Chemical Reactions Table of Contents Book L Ch

Chemical Reactions Table of Contents – Book L Ch 5. 1 Observing Chemical Change Ch 5. 2 Describing Chemical Reactions

Chemical Reactions Essential Question How is matter conserved in a chemical reaction?

Chemical Reactions Lesson 1 Objectives 1. Explain how changes in matter can be described. 2. Identify ways to tell that a chemical reaction has occurred.

Chemical Reactions Properties and Changes of Matter • Chemistry- the study of matter and how matter changes • Matter- anything that has mass and takes up space • Matter can be described in terms of two kinds of properties: 1. Physical properties- a characteristic of a substance that can be observed without changing the substance into another substance. • Ex. Melting point, freezing point, color, ability to dissolve in water, conducting heat and electricity

Chemical Reactions Properties and Changes of Matter • Chemical properties- a characteristic of a substance that describes its ability to change into other substances • To observe the chemical properties of a substance, you must change it to another substance. • Ex: Burning, tarnishing, rusting

Chemical Reactions - Observing Chemical Change Properties and Changes of Matter • Matter can undergo both physical change and chemical change.

Chemical Reactions Properties and Changes of Matter • Physical change - any change that alters the form or appearance of a substance but that does not make the substance into another substance • Ex: Squashing a marshmallow, freezing or melting water • Still the same substance, but in a changed form

Chemical Reactions Properties and Changes of Matter • Chemical change (chemical reaction) - a change in matter that produces one or more new substances • Ex: Burning of gasoline car exhaust Burning wood ashes
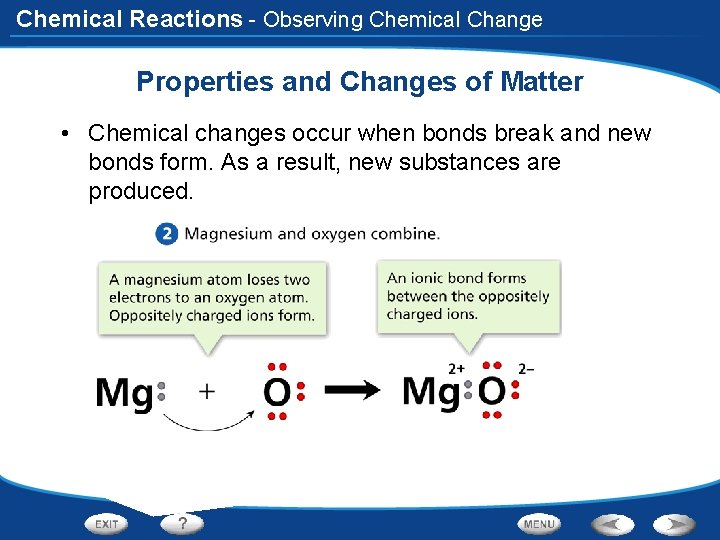
Chemical Reactions - Observing Chemical Change Properties and Changes of Matter • Chemical changes occur when bonds break and new bonds form. As a result, new substances are produced.

Chemical Reactions Evidence for Chemical Reactions • Chemical reactions involve two main kinds of changes that you can observe 1. Formation of new substances - color changes - a solid may appear - a gas might be produced from solids or liquids - other observable changes Ex. soft dough baking to form bread • Precipitate- a solid that forms from solution during a chemical reaction

Chemical Reactions Evidence for Chemical Reactions 2. Changes in Energy • As matter changes, it can either absorb or release energy. One common indication of this energy transfer is a change in temperature. • Endothermic reaction - a reaction in which energy is absorbed • Ex: an egg absorbing heat on a frying pan baking soda absorbing heat from vinegar making the solution feel cooler

Chemical Reactions Evidence for Chemical Reactions • Exothermic reaction- a reaction that releases energy in the form of heat • Ex: reaction between fuel and oxygen in an airplane engine releasing energy
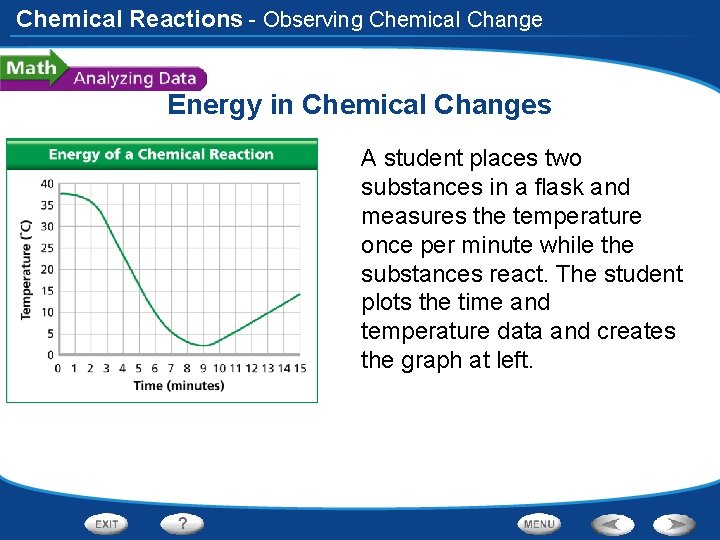
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes A student places two substances in a flask and measures the temperature once per minute while the substances react. The student plots the time and temperature data and creates the graph at left.
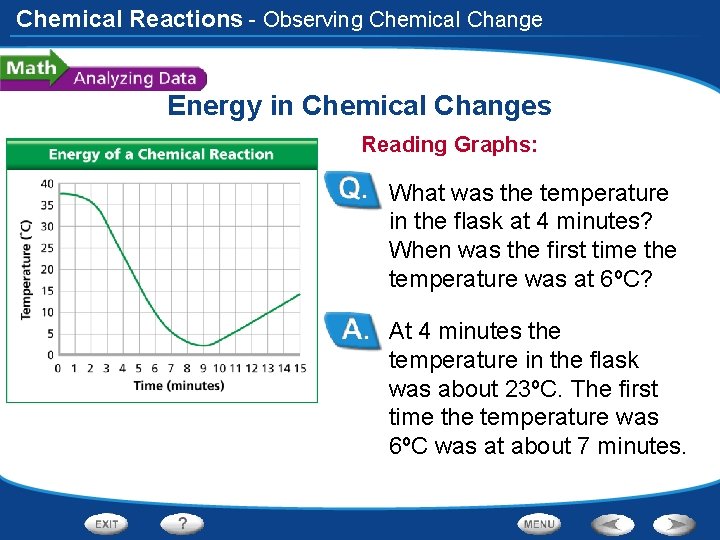
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Reading Graphs: What was the temperature in the flask at 4 minutes? When was the first time the temperature was at 6ºC? At 4 minutes the temperature in the flask was about 23ºC. The first time the temperature was 6ºC was at about 7 minutes.
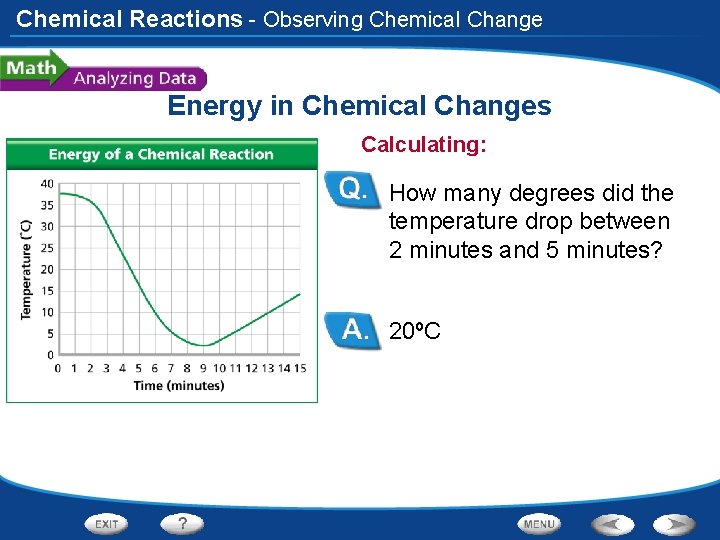
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Calculating: How many degrees did the temperature drop between 2 minutes and 5 minutes? 20ºC
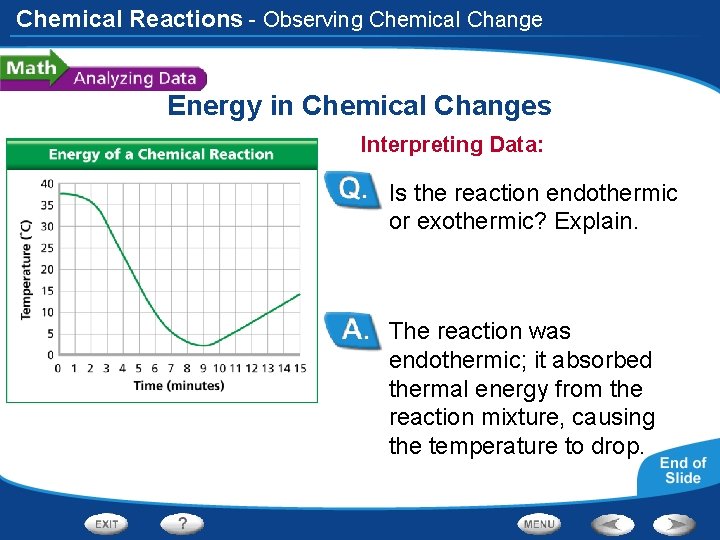
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Interpreting Data: Is the reaction endothermic or exothermic? Explain. The reaction was endothermic; it absorbed thermal energy from the reaction mixture, causing the temperature to drop.
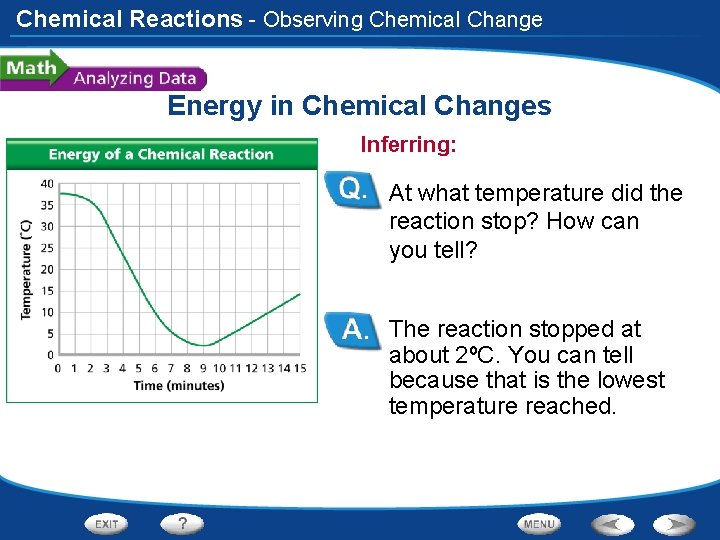
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Inferring: At what temperature did the reaction stop? How can you tell? The reaction stopped at about 2ºC. You can tell because that is the lowest temperature reached.
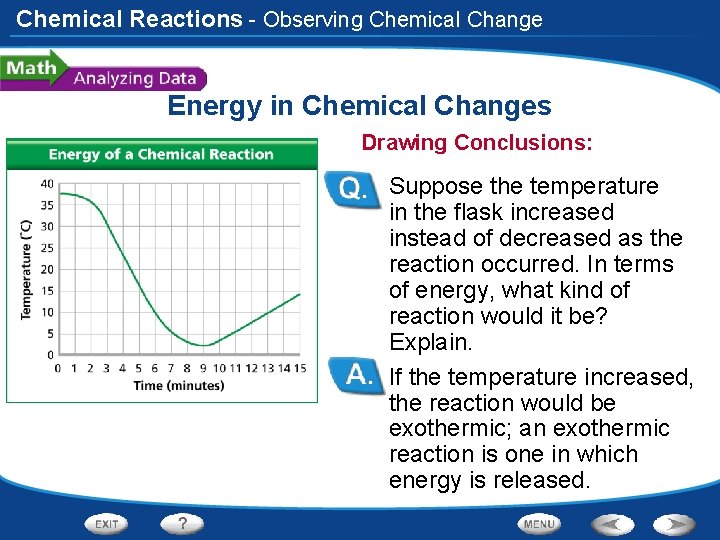
Chemical Reactions - Observing Chemical Change Energy in Chemical Changes Drawing Conclusions: Suppose the temperature in the flask increased instead of decreased as the reaction occurred. In terms of energy, what kind of reaction would it be? Explain. If the temperature increased, the reaction would be exothermic; an exothermic reaction is one in which energy is released.

Chemical Reactions Comparing Properties

Chemical Reactions Lesson 2 Objectives 1. Identify the information included in a chemical equation. 2. Explain how mass is conserved during a chemical reaction. 3. Identify the three categories of chemical reactions.

Chemical Reactions - Describing Chemical Reactions • Cellular phone messages make use of symbols and abbreviations to express ideas in shorter form. Similarly, chemists often use chemical equations in place of words.
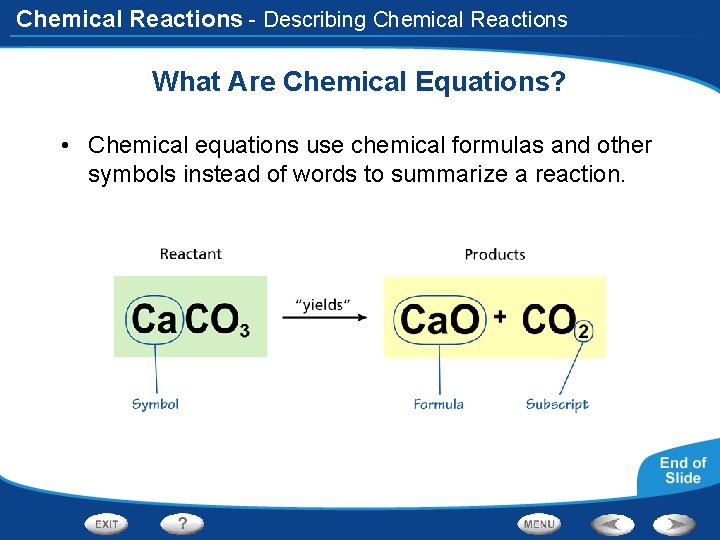
Chemical Reactions - Describing Chemical Reactions What Are Chemical Equations? • Chemical equations use chemical formulas and other symbols instead of words to summarize a reaction.

Chemical Reactions - Describing Chemical Reactions What Are Chemical Equations? • The formula of a compound identifies the elements in the compound and the ratios in which their atoms are present. • For example: CO 2 Must contain: 1 molecule of carbon (C) 2 molecules of oxygen (O)

Chemical Reactions What Are Chemical Equations? • Chemical equation - tells you the substances you start with in a reaction and the substances you get at the end. • Reactants- refers to the substances you have at the beginning of a reaction • Products- refers to the new substances created when the reaction is complete • Reactant + Reactant Product + Product • Read the arrow as “yields”

Chemical Reactions What Are Chemical Equations? • The formulas for the reactants are usually written on the left followed by an arrow. • The formulas for the products are written on the right. • Plus signs are used to separate two or more products and reactants. • Reactant + Reactant Product + Product C 6 H 12 O 6 + O 2 CO 2 + H 2 O + ATP (Energy) Chemical Formula for: Cellular Respiration

Chemical Reactions - Describing Chemical Reactions Conservation of Mass • Conservation of mass - states that in a chemical reaction, the total mass of the reactants must equal the total mass of the products. Iron and Sulfur yields Iron Sulfide

Chemical Reactions Conservation of Mass • During a chemical reaction, matter is not created or destroyed. • (all the atoms present at the start of the reaction are present at the end) • Sometimes difficult to measure ail the matter involved in a reaction. Ex: When burning a match, oxygen comes from the surrounding air and products escape into the air.

Chemical Reactions Conservation of Mass Open system matter can enter from or escape to the surroundings Ex: A burning match Closed system matter is not allowed to enter or leave Ex: Chemical reaction inside a sealed plastic bag • To measure all the matter before and after a reaction, you have to be able to contain it.
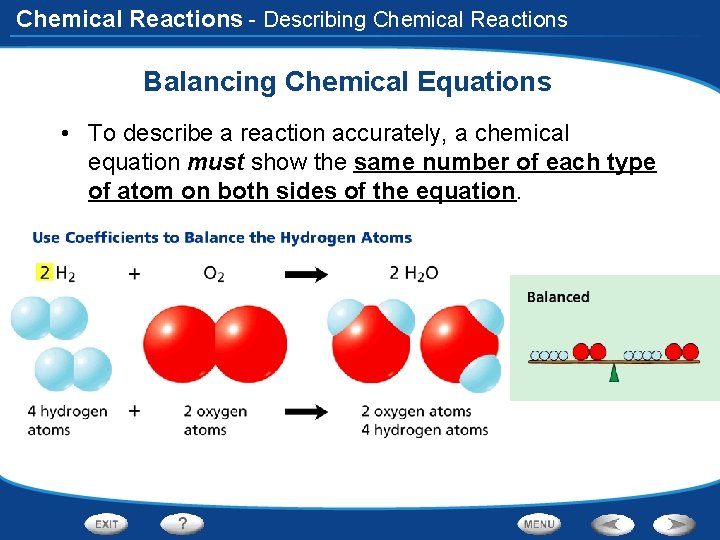
Chemical Reactions - Describing Chemical Reactions Balancing Chemical Equations • To describe a reaction accurately, a chemical equation must show the same number of each type of atom on both sides of the equation.

Chemical Reactions Balancing Chemical Equations How to Write a Balanced Chemical Equation: 1. Write the Equation • Place the reactants on the left side of the arrow separated by a plus sign, then write the product on the right side of the arrow. H 2 + O 2 H 2 O

Chemical Reactions Balancing Chemical Equations 2. Count the atoms • Count the number of atoms of each element on each side of the equation. You find two atoms of oxygen in the reactants, but only one atom of oxygen in the products. H 2 + O 2 H 2 O 2 Hydrogen 2 Oxygen 1 Oxygen 2 Hydrogen

Chemical Reactions Balancing Chemical Equations 3. Use Coefficients to Balance Atoms • Coefficient- a number placed in front of a chemical formula in an equation • Tells you how many atoms or molecules of a reactant or a product take part in the reaction. • If the coefficient is 1, you do not need to write it. H 2 + O 2 2 H 2 O

Chemical Reactions Balancing Chemical Equations 2 Hydrogen 2 Oxygen • H 2 + O 2 2 H 2 O 2 Oxygen 4 Hydrogen There are now 2 hydrogen atoms in the reactants and four in the product. How can you balance the hydrogen? • Double the number of hydrogens in the reactants by writing the coefficient 2 for hydrogen. 4 Hydrogen 2 Oxygen 2 H 2 + O 2 2 H 2 O Balanced! 2 Oxygen 4 Hydrogen

Chemical Reactions Balancing Chemical Equations 2 H 2 + O 2 2 H 2 O 4. Look Back and Check • The equation is balanced: Two molecules of hydrogen react with one molecule of oxygen to yield two molecules of water.

Chemical Reactions - Describing Chemical Reactions Classifying Chemical Reactions • Many chemical reactions can be classified in one of three categories: synthesis, decomposition, or replacement.

Chemical Reactions Classifying Chemical Reactions • Synthesize - means to put things together (to create) • In chemistry, when two or more elements or compounds combine to make a more complex substance, the process is called synthesis. • The reaction of hydrogen and oxygen to make water is a synthesis reaction.
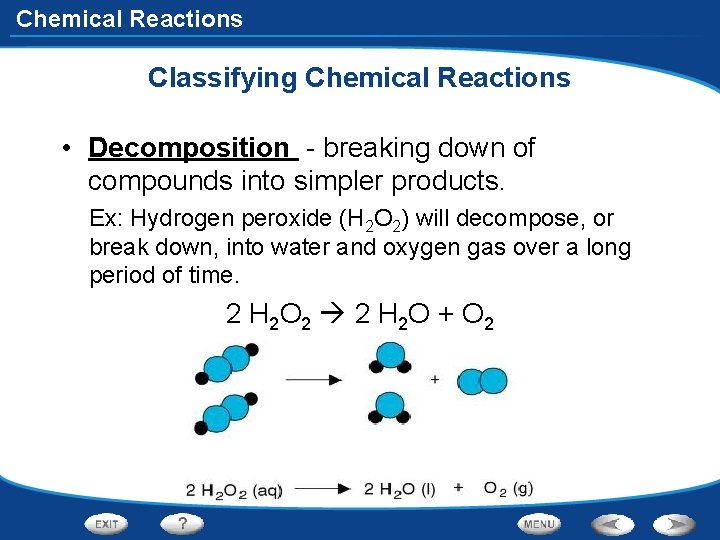
Chemical Reactions Classifying Chemical Reactions • Decomposition - breaking down of compounds into simpler products. Ex: Hydrogen peroxide (H 2 O 2) will decompose, or break down, into water and oxygen gas over a long period of time. 2 H 2 O 2 2 H 2 O + O 2

Chemical Reactions Classifying Chemical Reactions • Replacement – is when one element replaces another in a compound, • Also when two elements in different compounds trade places • Copper metal can be obtained by heating copper oxide with carbon. Carbon takes the place of copper. 2 Cu 2 O + C 4 Cu + CO 2

Chemical Reactions Classifying Chemical Reactions • Single replacement - one element replaces another element in the compound • Ex: 2 Cu 2 O + C 4 Cu + CO 2 • Double replacement - elements in one compound appear to “trade places” with elements in another compound. • Ex: Fe. S + 2 HCl Fe. Cl 2 + H 2 S
- Slides: 39