Chemical Reactions Synthesis and decomposition reactions were studied












- Slides: 13

Chemical Reactions

Synthesis and decomposition reactions were studied in the SNC 2 D course and reviewed earlier this year. This course has two additional types of synthesis reactions that are required learning: Synthesis and Decomposition Reactions A + B C and A B+C Reaction of Metal Oxides and Water When a metal oxide reacts with water, a basic solution results. This is because the product is a metal hydroxide which are a type of base. • Ca. O (s) + H 2 O (l) Ca(OH)2 (aq) • Li 2 O (s) + H 2 O (l) 2 Li. OH (aq) Reactions of Non-Metal Oxides with Water When a non metal oxide reacts with water, an acid is formed. For example: SO 3 (g) + H 2 O (l) H 2 SO 4 (aq) this is sulphuric acid – it is a very strong acid

Single Displacement Reactions One element displaces or replaces an element in a compound The Activity Series - a table of the reactivity of metals relative to each other. The most reactive metals are at the top of the activity series. The least reactive metals are found at the bottom. Hydrogen is included in the series, even though it is not a metal, because it forms a positively charged ion like a metal. The activity series is based on two important generalizations: • One element can displace elements below it from compounds in solution but cannot displace elements above it. • The farther apart two elements are, the more likely it is that the displacement will occur rapidly.

Single Displacement Reactions and the Metal Activity Series A + BC B + AC There are 3 types of single displacement reactions that involve metals: 1. Reactions involving a Metal and an Ionic Compound Examples: Mg(s) + Cu. SO 4(aq) Cu(s) + Mg. SO 4 (aq) Pb(s) + Zn(NO 3)2(aq) No reaction 2. Reactions involving a Metal and an Acid For example: Li(s) + H 2 SO 4(aq) H 2(g) + Li 2 SO 4 (aq) The reaction between Li and H 2 SO 4 occurs because: Li is more reactive than H 2 The reaction between Mg and Cu. SO 4 occurs because: Mg is more reactive than Cu 3. Reactions between a Metal and Water For example: Na(s) + H 2 O(l) H 2(g) + 2 Na. OH (aq) The reaction between Pb and Zn(NO 3)2 does not occur because: Pb is less reactive than Zn The reaction between Na and H 2 O occurs because: Na is more reactive than H 2

Single Displacement Reactions Involving Halogens A + BC BA + C The elements of the halogen family have their own activity series. The single displacement pattern for halogens is slightly different from the pattern used for the metals. This is because a negative ion (anion) is being displaced rather than a positive ion (cation). F 2 Examples: I 2 Cl 2(g) + 2 KI(aq) I 2(s) + 2 KCl (aq) This reaction occurs because: Cl 2 is more reactive than I 2(g) + Ca. Br 2(aq) No reaction This reaction does not occur because: I 2 is less reactive than Br 2 Cl 2 Br 2

Combustion of Hydrocarbons • Complete combustion of hydrocarbons: there is enough oxygen present to make H 2 O and CO 2 • Incomplete combustion of hydrocarbons: there is not enough oxygen to make CO 2 exclusively, so the products are H 2 O and a mixture of CO and CO 2. For example: Write the balanced chemical equation for: • The incomplete combustion of C 3 H 8 (assume no CO 2 is made) 2 C 3 H 8 + 7 O 2 6 CO + 8 H 2 O • The complete combustion of C 4 H 10 2 C 4 H 10 + 13 O 2 8 CO 2 + 10 H 2 O

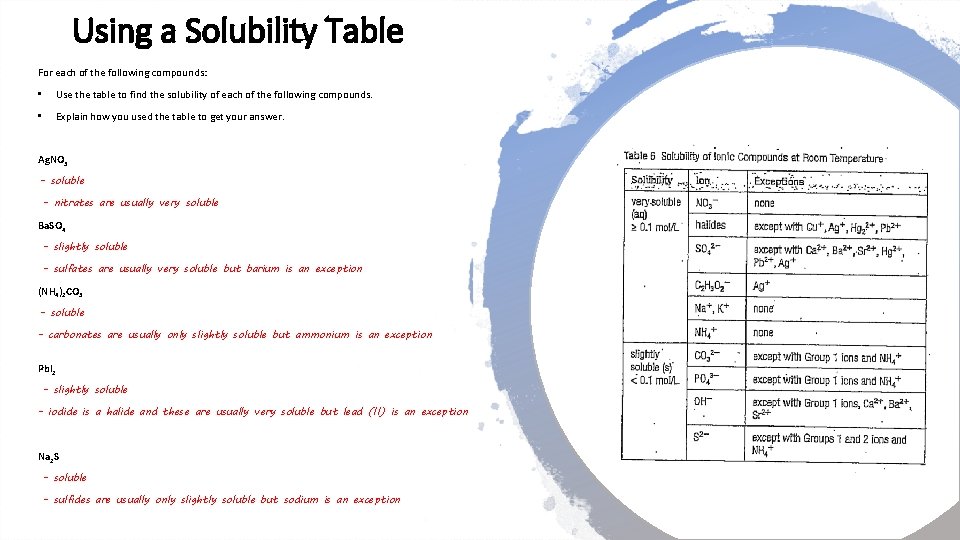
Using a Solubility Table For each of the following compounds: • Use the table to find the solubility of each of the following compounds. • Explain how you used the table to get your answer. Ag. NO 3 - soluble - nitrates are usually very soluble Ba. SO 4 - slightly soluble - sulfates are usually very soluble but barium is an exception (NH 4)2 CO 3 - soluble - carbonates are usually only slightly soluble but ammonium is an exception Pb. I 2 - slightly soluble - iodide is a halide and these are usually very soluble but lead (II) is an exception Na 2 S - soluble - sulfides are usually only slightly soluble but sodium is an exception

Compounds switch their cation and anion pairs. Double Displacement Reactions Double displacement reactions will only occur if: AB + CD AD + CB 2. A gas is formed. 1. A slightly soluble solid product (precipitate) is formed. or or 3. A molecular compound (usually water) is formed.

Reactions that produce a precipitate • https: //javalab. org/en/precipitation_reaction_en/

Reactions that produce a precipitate How do you know if a product will form a precipitate? - Look up each of them up on the solubility table. - If one of them is only slightly soluble, a precipitate is formed Examples: Do the following reactions proceed? If so, determine the products and balance the equation. Ag. NO 3(aq) + KCl(aq) Ag. Cl (s) + KNO 3 (aq) The reaction between Ag. NO 3 and KCl happens because: K 2 SO 4(aq) + Fe. Cl 3 (aq) A precipitate Ag. Cl (s) is formed No reaction The reaction between K 2 SO 4 and Fe. Cl 3 does not happen because: Both products are very soluble, so no precipitate is formed

Reactions that produce a gas Sometimes a gaseous product is formed during a double displacement reaction. When it is formed, it escapes into the air as bubbles of gas. A common gaseous product is H 2 S(g). It is formed when an acid reacts with a soluble sulfide such as sodium sulfide. Na 2 S(aq) + 2 HCl(aq) 2 Na. Cl (aq) + H 2 S (g)

Reactions that produce a gas Example 1: carbonic acid produced, then decomposes • Gases are also produced when an unstable product of a double displacement reaction decomposes. • This is a two step process. The common unstable products to be watching for are: H 2 CO 3 (carbonic acid) that decomposes into H 2 O and CO 2(g), and H 2 SO 3 (sulfurous acid) that decomposes into H 2 O and SO 2(g). Mg. SO 4(aq) + H 2 O(l) + CO 2(g) Overall: Mg. CO 3 (aq) + H 2 SO 4 (aq) Step 1: Mg. CO 3 (aq) + H 2 SO 4 (aq) Mg. SO 4(aq) + H 2 CO 3(aq) Step 2: H 2 CO 3(aq) H 2 O(l) + CO 2(g) Example 2: sulfurous acid produced, then decomposes Overall: Mg. SO 3 (aq) + HBr(aq) Mg. Br(aq) + H 2 O(l) + SO 2(g) Step 1: Mg. SO 3 (aq) + HBr(aq) Mg. Br(aq) + H 2 SO 3(aq) Step 2: H 2 SO 3(aq) H 2 O(l) + SO 2(g)

Reactions that produce a molecular compound Neutralization reaction: An acid reacts with a base to form salt and water. For example: HNO 3 (aq) + Na. OH (aq) Na. Cl(aq) + H 2 O(l) The molecular compound H 2 O is formed, so this reaction occurs https: //javalab. org/en/neutralization_reaction_model_en/