Chemical Reactions Reactions involve chemical changes in matter
































- Slides: 36

Chemical Reactions • Reactions involve chemical changes in matter resulting in new substances • Reactions involve rearrangement and exchange of atoms to produce new molecules Reactants Products 1

Evidence of Chemical Change Formation of a Gas Color Change 2 Emission of Light

Evidence of Chemical Change Formation of Precipitate ______ (solid formed from two liquids mixed) Release or Absorption of Heat 3

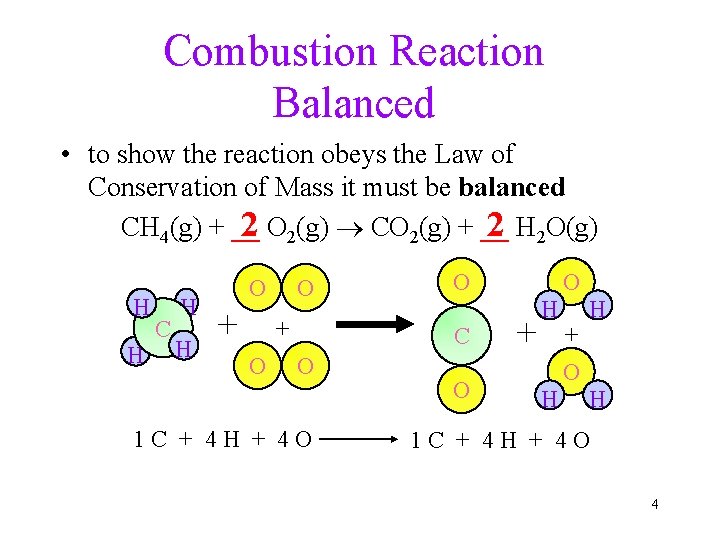
Combustion Reaction Balanced • to show the reaction obeys the Law of Conservation of Mass it must be balanced 2 O 2(g) CO 2(g) + __ 2 H 2 O(g) CH 4(g) + __ H H C H H O + O O C O 1 C + 4 H + 4 O O + H H O + O H H 1 C + 4 H + 4 O 4

Chemical Equations CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) • CH 4 and O 2 are the reactants, and CO 2 and H 2 O are the products • the (g) after the formulas tells us the state of the chemical • the number in front of each substance tells us the numbers of those molecules in the reaction coefficients ücalled the ______ 5

Symbols Used in Equations • symbols used to indicate state liquid (s) = _____ solid ü(g) = gas ____; (l) = _______; aqueous = dissolved in water ü(aq) = _____ • energy symbols used above the arrow for decomposition reactions heat ü D = _____ ü hn = light _____ 6

Combustion Reactions • Reactions in which O 2(g) is a reactant are called Combustion Reactions • Combustion reactions release lots of energy 2 C 8 H 18(g) + 25 O 2(g) 16 CO 2(g) + 18 H 2 O(g) 7

Classifying Reactions • another scheme classifies reactions by what the atoms do Type of Reaction Synthesis General Equation A + B AB Decomposition AB A + B Displacement A + BC AC + B Double Displacement AB + CD AD + CB 8

Synthesis Reactions • also known as Composition or Combination reactions • two (or more) reactants combine together to one product make ____ üsimpler substances combining together 2 CO + O 2 2 CO 2 2 Mg + O 2 2 Mg. O 9

Decomposition Reactions • a large molecule is broken apart into smaller molecules or its elements ücaused by addition of energy into the molecule • have only one reactant, make 2 or more products Decomposition of Water 10

Displacement Reactions • also known as single-displacement reactions • reactions that involve one anion being transferred from one cation to another X+ Y- + A X + A + YZn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) Fe 2 O 3(s) + Al(s) Fe(s) + Al 2 O 3(s) 2 Na(s) + 2 H 2 O(aq) 2 Na. OH(aq) + H 2(g) 11

Double Displacement Reactions • two ionic compounds exchange ions • may be followed by decomposition of one of the products to make a gas • X +Y- (aq) + A+B- (aq) XB + AY • precipitation, acid-base and gas-evolving reactions are also double displacement reactions 12

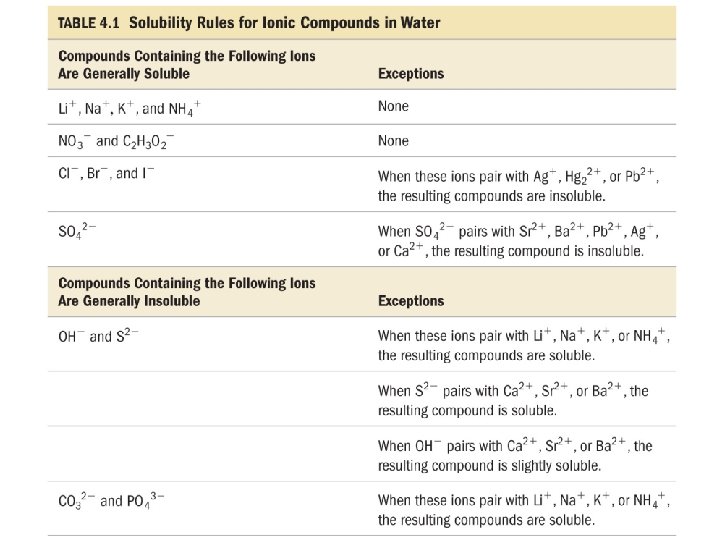
Dissociation • when ionic compounds dissolve in water, the anions and cations are separated from each other - this is called _______ dissociation ü however not all ionic compounds are soluble in water! • when compounds containing polyatomic ions dissociate, the polyatomic group stays together as one ion 13

When will a Salt Dissolve? • a compound is soluble in a liquid if it dissolves in that liquid ü Na. Cl is soluble in water, but Ag. Cl is not • a compound is insoluble if a significant amount does not dissolve in that liquid ü Ag. Cl is insoluble in water Ø though there is a very small amount dissolved, but not enough to be significant 14


Determine if Each of the Following is Soluble in Water • • • KOH ______ soluble Ag. Br insoluble _______ soluble Ca. Cl 2 ______ soluble Pb(NO 3)2 ______ Pb. SO 4 insoluble _______ 16

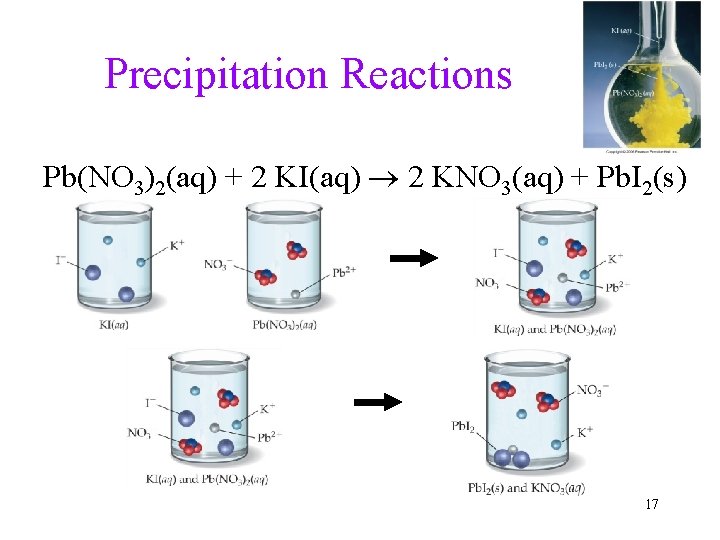
Precipitation Reactions Pb(NO 3)2(aq) + 2 KI(aq) 2 KNO 3(aq) + Pb. I 2(s) 17

No Precipitate Formation = No Reaction KI(aq) + Na. Cl(aq) KCl(aq) + Na. I(aq) all ions still present, no reaction (no rxn) 18

Ionic Equations • equations which describe the complete formulas of all equations reactant and products are called molecular __________ 2 KOH(aq) + Mg(NO 3)2(aq) 2 KNO 3(aq) + Mg(OH)2(s) • equations which describe the actual dissolved species are complete ionic equations called ____________ ü aqueous electrolytes are written as ions Ø soluble salts, strong acids, strong bases ü insoluble substances and nonelectrolytes written in molecule form Ø solids, liquids and gases are not dissolved, therefore molecule form 2 K+(aq) + 2 OH-(aq) + Mg 2+(aq) + 2 NO 3 -(aq) 2 K+(aq) + 2 NO 3 -(aq) + Mg(OH)2(s) 19

Ionic Equations • ions that do not participate directly in a reaction in solution spectator ions are called _______ 2 K+(aq) + 2 OH-(aq) + Mg 2+(aq) + 2 NO 3 -(aq) 2 K+(aq) + 2 NO 3 -(aq) + Mg(OH)2(s) • an ionic equation in which the spectator ions are ionic equation removed is called a net _________ 2 OH-(aq) + Mg 2+(aq) Mg(OH)2(s) 20

Example – Write the molecular, complete ionic and net-ionic equation for the reaction of aqueous nitric acid with aqueous calcium hydroxide Molecular Equation: 2 HNO 3(aq) + Ca(OH)2(aq) Ca(NO 3)2(aq) + 2 H 2 O(l) Complete Ionic Equation: 2 H+(aq) + 2 NO 3 -(aq) + Ca 2+(aq) + 2 OH-(aq) Ca 2+(aq) + 2 NO 3 -(aq) + 2 H 2 O(l) Net Ionic Equation: H+(aq) + OH-(aq) H 2 O(l) 21

Acid-Base Reactions • also called neutralization reactions because the acid and base neutralize each others properties • in the reaction of an acid with a base, the H+ from the acid combines with the OH- from the base to make water • the cation from the base combines with the anion from the acid to make the salt acid + base salt + water 2 HNO 3(aq) + Ca(OH)2(aq) Ca(NO 3)2(aq) + 2 H 2 O(l) 22

Gas Evolving Reactions • Some reactions form a gas directly from the ion exchange K 2 S(aq) + H 2 SO 4(aq) K 2 SO 4(aq) + H 2 S(g) • Other reactions form a gas by the decomposition of one of the ion exchange products into a gas and water K 2 SO 3(aq) + H 2 SO 4(aq) K 2 SO 4(aq) + H 2 SO 3(aq) H 2 SO 3 H 2 O(l) + SO 2(g) K 2 SO 3(aq) + H 2 SO 4(aq) K 2 SO 4(aq) + H 2 O(l) + SO 2(g) 23

Compounds that Undergo Gas Evolving Reactions Reactant Type Reacting Ion Exchange With Product Decompose ? Gas Formed Example metaln. S, metal HS acid H 2 S no H 2 S K 2 S(aq) + 2 HCl(aq) 2 KCl(aq) + H 2 S(g) metaln. CO 3, metal HCO 3 acid H 2 CO 3 yes CO 2 K 2 CO 3(aq) + 2 HCl(aq) 2 KCl(aq) + CO 2(g) + H 2 O(l) metaln. SO 3 metal HSO 3 acid H 2 SO 3 yes SO 2 K 2 SO 3(aq) + 2 HCl(aq) 2 KCl(aq) + SO 2(g) + H 2 O(l) (NH 4)nanion base NH 4 OH yes NH 3 KOH(aq) + NH 4 Cl(aq) KCl(aq) + NH 3(g) + H 2 O(l) 24

Oxidation-Reduction Reactions • oxidation-reduction reactions are also called redox reactions • all redox reactions involve the transfer of electrons from one atom to another • atoms that lose electrons are being oxidized, atoms that gain electrons are being reduced Ger 2 Na(s) + Cl 2(g) → 2 Na+Cl–(s) Na → Na+ + 1 e– oxidation Cl 2 + 2 e– → 2 Cl– reduction Leo 25

Oxidation–Reduction • oxidation and reduction must occur simultaneously ü if an atom loses electrons another atom must take them • the reactant that reduces an element in another reactant reducing agent is called the _______ ü the reducing agent contains the element that is oxidized • the reactant that oxidizes an element in another reactant oxidizing agent is called the _______ ü the oxidizing agent contains the element that is reduced 2 Na(s) + Cl 2(g) → 2 Na+Cl–(s) Na is oxidized, Cl is reduced Na is the reducing agent, Cl 2 is the oxidizing agent 26

Practice – Identify the Element being Oxidized and Element being Reduced; and the Oxidizing and Reducing Agents • 2 Mg(s) + O 2(g) → 2 Mg. O(s) Mg is oxidized, O is reduced Mg is the reducing agent, O 2 is the oxidizing agent • Fe(s) + Cl 2(g) → Fe. Cl 2(s) Fe is oxidized, Cl is reduced Fe is the reducing agent, Cl 2 is the oxidizing agent • Zn(s) + Fe 2+(aq) → Zn 2+(aq) + Fe(s) Zn is oxidized, Fe is reduced Zn is the reducing agent, Fe 2+ is the oxidizing agent 27

Electron Bookkeeping • for reactions that are not metal + nonmetal, or do not involve O 2, we need a method for determining how the electrons are transferred • chemists assign a number to each element in a oxidation state that allows them reaction called an _______ to determine the electron flow in the reaction ü even though they look like them, oxidation states are not ion charges! Øoxidation states are imaginary charges assigned based on a set of rules Øion charges are real, measurable charges 28

Rules for Assigning Oxidation States • rules are in order of priority 1. free elements have an oxidation state = 0 ü Na = 0 and Cl 2 = 0 in 2 Na(s) + Cl 2(g) 2. monatomic ions have an oxidation state equal to their charge ü Na = +1 and Cl = -1 in Na+ & Cl- 3. (a) the sum of the oxidation states of all the atoms in a compound is 0 ü Na = +1 and Cl = -1 in Na. Cl, (+1) + (-1) = 0 29

Rules for Assigning Oxidation States 3. (b) the sum of the oxidation states of all the atoms in a polyatomic ion equals the charge on the ion ü N = +5 and O = -2 in NO 3–, (+5) + 3(-2) = -1 4. (a) Group I metals have an oxidation state of +1 in all their compounds ü Na = +1 in Na. Cl (b) Group II metals have an oxidation state of +2 in all their compounds ü Mg = +2 in Mg. Cl 2 30

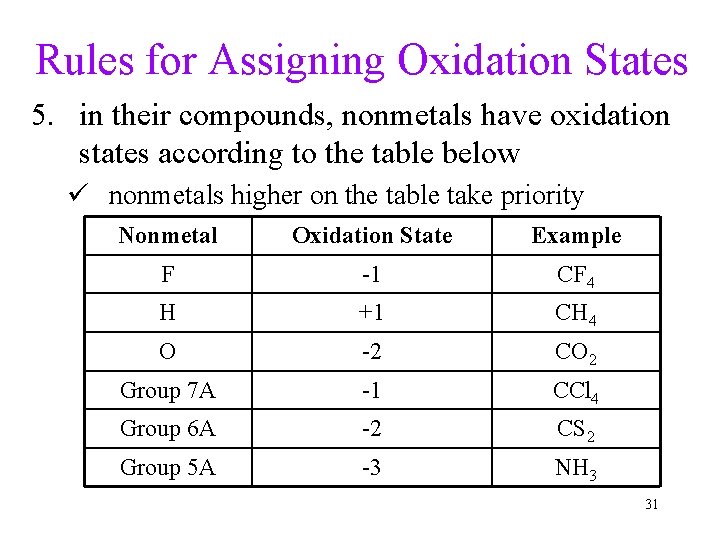
Rules for Assigning Oxidation States 5. in their compounds, nonmetals have oxidation states according to the table below ü nonmetals higher on the table take priority Nonmetal Oxidation State Example F -1 CF 4 H +1 CH 4 O -2 CO 2 Group 7 A -1 CCl 4 Group 6 A -2 CS 2 Group 5 A -3 NH 3 31

Practice – Assign an Oxidation State to each Element in the following • Br 2 Br = 0, (Rule 1) • K+ • Li. F K = +1, (Rule 2) Li = +1, (Rule 4 a) & F = -1, (Rule 5) • CO 2 O = -2, (Rule 5) & C = +4, (Rule 3 a) • SO 42 - O = -2, (Rule 5) & S = +6, (Rule 3 b) • Na 2 O 2 Na = +1, (Rule 4 a) & O = -1, (Rule 3 a) 32

Oxidation and Reduction • oxidation occurs when an atom’s oxidation increases during a reaction state ____ • reduction occurs when an atom’s oxidation state decreases ____ during a reaction CH 4 + 2 O 2 → CO 2 + 2 H 2 O -4 +1 0 +4 – 2 +1 -2 oxidation reduction 33

Listing of Metals by Reactivity 34

Predict the Product • Mg(s) + Cu 2+(aq) Mg 2+(aq) + Cu(s) Mg(s) Mg 2+(aq) + 2 e. Cu 2+(aq) + 2 e- Cu(s) Mg(s) + Cu 2+(aq) Mg 2+(aq) + Cu(s) • Cu(s) + Mg 2+(aq) No Reaction • Zn(s) + 2 H+(aq) Zn 2+(aq) + H 2(g) Zn(s) Zn 2+(aq) + 2 e 2 H+(aq) + 2 e- H 2(g) Zn(s) + 2 H+(aq) Zn 2+(aq) + H 2(g) 35

Mg will react with Cu 2+ to form Mg 2+ and Cu metal Mg is above Cu on the Activity Series but Cu will not react with Mg 2+ 36