Chemical Reactions Physical vs Chemical What is the

















- Slides: 20

Chemical Reactions

Physical vs. Chemical �What is the difference between a chemical and physical property?

Physical Change �Physical change: a change that occurs that does not change the identity of the substance �Melting ice �Freezing Kool-aid �Tearing paper �Boiling water

Chemical Changes �Chemical change: a change that occurs causing the identity of the substance to change �Burning paper �Digesting food �Electrolysis of water �A chemical change is called a chemical reaction

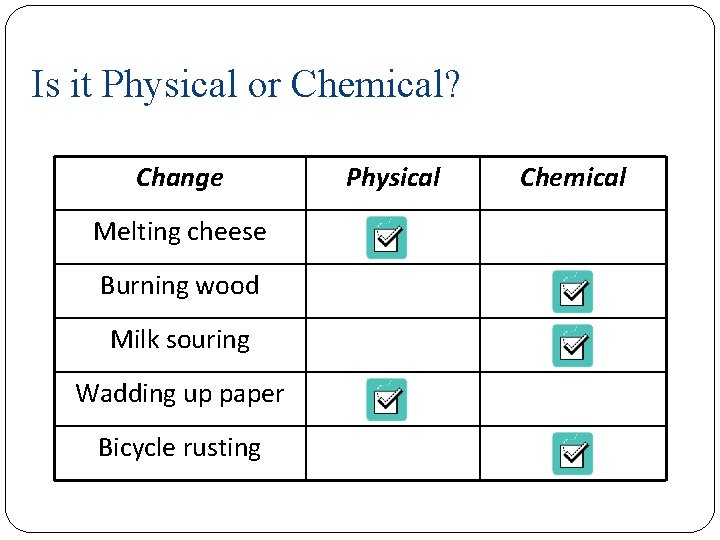
Is it Physical or Chemical? Change Melting cheese Burning wood Milk souring Wadding up paper Bicycle rusting Physical Chemical

Chemical Reactions �A chemical reaction produces new substances by changing the way in which atoms are arranged. �Bonds between atoms are broken and new bonds form between different atoms �The new arrangements of atoms form different substances

Reactants and Products �Reactants are the substances present at the beginning of a chemical reaction. �Products are the substances formed by a chemical reaction �Reactant + Reactant Product �The mass of the reactants is always equal to the mass of the products

Detecting a Chemical Reaction 1. 2. 3. 4. 5. Color Change Formation of a Precipitate Formation of a Gas Temperature Change Evolution of Light

Color Change �Substance often change color during a chemical reaction. �Example: when gray iron rusts, the product that forms is brown.

Formation of a Precipitate �Precipitate: A solid product formed when chemicals in two liquids react. �Example: Seashells are formed when sea creatures release a liquid that reacts with seawater.

Formation of a Gas �Chemical reactions may produce a gas, like that often formed when antacid pills are mixed with excess stomach acid.

Temperature Change �Most chemical reactions involve temperature change �Example: Concrete gets warmer as it hardens

Evolution of Light �Energy can be released in the form of light during a chemical reaction �Example—Glow Sticks

Endothermic and Exothermic Reactions �Exothermic Reactions: If more energy is released when the products form than is needed to break the bonds in the reactants then energy is released during the reaction �Energy is released �Endothermic Reactions: If more energy is required to break the bonds in the reactants than is released when the product is formed then energy must be added to the reaction �Energy is absorbed

Chemical Equations

Chemical Equation � Shorthand form for writing what reactants are used and what products are formed in a chemical reaction � Sometimes shows whether energy is produced or absorbed � Example: C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy

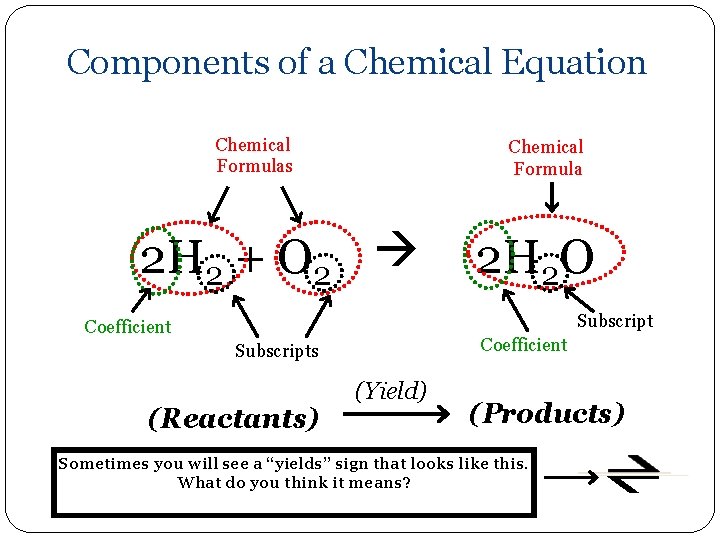
Components of a Chemical Equation Chemical Formulas Chemical Formula 2 H 2 + O 2 2 H 2 O Subscript Coefficient Subscripts (Reactants) (Yield) (Products) Sometimes you will see a “yields” sign that looks like this. What do you think it means?

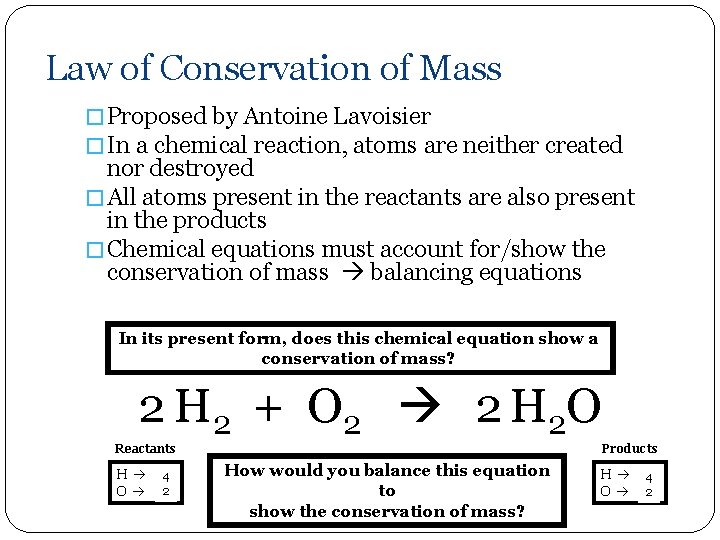
Law of Conservation of Mass � Proposed by Antoine Lavoisier � In a chemical reaction, atoms are neither created nor destroyed � All atoms present in the reactants are also present in the products � Chemical equations must account for/show the conservation of mass balancing equations In its present form, does this chemical equation show a conservation of mass? 2 H 2 + O 2 2 H 2 O Reactants H 24 O 22 Products How would you balance this equation to show the conservation of mass? H 24 O 12

Hints For Balancing Equations � Count the atoms �List the number of atoms of each element to see which elements must be balanced � Use a coefficient to add atoms to one side of the equation �Start with the reactant or product that has the greatest number of different elements � Add a coefficient to another reactant or product �Make sure that the coefficients in your balanced equation are the smallest whole numbers possible (they should have no common factor other than one)

Tutorial on Balancing Equations �http: //users. wfu. edu/ylwong/balanceeq/balanceq. html