Chemical Reactions Part 1 Balancing Chemical Equations Unit

Chemical Reactions – Part 1 Balancing Chemical Equations Unit 7: Stoichiometry - Part III Mrs. Callender

Lesson Essential Question… How do I balance a chemical equation?

Parts of a Chemical Reaction 2 Na. Cl(aq) + Ag 2 SO 4(aq) Na 2 SO 4(aq) + 2 Ag. Cl(s) States of Matter ? ? ? COEFFICIENTS (aq) – aqueous State of matter ? ? ? the reaction. (s) – solid (ppt or solid)after States of matter at room temperature ? ? ? are Reactants ? ? ? Yields ? ? ? These numbers from the? ? ? equation. Use solubility rules determine state. (l)- liquid (ex. H 2 balancing 0, Br 2)to Products before reaction They are the numberstarted. of “moles” of each (g) – gas (ex. H 2, O 2) compound indicating the ratio of each reactant to product formed.

Think back to the Law of Conservation of Matter What did it say? “No matter can be created nor destroyed in an ordinary chemical reaction. ”
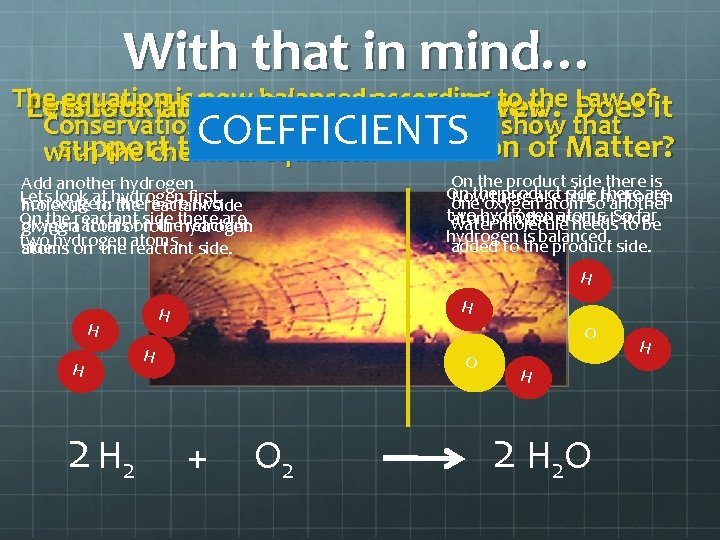
With that in mind… The equation now according to the Law Evaluate Lets lookthe atis it chemical onbalanced the molecular reaction below. level. Doesofit Conservation of Matter so how do we show that support the Law of Conservation of Matter? with the chemical equation? COEFFICIENTS On the product side there is On thethere product side hydrogen there are Now are four one oxygen atom so another two hydrogen atoms. So far atoms on the product water molecule needs side. to be hydrogen is balanced. added to the product side. Add another hydrogen Lets look at For oxygen: there arefirst. two molecule tohydrogen the reactant side On theareactant side are oxygen atoms thethere reactant giving total ofon four hydrogen two atoms. side. hydrogen atoms on the reactant H H H 2 O O H + O 2 H H O O H 2 O H
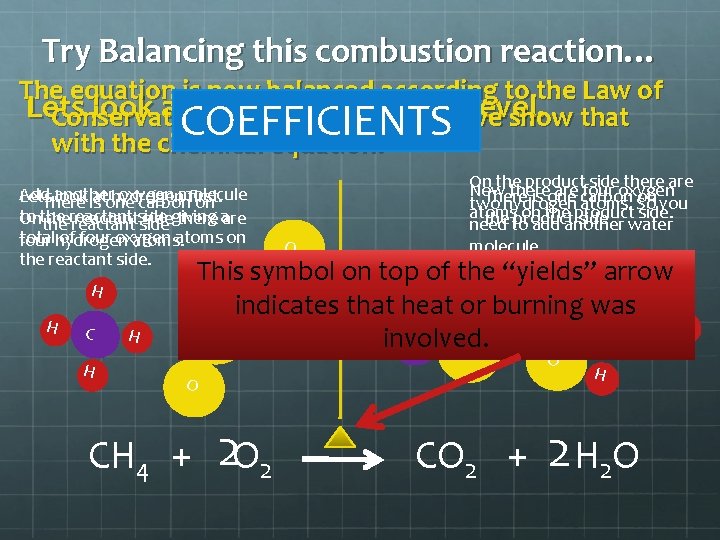
Try Balancing this combustion reaction… The equation is now balanced according to the Law of Lets look at itof on. Matter the molecular level. Conservation so how do we show that with the chemical equation? COEFFICIENTS Add another oxygen molecule Lets look is atone hydrogen first. There carbon on to the reactant side giving On the reactant side there aare total of four oxygen atoms on four hydrogen atoms. the reactant side. This symbol on top. Oof the “yields” arrow O indicates that heat or burning was. O H H O C involved. O H H C H O On the product sideoxygen there are Now there are four There is one carbon on you two hydrogen atoms. So atoms on the product side. the to product side water need add another molecule. H H O O CH 4 + 2 O 2 CO 2 + H 2 O

Try Balancing These Equations… 1. 2 Al 2. 2 Na. Cl. O 3 3. 2 BF 3 + 2 Al. N Fe 3 N 2 + 2 Na. Cl 3 Li 2 SO 3 4. 3 Sn(NO 2)4 5. B 2 Br 6 + + Pt 3 N 4 6 HNO 3 + + 3 Fe 3 O 2 B 2(SO 3)3 + Sn 3 N 4 + 6 LIF 3 Pt(NO 2)4 2 B(NO 3)3 + 6 HBr
- Slides: 7