Chemical Reactions https www youtube comwatch vCg WHbp
Chemical Reactions - + + - https: //www. youtube. com/watch? v=Cg. WHbp. MVQ 1 U 1

Chemical Reactions Learning Targets: I will be able to: 1. describe what happens to the elements in a chemical reaction. 2. review chemical change indicators such as precipitate, evolution of gas, color change, and absorption/release of heat. 3. describe how the rate of a reaction can be changed. 4. apply the Law of Conservation of Mass to chemical reactions. 2

Chemical Reaction • chemical reaction: the process by which a chemical change occurs • Atoms are rearranged, and chemical bonds are broken and reformed. • One or more substances change to produce one or more different substances. • Types of Reactions What does H 2 & O 2 gas create? 1. 2. 3. 4. Synthesis (combining) Decomposition (separating) Combustion (burning) Single, Displacement/Replacement (single switch) 5. Double Displacement/Replacement (double switch) Out of the five types of reactions, which would describe the chemical reaction taking place to the right? 3 H 2 O 2

Physical Change • physical change: a change in shape, size, color, or state without a change in chemical composition • Physical change is a change that is reversible. • Examples – tearing paper Physical changes are – cutting your hair not indicative of a chemical – change in state reaction. 4

Chemical Change • chemical change: a change in which a substance becomes another substance having different properties and chemical composition • Chemical changes are not reversible using ordinary physical means. • Chemical changes usually cause production of heat, sound, light, odor, fizzing/foaming, color changes. You usually need more than one of the above characteristics to be considered a chemical change! Examples mixing vinegar & baking soda burning a piece of wood milk 100% way to know a A chemical analysissoured is the only chemical change has occurred. 5
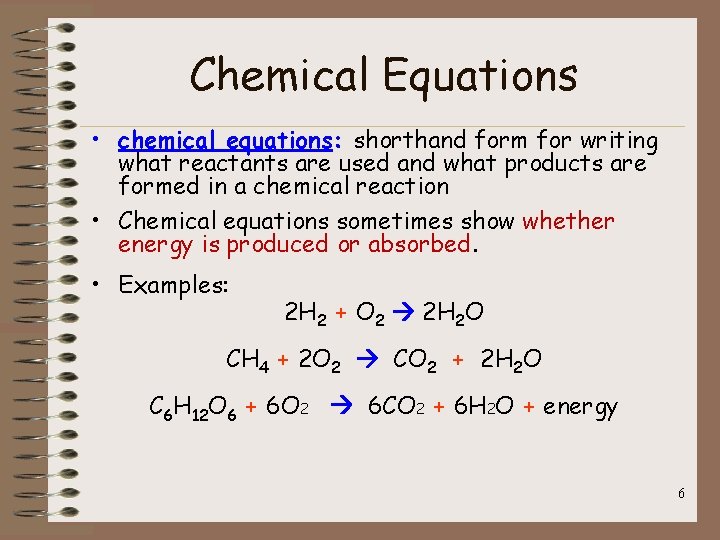
Chemical Equations • chemical equations: shorthand form for writing what reactants are used and what products are formed in a chemical reaction • Chemical equations sometimes show whether energy is produced or absorbed. • Examples: 2 H 2 + O 2 2 H 2 O CH 4 + 2 O 2 CO 2 + 2 H 2 O C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy 6

Chemical Equations • chemical formula: a combination of symbols that indicates the chemical composition of a substance • coefficient: the number in front of a chemical formula that tells us how many molecules of a given formula are present • subscript: a number after an element’s chemical symbol that tells us how many atoms there are within a compound 7
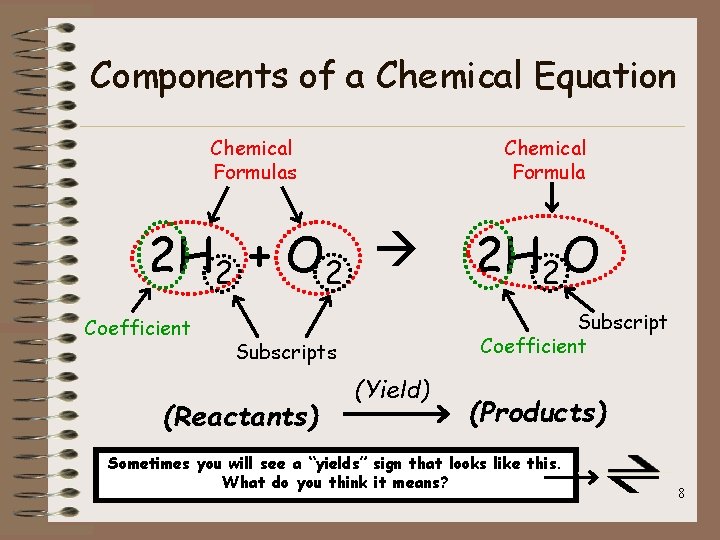
Components of a Chemical Equation Chemical Formulas Chemical Formula 2 H 2 + O 2 Coefficient Subscripts (Reactants) 2 H 2 O (Yield) (Products) Sometimes you will see a “yields” sign that looks like this. What do you think it means? 8

Energy and Chemical Reactions Exothermic Reaction Endothermic Reaction • A chemical reaction in which thermal energy is released. • The reactants have greater bond energy than the products. • A chemical reaction in which thermal energy is absorbed. • The products have greater bond energies than the reactants. C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy (respiration) 6 CO 2 + 6 H 2 O + energy C 6 H 12 O 6 + 6 O 2 (photosynthesis) Chemistry Comes Alive! Sample Movies Teacher. Tube - Videos Chemistry Demonstration Videos Can you think of other reactions where energy is gained or released? 9

Rates of Chemical Reactions The rates at which chemical reactions can take place are based on the interaction (collisions) between the different particles. These rates can be impacted by the following: • Temperature: a measure of the average kinetic energy of the particles in a sample of matter – Ex. Increasing the temperature when cooking • Surface area: amount of material that comes into contact with the reactants – Ex. Cutting a potato into smaller pieces when cooking • Concentration: amount of substance per volume – Ex. Turning the valve on a gas stove to increase the concentration of methane molecules • Catalysts (enzymes): organic substances that help speed up chemical reactions, but are not consumed in the reaction 10

Law of Conservation of Mass • Proposed by Antoine Lavoisier 1. In a chemical reaction, atoms are neither created nor destroyed. 2. All atoms present in the reactants are also present in the products. 3. Chemical equations must account for/show the conservation of mass in balancing equations. In its present form, does this chemical equation show a conservation of mass? 2 H 2 + O 2 2 H 2 O Reactants H 24 O 22 How would you balance this equation to show the conservation of mass? Products H 24 O 12 11

Do the following chemical equations show a conservation of mass? If not, which elements are not balanced? 2 Fe + 3 Cl 2 2 Fe. Cl 3 2 Fe+ O 2 Fe 2 O 3 2 H 2 + O 2 2 H 2 O N 2 + H 2 2 NH 3 12

Hints For Balancing Equations • Count the atoms – List the number of atoms of each element to see which elements must be balanced • Use a coefficient to add atoms to one side of the equation – Start with the reactant or product that has the greatest number of different elements • Add a coefficient to another reactant or product – Make sure that the coefficients in your balanced equation are the smallest whole numbers possible (they should have no common factor other than one) Tutorial on Balancing Equations 13

Chemical Reactions • https: //www. youtube. com/watch? v=Cg. WHbp. MVQ 1 U • Objectives: 1. Predict products of chemical reactions such as synthesis, decomposition, replacement, and combustion. 2. Balance chemical equations with adherence to law of conservation of matter. 3. Review chemical change indicators such as precipitate, evolution of gas, color change, and absorption/release of heat. 14
- Slides: 14