Chemical Reactions Different Types of Reactions ReviewRemember Whats

Chemical Reactions Different Types of Reactions

Review/Remember: • What’s the difference between a Physical and Chemical Change?

Physical Change: Changes the way something looks Chemical Change: Changes the chemical makeup of the substance (it makes something new!) Can’t be undone or reversed by physical changes Indicators of a chemical change: 1. light 2. Heat 3. Gas (bubbles) 4. Color change 5. Precipitate (formation of a solid from liquids)

CHEMICAL REACTIONS • Occur when substances undergo chemical changes to form new substances. • Atoms are rearranged (NOT created or destroyed). Parts of a Reaction – Reactants • Substance or molecule that participates in a chemical reaction. • On left side of arrow in chemical equation – Products • Substance that forms in a chemical reaction. • On right side of arrow in chemical equation 2 H 2 + O 2 2 H 2 O (Reactants) (Products)
CHEMICAL BONDS BETWEEN ATOMS (Ionic and Covalent) • Sometimes energy must be added to break bonds so atoms can be rearranged. – Activation energy = energy required to begin or finish a reaction - Many forms of energy can be used to break bonds such as heat, light, electricity, and sound. • Often forming bonds releases energy (like explosions).
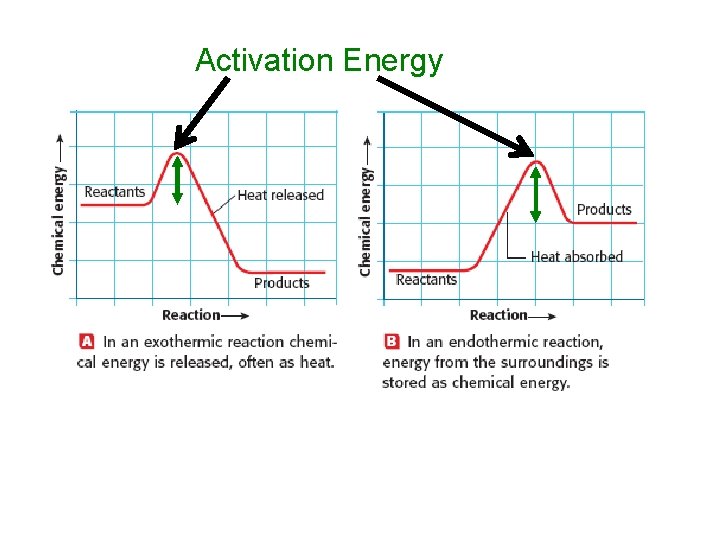
Activation Energy

CONSERVATION OF ENERGY AND MASS • Mass and energy are conserved in chemical reactions. • The total energy and mass that exists before the reaction is equal to the total energy and mass of the products and their surroundings. HEAT AND CHEMICAL REACTIONS • Exothermic reaction = chemical reaction in which heat is released to the surroundings. • Endothermic reaction = chemical reaction that absorbs heat

SPEED OF A REACTION • CATALYST = substance that changes the rate of a chemical reaction without being consumed or changed significantly. • Catalysts are not reactants or products, because they are not used up in the reaction. • Catalysts are often used in industry to make reactions go faster (saves money and time). • Catalyst in your body are called enzymes

Types of Reactions

#1 Reaction • Synthesis Reaction • Two or more substances combine to form a new compound. • Synthesis reactions have the following general form: A + B AB • Example: 2 Na + Cl 22 2 Na. Cl
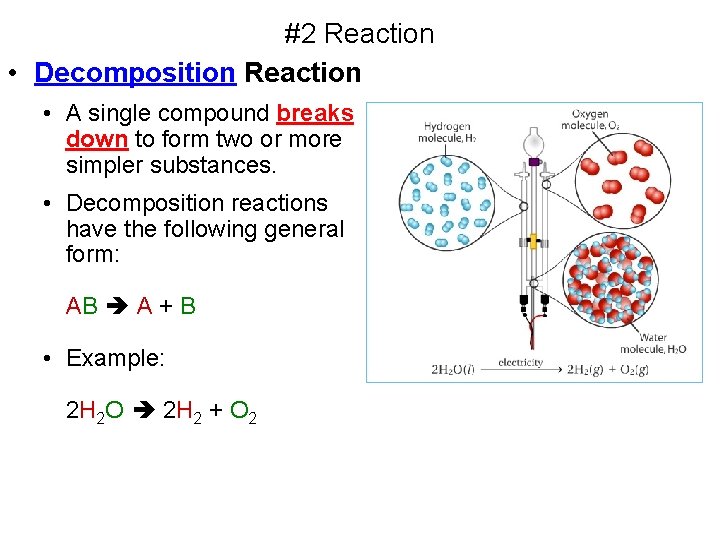
#2 Reaction • Decomposition Reaction • A single compound breaks down to form two or more simpler substances. • Decomposition reactions have the following general form: AB A + B • Example: 2 H 2 O 2 H 2 + O 2

# 3 Reaction • Single-Displacement Reaction • One element or ion takes the place of another element or ion in the compound. • Single-displacement reactions have the following general form: AX + B BX + A • Example: 3 Cu. Cl 2 + 2 Al. Cl 3 + 3 Cu

#4 Reaction • Double-displacement Reaction • A gas, a solid precipitate, or a molecular compound forms from the apparent exchange of atoms or ions between two compounds. • Double-displacement reactions have the following general form: AX + BY AY + BX • Example: Pb(NO 3)2 + K 2 Cr. O 4 Pb. Cr. O 4 + 2 KNO 3
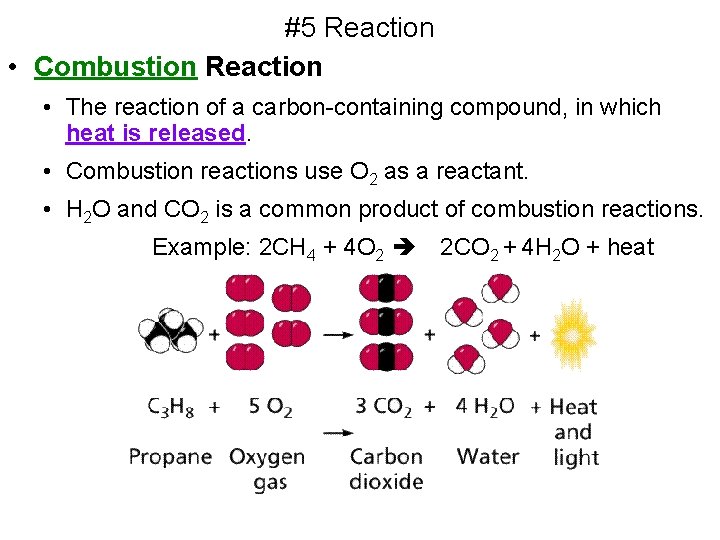
#5 Reaction • Combustion Reaction • The reaction of a carbon-containing compound, in which heat is released. • Combustion reactions use O 2 as a reactant. • H 2 O and CO 2 is a common product of combustion reactions. Example: 2 CH 4 + 4 O 2 2 CO 2 + 4 H 2 O + heat
- Slides: 14