Chemical Reactions Describing Chemical Reactions Chemical Reaction process

Chemical Reactions

Describing Chemical Reactions Chemical Reaction – process where a new substance is produced û Indications of a chemical reaction û heat and light û production of a gas û color change û formation of a precipitate û Precipitate – a solid formed when two ionic solutions are mixed û

Types of Reactions There are five types of chemical reactions we will talk about: 1. Combination 2. Decomposition 3. Single Replacement 4. Double Replacement 5. Combustion • You need to be able to identify the type of reaction and predict the product(s) •

Combination Reactions aka Synthesis reactions • occur when two or more substances combine and form a compound • Reactant + Reactant 1 Product • • Ex: Ca (s) + Cl 2 (g) Ca. Cl 2 (s)

Practice Predict the products. Write and balance the following synthesis reaction equations. • Example #1 - Aluminum metal reacts with fluorine gas 2 Al(s) + 3 F 2(g) 2 Al. F 3 (s) •

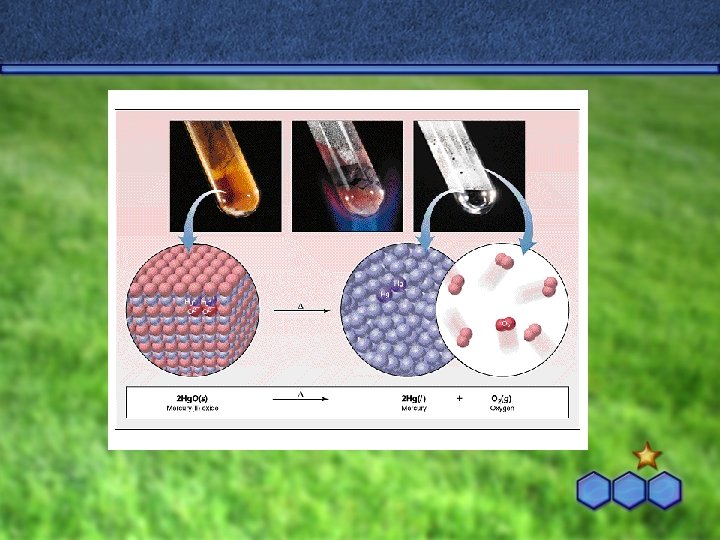
Decomposition Reactions occurs when a compound breaks up into it’s elements when heated • 1 Reactant Product + Product • • Ex: 2 KCl (s) 2 K (s) + Cl 2 (g)

Practice Predict the products. Then, write and balance the following decomposition reaction equations: • Example #2 – Solid lead (IV) oxide decomposes • using a catalyst Catalyst Pb. O 2(s) Pb(s) + O 2(g)

Single Replacement Reactions occur when one element replaces another in a compound • a metal can replace a metal (+) OR a nonmetal can replace a nonmetal (-) • • Ex: 2 K (s) + Mg. Cl 2 (aq) 2 KCl (aq) + Mg (s) • Ex: Br 2 (g) + 2 Li. Cl (aq) 2 Li. Br (aq) + Cl 2 (g)

Single Replacement Reactions • Write and balance the following single replacement reaction equation: • Example #3 - Zinc reacts with aqueous hydrogen monochloride Zn(s) + 2 HCl(aq) Zn. Cl 2(aq) + H 2(g) Note: Zinc replaces the hydrogen ion in the reaction • Example #4 - Sodium chloride solid reacts with fluorine 2 Na. Cl(s) + F 2(g) 2 Na. F(s) + Cl 2(g)

Double Replacement Reactions occur when a metal replaces a metal and a nonmetal replaces a nonmetal in a compound • Example: Ag. NO 3(aq) + Na. Cl(s) Na. NO 3(aq) + Ag. Cl(s) • • Another example: K 2 SO 4(aq) + Ba(NO 3)2(aq) 2 KNO 3(aq) + Ba. SO 4(s) • Think about it like “foil”ing in algebra, first and last ions go together + inside ions go together

Practice • Predict the products. Balance the equation Example # 5 – Aqueous calcium chloride reacts with aqueous sodium phosphate 3 Ca. Cl 2(aq) + 2 Na 3 PO 4(aq) Ca 3(PO 4)2 (aq) + 6 Na. Cl(s) • Example # 6 – Aqueous iron (III) chloride reacts with aqueous sodium hydroxide Fe. Cl 3(aq) + 3 Na. OH(aq) Fe(OH)3 (aq) + 3 Na. Cl (s) •

Combustion Reactions occur when a hydrocarbon or alcohol reacts with oxygen gas • Cx. Hy (or Cx. Hy. OH) + O 2 CO 2 + H 2 O • products are ALWAYS carbon dioxide and water • Ex : 2 CH 3 OH + 3 O 2 4 H 2 O + 2 CO 2 •

Combustion Write the products and balance the following combustion reaction: • Example #7 • C 5 H 12 + 8 O 2 6 H 2 O + 5 CO 2 •

û This is also called burning!!! In order to burn something you need the 3 things in the “fire triangle”: 1) A Fuel (hydrocarbon) 2) Oxygen to burn it with 3) Something to ignite the reaction (spark)

- Slides: 16