CHEMICAL REACTIONS CHEMICAL REACTIONS The process by which

CHEMICAL REACTIONS

CHEMICAL REACTIONS The process by which one or more substances are changed into one or more different substances

WHAT ARE REACTIONS? Reactions are represented by a chemical equation Reactants -----> Products Must have the same number and type of atoms on both sides of the equation

SYNTHESIS REACTIONS Two reactants produce one product Examples: ___NH 3 + ___ HCl ___ NH 4 Cl ___ Al ___ Cl 2 ___ Al. Cl 3 +

MORE SYNTHESIS __Ag + __ S 8 __Ag 2 S __Cr + __O 2 __Cr 2 O 3

DECOMPOSITION REACTIONS One reactant produces two products Examples ___ Ag 2 O ___ Ag + ___ O 2 ___ PCl 5 ___ PCl 3 + ___ Cl 2

SINGLE DISPLACEMENT REACTION One element and one compound producing a different element and compound Examples – we need to balance… ___ Fe + ___ H 3(PO 4) ___ H 2 + ___ Fe 3(PO 4)2 ___ H 2 + ___ Cu. O ___ Cu + ___ H 2 O

SINGLE DISPLACEMENT EXAMPLE Let’s practice balancing some more… ____ Mg + ____ Fe 2 O 3 ____ Fe + ____ Mg. O ____ Al+ ____ Cu. Cl 2 ____ Al. Cl 3 + ____ Cu

DOUBLE DISPLACEMENT Two compounds producing two different compounds Examples: (guess what we have to do? ) ___ Al(NO 3)3 + ___ Na. OH ___ Al(OH)3 + ___ Na. NO 3 ___ Pb. Cl 2 + ___ Li 2 SO 4 ___ Pb. SO 4 + ___ Li. Cl

DOUBLE DISPLACEMENT Some more practice (I know how excited you are…) ____ Na. Br + ____ H 3 PO 4 ____ Na 3 PO 4 + ____ HBr ____ Ca(OH)2 + ____ Al 2(SO 4)3 ____ Ca. SO 4 + ____ Al(OH)3

COMBUSTION REACTIONS A hydrocarbon reacts with oxygen to produce carbon dioxide and water Examples: (You know what to do!) ___ CH 4 + ___ O 2 ___ CO 2 + ___ H 2 O ___ C 6 H 12 O 6 + ___ O 2 ___ CO 2 + ___ H 2 O
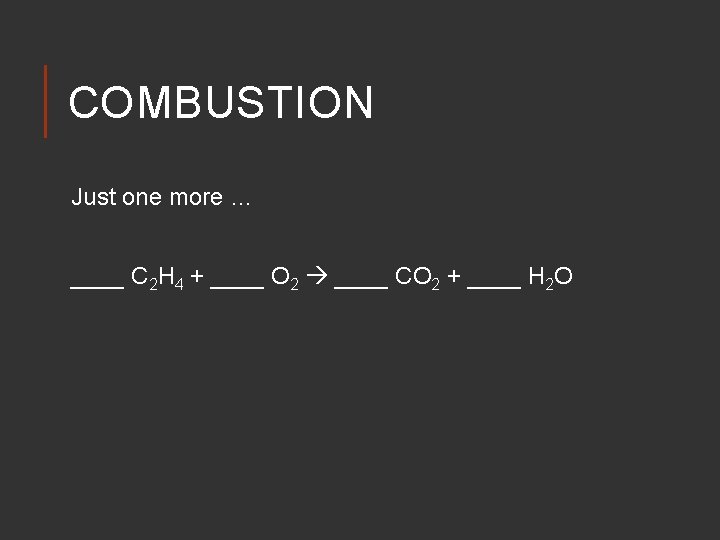
COMBUSTION Just one more … ____ C 2 H 4 + ____ O 2 ____ CO 2 + ____ H 2 O
- Slides: 12