Chemical Reactions Chemical Equations The starting substances are

Chemical Reactions Chemical Equations
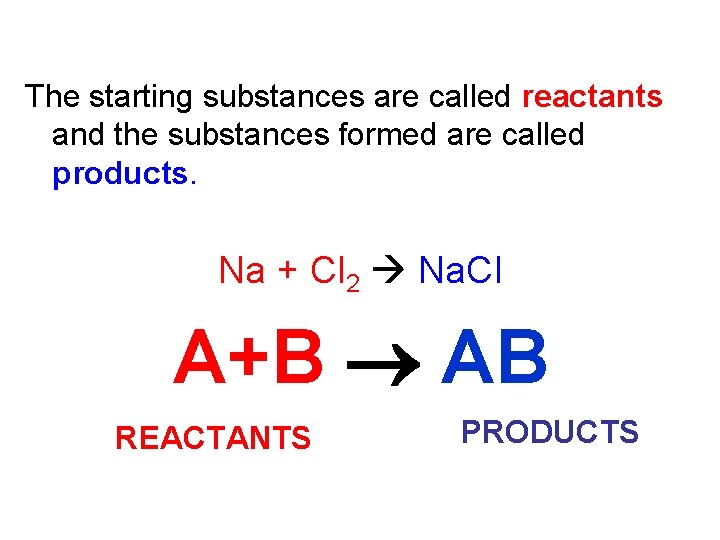
The starting substances are called reactants and the substances formed are called products. Na + Cl 2 Na. Cl A+B AB REACTANTS PRODUCTS

Describing Chemical Reactions

Writing Equations 2 H 2(g) + O 2(g) 2 H 2 O(g) • Identify the substances involved. • Use symbols to show: – How many? - coefficient – Of what? - chemical formula – In what state? – (physical state) • Remember the diatomic elements.

Law of Conservation of Mass • The Law of Conservation of Mass states that the mass of all substances present before a chemical change is equal to the mass of all substances after the change. 10 g Before Reaction 10 g After Reaction

Law of Conservation of Mass Sometimes the mass of the reactants and the mass of the products appears to be unequal. This is usually because some of the mass is lost as a gas. Wood + Fuel 99 g + 1 g Total Mass = 100 g ash + released gas = 99. 5 g + 0. 5 g Total Mass = 100 g Although it appears mass is lost, 0. 5 g of mass is released into the air. The sum on both sides of the reaction is equal.
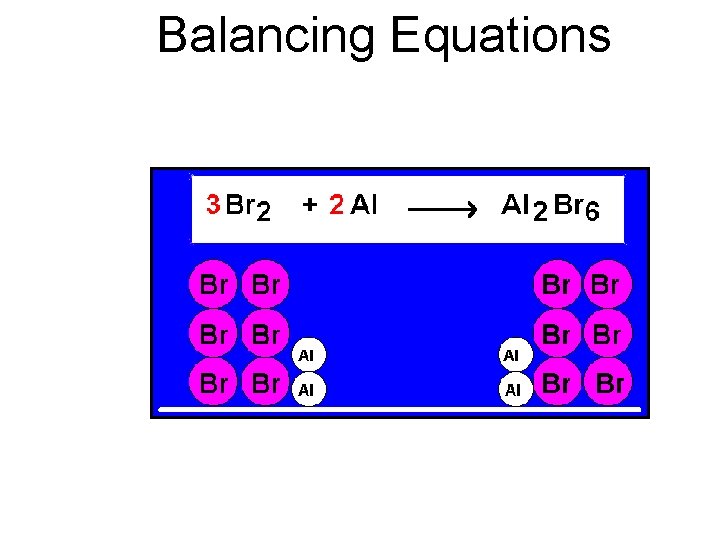
Balancing Equations

Balancing Steps 1. Write the unbalanced equation. 2. “Take Inventory”- Count atoms on each side. 3. Add coefficients to make #s equal. Coefficient subscript = # of atoms 4. Reduce coefficients to lowest possible ratio, if necessary. 5. Double check atom balance!!!

Helpful Tips • Balance one element at a time. • Update “inventory” after adding a coefficient. • If an element appears more than once per side, balance it last. • Balance polyatomic ions as single units. – “ 1 SO 4” instead of “ 1 S” and “ 4 O”
- Slides: 9