CHEMICAL REACTIONS Chemical Equations Reactants substances that undergo

CHEMICAL REACTIONS

Chemical Equations Reactants – substances that undergo changes Products – new substances formed from reaction n Reactants N 2 + 3 H 2 Products 2 NH 3

Conservation of Mass *Mass is not created in chem. reaction *Mass in not destroyed in chem. Reaction

Types of Reactions Synthesis Decomposition Single-replacement Double-replacement Combustion

Synthesis Two or more substances react to form a single substance A+B AB Example: 2 H 2 + O 2 H 2 O

Decomposition A compound breaks down into two or more simpler substances n n Reactant MUST be a compound! Products may be elements or compounds AB 2 H 2 O A+B 2 H 2 + O 2
Single Replacement One element takes the place of another element in a reaction A + BC B + AC Cu + 2 Ag. NO 3 2 Ag + Cu(NO 3)2
Double Replacement Two different compounds exchange positive ions to form two new compounds AB + CD AD + CB Ca. CO 3 + 2 HCl Ca. Cl 2 + H 2 CO 3
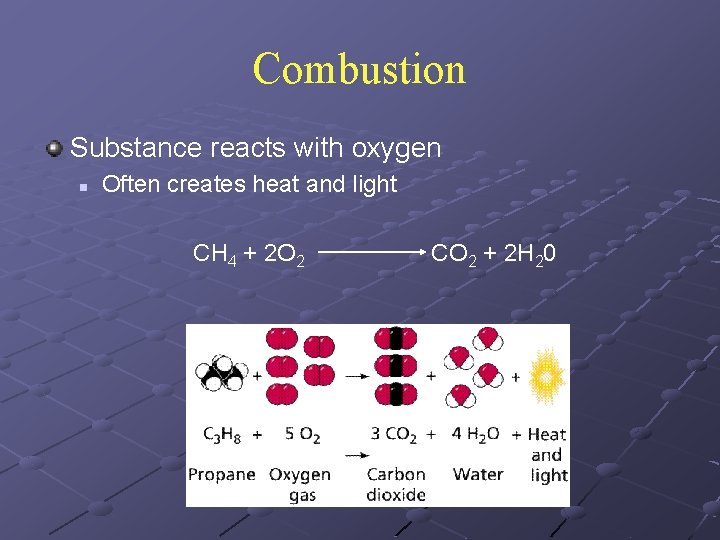
Combustion Substance reacts with oxygen n Often creates heat and light CH 4 + 2 O 2 CO 2 + 2 H 20
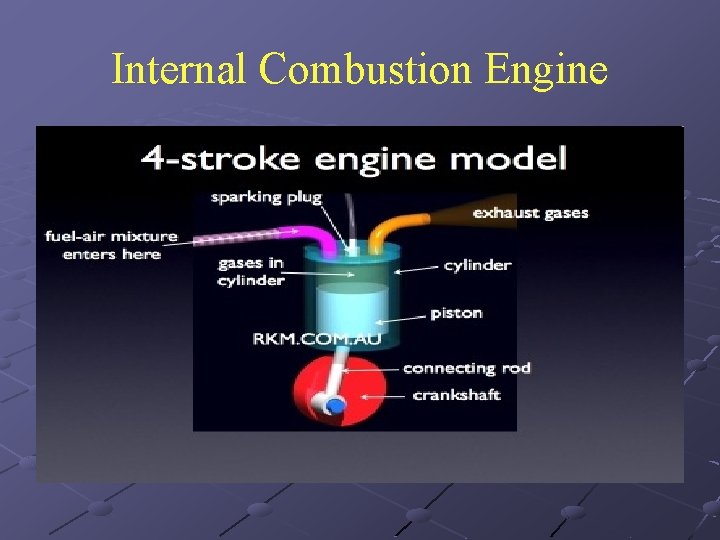
Internal Combustion Engine

Reaction Rates Reaction Rate – rate at which reactants change into products over time. TNT vs. Leaves changing

Reaction Rates ~ 200 – 350 calories -A Cal X-Country runner burns calories- energy is released OR -Just burn the bar! -energy released faster! **Same amount**

Factors affecting reaction rates… An increase in TEMPERATURE increases reaction rate. n Decrease in temp = decrease in reaction rate.

Surface area Increase in surface area of a reactant increases reaction rate. (and vice versa)

Stirring reactants increases reaction rate.

Concentration The more reacting particles, the more likely a reaction will occurr. n n Concentrated detergent Grape juice concentrate

Catalysts Catalyst – substance that affects reaction rate without being used up. n Making friends…

Equilibrium When opposing changes take place at the same rate. n Physical and Chemical equilibria. Reversible Reaction: n A+B C

Reactions… Exothermic – releases energy to its surroundings. n Warm test tube? ? ? Endothermic – absorbs energy from surroundings.

MOLES Mole – amount of a substance that contains ~ 6. 02 x 1023 particles of that substance Avagadro’s number (don’t worry about it. ) Marbles?

Mole vs. Dozen Eggs vs. Donuts A dozen eggs has a different mass than a dozen donuts. *But its still a dozen! **A mole works the same as a dozen**

Molar Mass *The mass of one mole of a substance = molar mass *Molar Mass = same as Atomic Mass but expressed in Grams! ? ? ? What’s the molar mass of Carbon? ? ? Sulfur? ? ?

Mole - Mass Conversions CO 2 = ~ 44 grams C = ~ 12 grams O 2 = ~ 32 grams Conversion Factor: 44. 0 g CO 2 OR 1 mol CO 2 44. 0 g CO 2

Mole Conversions… Say you have… 50 g of CO 2 How many moles of CO 2 do you have? ? ? Use conversion factor: 50 g CO 2 X 1 mol CO 2 44. 0 g CO 2 = 50. 0 g CO 2 44. 0 g CO 2 ANSWER = 1. 14 moles of CO 2

Converting… Convert 1. 25 mol Mg. Cl 2 to grams! 1. Find out 1 mole of Mg. Cl 2 by adding atomic mass of each element. Mg = ~24 + Cl = ~35 = 94 2. Use correct conversion factor: 1. 25 mol Mg. Cl 2 X 94 g Mg. Cl 2 1 mol Mg. Cl 2 3. Multiply across and cancel like units! ANSWER = ~ 118 g Mg. Cl 2
- Slides: 25