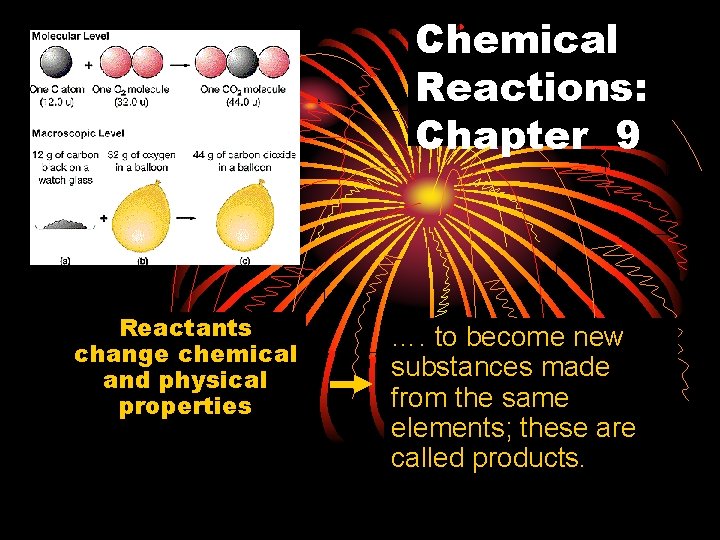
Chemical Reactions Chapter 9 Reactants change chemical and











- Slides: 26

Chemical Reactions: Chapter 9 Reactants change chemical and physical properties …. to become new substances made from the same elements; these are called products.

Types of Reactions • • • Synthesis or Composition Decomposition Single Replacement Double Displacement Combustion Reactions

Composition/ Synthesis Reactions • A + X AX • An element may react with oxygen to form the oxide (oxidation) A X

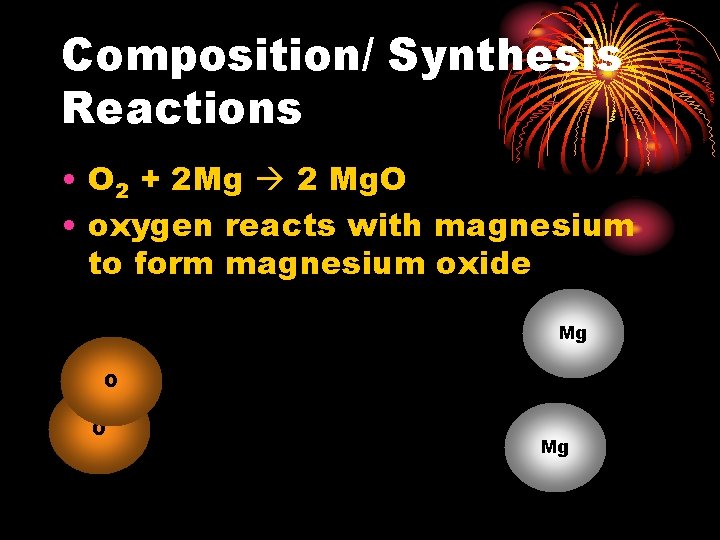
Composition/ Synthesis Reactions • O 2 + 2 Mg 2 Mg. O • oxygen reacts with magnesium to form magnesium oxide Mg O O Mg

Composition/ Synthesis Reactions • C + O 2 CO 2 • carbon reacts with oxygen to form carbon dioxide O O C

Practice predicting reactions with oxide and sulfides O 2 2 Fe. O 1. 2 Fe + ____ O 2 Fe O 2. 4 Fe + 3 ___ 2 3 SO 2 3. S 8 + 8 O 2 8 ____ Ba. S 4. 8 Ba + S 8 8 ____

Metals react with halogens. • • • 2 Na + Cl 2 2 Na. Cl 2 Co + 3 F 2 2 Co. F 3 2 K + I 2 2 KI Sr + Br 2 Sr. Br 2 2 Mg + O 2 2 Mg. O

Synthesis reactions can also occur with oxides. • Active metal oxides react with water to form hydroxides. ex: • Ca. O + H 2 O Ca(OH)2 • Many oxides of nonmetals react with water to form oxyacids. ex: • SO 2 + H 2 O H 2 SO 3 • Some metal oxides react with nonmetal oxides to form salts. • Ca. O + SO 2 Ca. SO 3

Practice • Active metal oxides react with water to form hydroxides. ex: Mg(OH)2 • Mg. O + H 2 O ____ • Many oxides of nonmetals react with water to form oxyacids. ex: H 2 SO 4 • SO 3 + H 2 O _____ • Some metal oxides react with nonmetal oxides to form salts. Mg(NO 3)2 • Mg. O + NO 2 ____

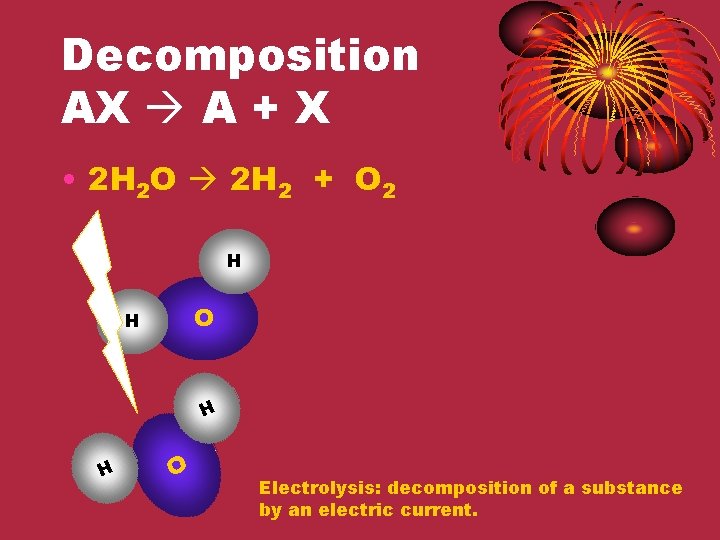
Decomposition AX A + X • 2 H 2 O 2 H 2 + O 2 H O H H H O Electrolysis: decomposition of a substance by an electric current.

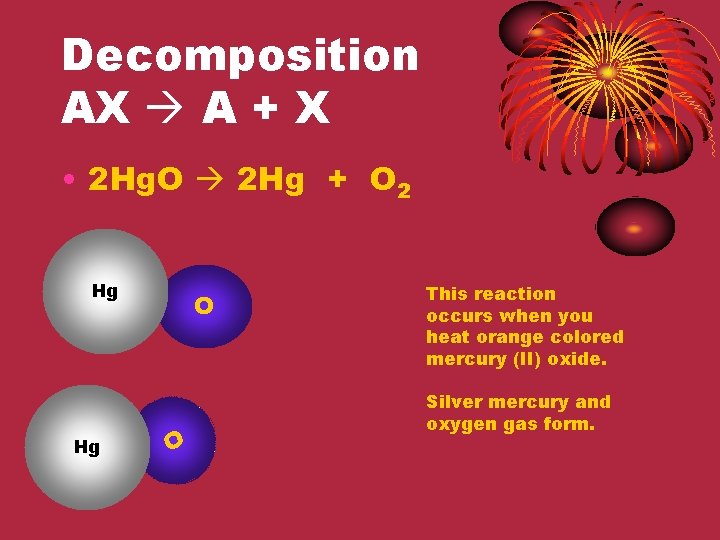
Decomposition AX A + X • 2 Hg. O 2 Hg + O 2 Hg Hg O O This reaction occurs when you heat orange colored mercury (II) oxide. Silver mercury and oxygen gas form.

Decomposition of metal carbonates ACO 3 AO + CO 2 (g) • Ca. CO 3 (s) Ca. O (s) + CO 2 (g) O Ca C O O This reaction occurs when you heat calcium carbonate. calcium oxide and carbon dioxide form.

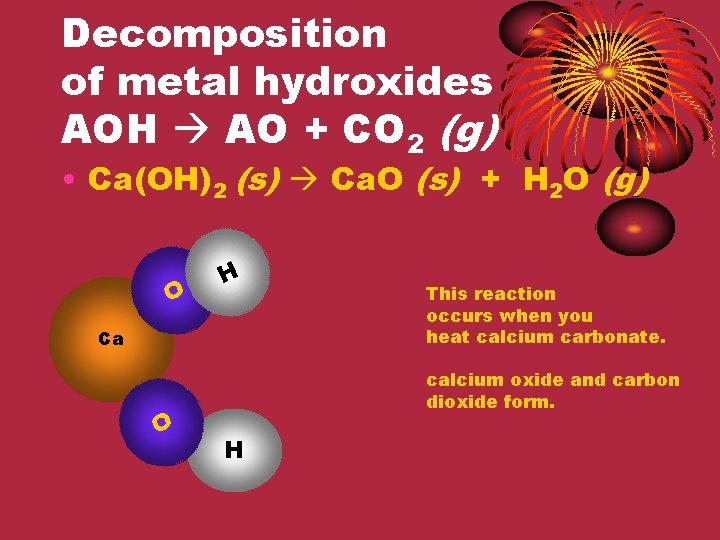
Decomposition of metal hydroxides AOH AO + CO 2 (g) • Ca(OH)2 (s) Ca. O (s) + H 2 O (g) O H Ca O This reaction occurs when you heat calcium carbonate. calcium oxide and carbon dioxide form. H

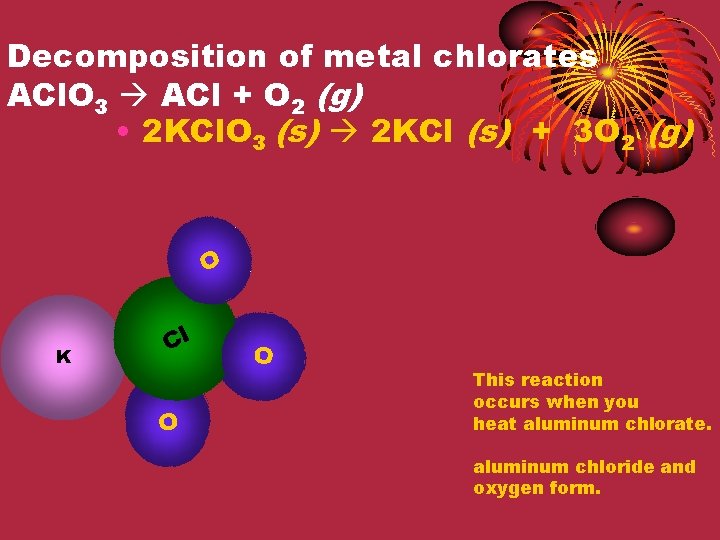
Decomposition of metal chlorates ACl. O 3 ACl + O 2 (g) • 2 KCl. O 3 (s) 2 KCl (s) + 3 O 2 (g) O Cl K Cl O O This reaction occurs when you heat aluminum chlorate. aluminum chloride and oxygen form.

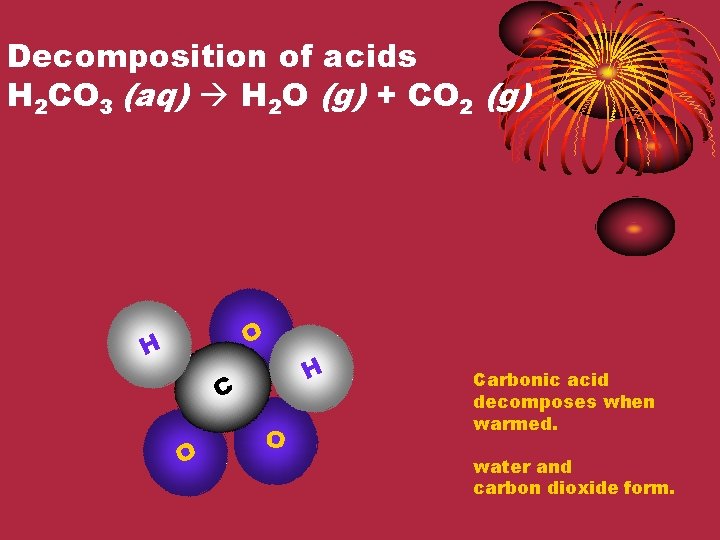
Decomposition of acids H 2 CO 3 (aq) H 2 O (g) + CO 2 (g) O H H C O O Carbonic acid decomposes when warmed. water and carbon dioxide form.

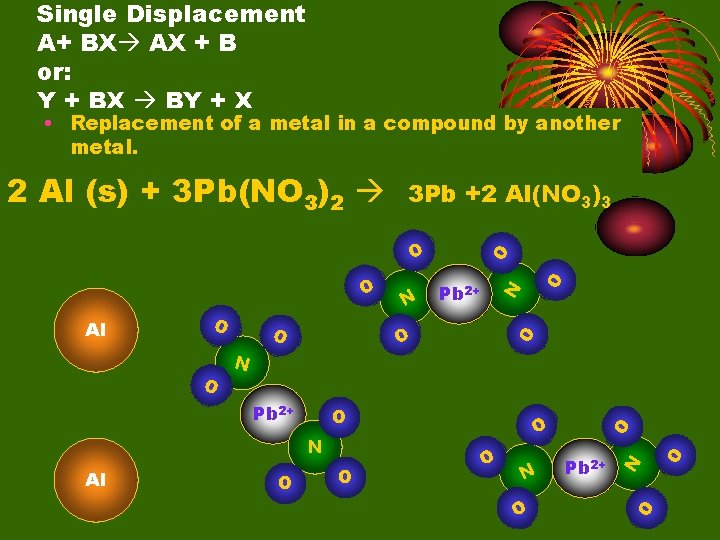
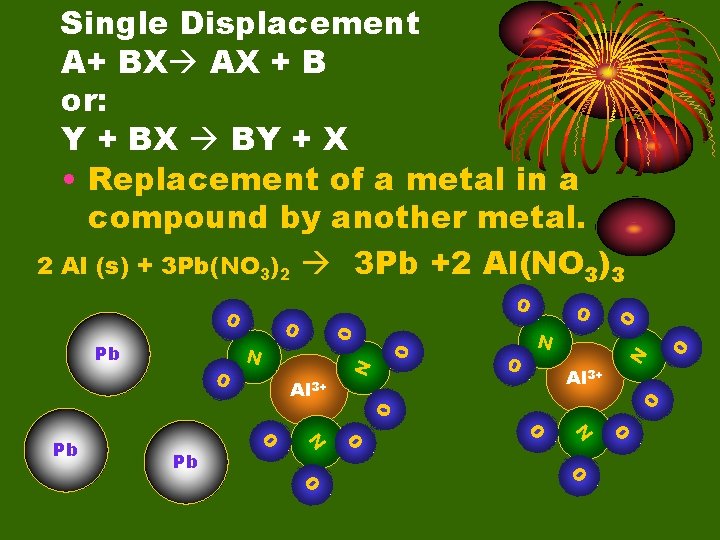
Single Displacement A+ BX AX + B or: Y + BX BY + X • Replacement of a metal in a compound by another metal. 2 Al (s) + 3 Pb(NO 3)2 3 Pb +2 Al(NO 3)3 O O N N O O O Pb 2+ O O N Pb 2+ O N Al O O Al Pb 2+

O O N O O O Pb O O N Al 3+ N O Al 3+ O Pb O Single Displacement A+ BX AX + B or: Y + BX BY + X • Replacement of a metal in a compound by another metal. 2 Al (s) + 3 Pb(NO 3)2 3 Pb +2 Al(NO 3)3

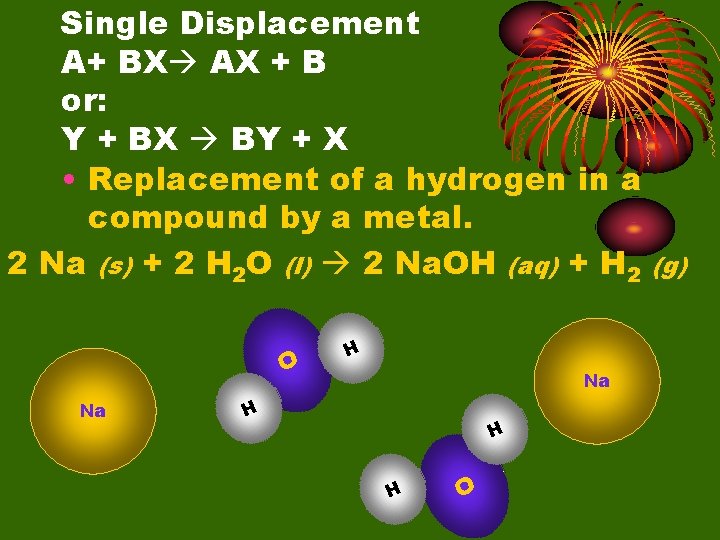
Single Displacement A+ BX AX + B or: Y + BX BY + X • Replacement of a hydrogen in a compound by a metal. 2 Na (s) + 2 H 2 O (l) 2 Na. OH (aq) + H 2 (g) O Na H H H O

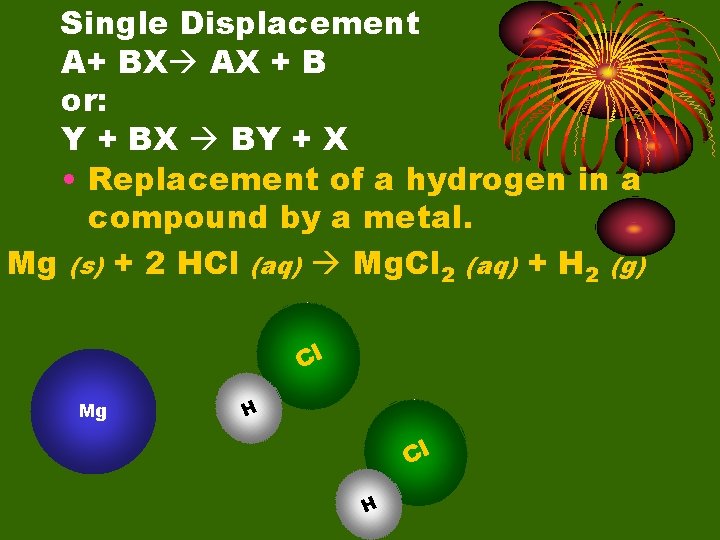
Single Displacement A+ BX AX + B or: Y + BX BY + X • Replacement of a hydrogen in a compound by a metal. Mg (s) + 2 HCl (aq) Mg. Cl 2 (aq) + H 2 (g) Cl Mg H Cl H

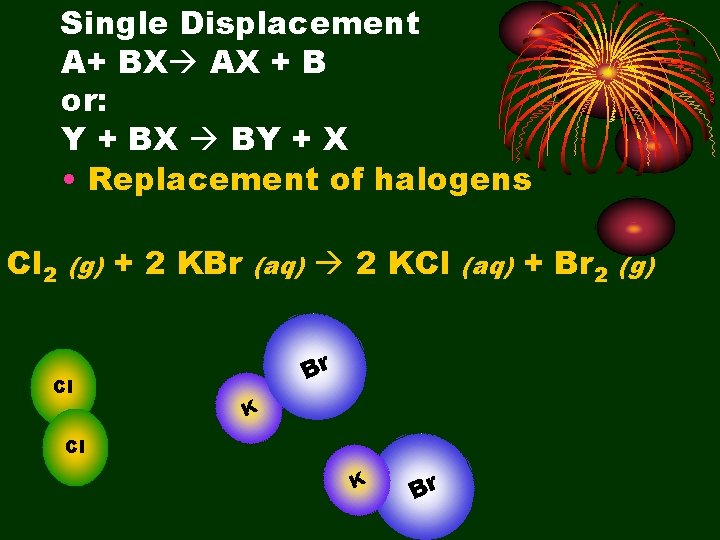
Single Displacement A+ BX AX + B or: Y + BX BY + X • Replacement of halogens Cl 2 (g) Cl + 2 KBr (aq) 2 KCl Br K Cl K Br (aq) + Br 2 (g)

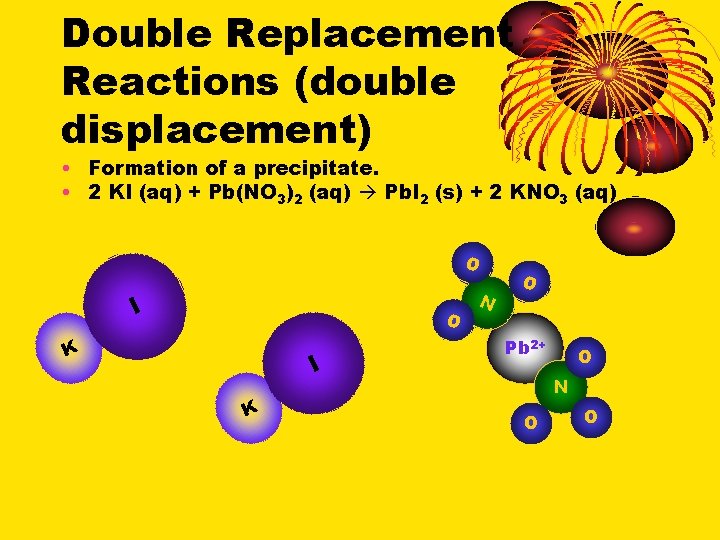
Double Replacement Reactions (double displacement) • Formation of a precipitate. • 2 KI (aq) + Pb(NO 3)2 (aq) Pb. I 2 (s) + 2 KNO 3 (aq) O I K N K O O I Pb 2+ O N O O

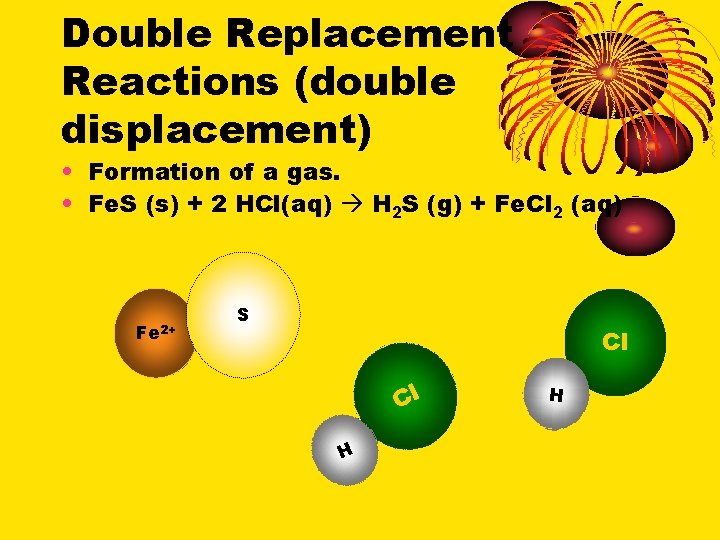
Double Replacement Reactions (double displacement) • Formation of a gas. • Fe. S (s) + 2 HCl(aq) H 2 S (g) + Fe. Cl 2 (aq) S Cl Cl H H Fe 2+

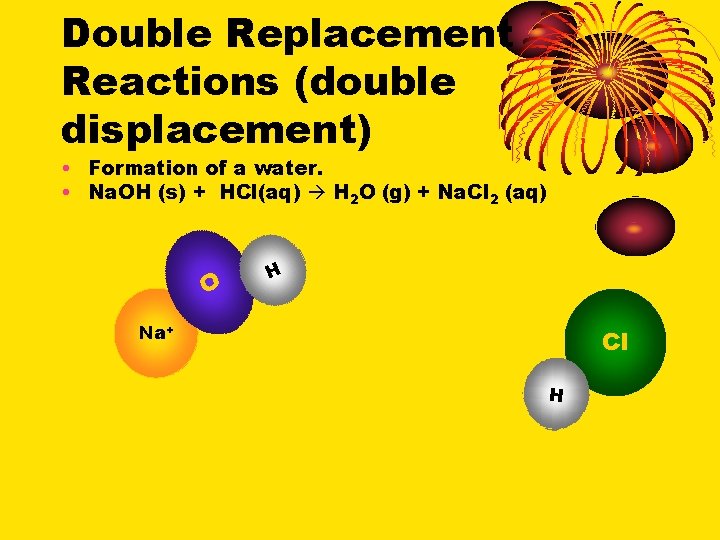
Double Replacement Reactions (double displacement) • Formation of a water. • Na. OH (s) + HCl(aq) H 2 O (g) + Na. Cl 2 (aq) O H Na+ Cl H

Combustion Reactions • A substance combines with oxygen, releasing a large amount of energy in the form of light and heat. 2 H 2 (g) + O 2 (g) 2 H 2 O O H H H

Combustion Reactions • A substance combines with oxygen, releasing a large amount of energy in the form of light and heat. C 3 H 8 (g) +5 O 2 (g) 4 H 2 O + 3 CO 2 O O H H C H O O O

Combustion Reactions • A substance combines with oxygen, releasing a large amount of energy in the form of light and heat. C 3 H 8 (g) +5 O 2 (g) 4 H 2 O + 3 CO 2 O H H H O O H C O C H O O O C