Chemical Reactions Chapter 4 Stoichiometry 1 Balancing Chemical

Chemical Reactions Chapter 4 Stoichiometry 1

Balancing Chemical Equations “Matter is conserved in chemical change” Antoine Lavoisier, 1789 An equation must be balanced: It must have the same number of atoms of each kind on both sides 4

Combustion Reactions In combustion, a hydrocarbon or C–H–O fuel combines with O 2 to form CO 2 and H 2 O __ CH 4 + __ O 2 __ CO 2 + __ H 2 O 1 CH 4 + 2 O 2 1 CO 2 + 2 H 2 O Balanced equation shows 1 C, 4 H, and 4 O on each side If N or S are in the formula for the fuel, assume it is oxidized to NO 2 or SO 2 6

Example 4 -2 Write a balanced equation for the complete combustion of glycerol, C 3 H 8 O 3 Write a balanced equation for the complete combustion of thiosalicylic acid, C 7 H 6 O 2 S 8

Stoichiometry is chemical accounting The heart of stoichiometry is the mole ratio given by the coefficients of the balanced equation 9

Stoichiometry is chemical accounting The heart of stoichiometry is the mole ratio given by the coefficients of the balanced equation moles A mole ratio moles B moles A moles B 10

Example 4 -3 B How many moles of Ag are produced in the decomposition of 1. 00 kg of silver (I) oxide: 2 Ag 2 O (s) 4 Ag (s) + O 2 (g) 12

Example 4 -6 B The model problem describes an Al-Cu alloy composed of 93. 7% Al and 6. 3% Cu by mass, with a density of 2. 85 g/cm 3. The Al (but not the Cu) reacts with HCl: 2 Al (s) + 6 HCl (aq) 2 Al. Cl 3 (aq) + 3 H 2 (g) How many grams of Cu are present in a sample of alloy that yields 1. 31 g H 2 when it reacts with HCl? 18

Example 4 -7 B A vinegar contains 4. 0% HC 2 H 3 O 2 by mass and has a density of 1. 01 g/m. L. It reacts with sodium hydrogen carbonate: HC 2 H 3 O 2 (aq) + Na. HCO 3 (s) Na. C 2 H 3 O 2 (aq) + H 2 O (l) + CO 2 (g) How many grams of CO 2 are produced by the reaction of 5. 00 m. L of this vinegar with Na. HCO 3? 20

Chemical Reactions in Solution Most reactions occur in aqueous solution SOLUTE is the substance to be dissolved in solution SOLVENT is the substance (often a liquid) the solute dissolves in The concentration of the solution is Molarity (M) = moles solute L solution 21

Example 4 -8 B 15. 0 m. L of concentrated acetic acid, HC 2 H 3 O 2 (d = 1. 048 g/m. L), are dissolved in enough water to produce 500. 0 m. L of solution. What is the concentration of the solution? 23

Example 4 -9 B How many grams of Na 2 SO 4 • 10 H 2 O are needed to prepare 355 m. L of 0. 445 M Na 2 SO 4? 25

Dilution problems It is common to prepare a solution by diluting a more concentrated solution (the stock solution). The moles of solute taken from the stock solution are given by moles solute = volume x molarity All the solute taken from the stock appears in the diluted solution, so moles solute are constant: Vstock. Mstock = Vdilute. Mdilute 26

Example 4 -10 A 15. 00 m. L of 0. 450 M K 2 Cr. O 4 solution are diluted to 100. 00 m. L. What is the concentration of the dilute solution? 27

Example 4 -10 B After being left out in an open beaker, 275 m. L of 0. 105 M Na. Cl has evaporated to only 237 m. L. What is the concentration of the solution after evaporation? 28

Stoichiometry in Solution Stoichiometry in solution is just the same as for mass problems, except the conversion into or out of moles uses molarity instead of molar mass: grams A grams B moles A m. L A mole ratio moles B moles A moles B m. L B 29

Example 4 -11 B K 2 Cr. O 4 (aq) + 2 Ag. NO 3 (aq) Ag 2 Cr. O 4 (s) + 2 KNO 3 (aq) How many m. L of 0. 150 M Ag. NO 3 must react with excess K 2 Cr. O 4 to produce exactly 1. 00 g Ag 2 Cr. O 4? 31

Limiting reactant In a given reaction, often there is not enough of one reactant to use up the other reactant completely The reactant in short supply LIMITS the quantity of product that can be formed 32
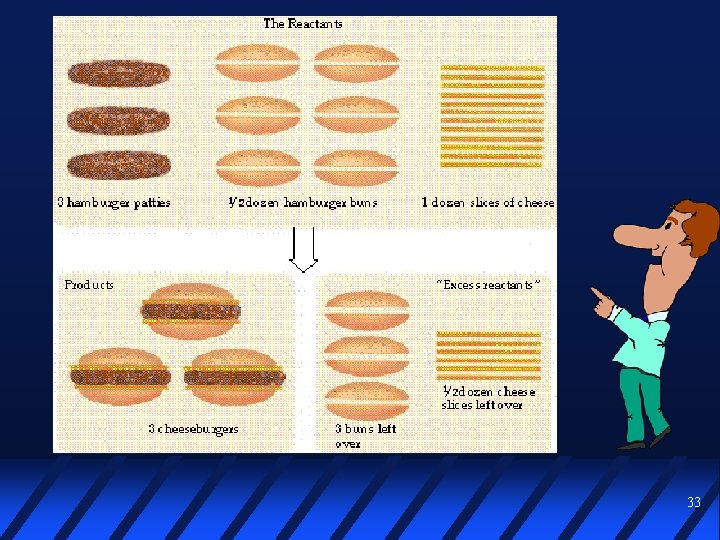
33

Goldilocks Chemistry Imagine reacting different amounts of Zn with 0. 100 mol HCl: Zn (s) + 2 HCl (aq) Zn. Cl 2 (aq) + H 2 (g) Mass Zn Moles HCl Ratio mol HCl mol Zn Rxn 1 6. 54 g 0. 100 mol 1. 00 Rxn 2 3. 27 g 0. 0500 mol 0. 100 mol 2. 00 Rxn 3 1. 31 g 0. 0200 mol 0. 100 mol 5. 00 34

Limiting reactant problems The easiest way to do these is to do two stoichiometry calculations Find the amount of product possible from each reactant The smaller answer is the amount of product you can actually make (you just ran out of one reactant) The reactant on which that answer was based is the limiting reactant 35

Example 4 -13 A When 215 g P 4 react with 725 g Cl 2 P 4 (s) + 6 Cl 2 (g) 4 PCl 3 (l) (example 4 -12 A) which reactant is in excess and what mass of that reactant remains after the reaction is finished? 38

Example 4 -13 B 12. 2 g H 2 and 154 g O 2 are allowed to react. Identify the limiting reactant, which gas remains after the reaction, and what mass of it is left over. 2 H 2 (g) + O 2 (g) 2 H 2 O (l) 39

Percent Yield In real experiments we often do not get the amount of product we calculate we should, because the reactants may participate in other reactions (side reactions) that produce other products (by-products) The reaction often does not go to completion. Percent yield tells the ratio of actual to theoretical amount formed. 40

Percent Yield Suppose you calculate that a reaction will produce 50. 0 g of product. This is theoretical yield. The reaction actually produces only 45. 0 g of product. This is the actual yield. Percent yield = 45. 0 g (actual) x 100 = 90. 0% 50. 0 g (theoretical) 41

Example 4 -14 B What is the percent yield if 25. 0 g P 4 reacts with 91. 5 g Cl 2 to produce 104 g PCl 3: P 4 (s) + 6 Cl 2 (g) 4 PCl 3 (l) 43

Example 4 -15 B What mass of C 6 H 11 OH should you start with to produce 45. 0 g C 6 H 10 if the reaction has 86. 2% yield and the C 6 H 11 OH is 92. 3% pure: C 6 H 11 OH (l) C 6 H 10 + H 2 O (l) 45

Exercise 26 Balance these equations by inspection (NH 4)2 Cr 2 O 7 (s) Cr 2 O 3 (s) + N 2 (g) + H 2 O (g) NO 2 (g) + H 2 O (l) HNO 3 (aq) + NO (g) H 2 S (g) + SO 2 (g) S (g) + H 2 O (g) SO 2 Cl 2 + HI H 2 S + H 2 O + HCl + I 2 46

Exercise 30 Write balanced equations for these reactions: Sulfur dioxide gas with oxygen gas to produce sulfur trioxide gas Solid calcium carbonate with water and dissolved carbon dioxide to produce aqueous calcium hydrogen carbonate Ammonia gas and nitrogen monoxide gas to produce nitrogen gas and water vapor 47

Exercise 32 3 Fe (s) + 4 H 2 O (g) Fe 3 O 4 (s) + H 2 (g) How many moles of H 2 can be produced from 42. 7 g Fe and excess steam? How many grams of H 2 O are consumed in the conversion of 63. 5 g Fe to Fe 3 O 4? If 7. 36 mol H 2 are produced, how many grams of Fe 3 O 4 must also be produced? 48

Exercise 36 Silver oxide decomposes above 300 °C to yield metallic silver and oxygen gas. 3. 13 g impure silver oxide yields 0. 187 g O 2. Assuming there is no other source of O 2, what is the % Ag 2 O by mass in the original sample? 49

Exercise 42 How many grams of CO 2 are produced in the complete combustion of 406 g of a bottled gas that consists of 72. 7% C 3 H 8 (propane) and 27. 3% C 4 H 10 (butane), by mass? 50

Exercise 45 What are the molarities of these solutes? 150. 0 g sucrose (C 12 H 22 O 11) in 250. 0 m. L aqueous solution 98. 3 mg of 97. 9% pure urea, CO(NH 2)2, in 5. 00 m. L aqueous solution 12. 5. 0 m. L methanol (CH 3 OH, density = 0. 792 g/m. L) in 15. 0 L aqueous solution 51

Exercise 52 After 25. 0 m. L of aqueous HCl solution is diluted to 500. 0 m. L, the concentration of the diluted solution is found to be 0. 085 M HCl. What was the concentration of the original HCl solution? 52

Exercise 56 Ca(OH)2 (s) + 2 HCl (aq) Ca. Cl 2 (aq) + 2 H 2 O (l) How many grams of Ca(OH)2 will react completely with 415 m. L of 0. 477 M HCl? How many kilograms of Ca(OH)2 will react with 324 L of an HCl solution that is 24. 28% HCl by mass, density = 1. 12 g/m. L? 53
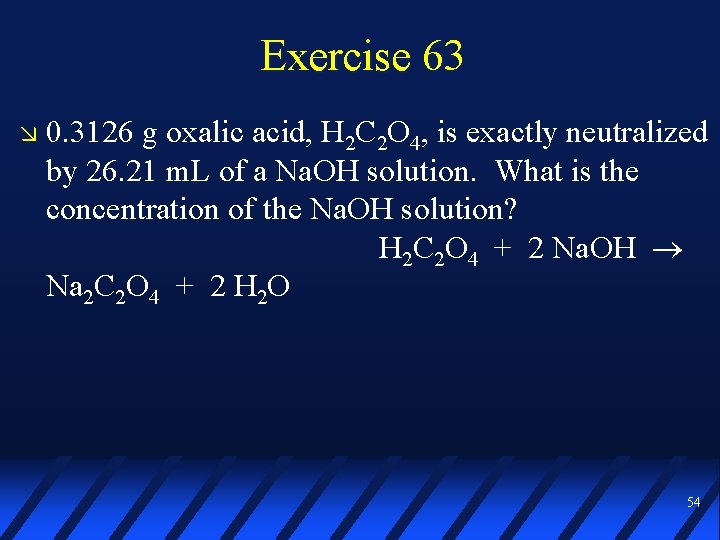
Exercise 63 0. 3126 g oxalic acid, H 2 C 2 O 4, is exactly neutralized by 26. 21 m. L of a Na. OH solution. What is the concentration of the Na. OH solution? H 2 C 2 O 4 + 2 Na. OH Na 2 C 2 O 4 + 2 H 2 O 54

Exercise 70 Chlorine can be generated by heating calcium hypochlorite and hydrochloric acid to form chlorine gas, calcium chloride, and water. If 50. 0 g Ca(OCl)2 and 275 m. L 6. 00 M HCl react, how many grams of Cl 2 gas form? Which reactant is left over, and how much (in grams)? 55

Exercise 72 2 C 6 H 5 NO 2 + 4 C 6 H 14 O 4 (C 6 H 5 N)2 + 4 C 6 H 12 O 4 + 4 H 2 O nitrobenzene triethylene glycol azobenzene If 0. 10 L nitrobenzene (d = 1. 20 g/m. L) react with 0. 30 L triethylene glycol (d = 1. 12 g/m. L) to form 55 g azobenzene, find Theoretical yield Actual yield Percent yield 56
- Slides: 38