Chemical Reactions Chapter 16 Sections 1 2 3



























- Slides: 30

Chemical Reactions Chapter 16 Sections 1, 2, 3

What is a Chemical Reaction? • Also referred to as a “chemical change” • The process in which matter changes producing one or more new substances. • This occurs when chemical bonds break between atoms.

What happens in a Chemical Reaction? • Chemical bonds in the reactants are broken, then atoms are rearranged to form new substances (products). • The amount of matter does not change during a chemical reaction, the atoms are only rearranged to form new substances. • This is evidenced (shown) in a balanced chemical equation.

What signs indicate a chemical reactions occurs? • A change in physical properties • A color change • Bubbles produced • An insoluble solid is produced called a precipitate. • A change in thermal energy. • An increase in temperature is called an exothermic reaction. • A decrease in temperature is called an endothermic reaction.

What is a chemical equation? Reactant + Reactant Product + Product 2 Mg + 2 HCl 2 Mg. Cl + H 2 Reactants- substances at the beginning of a reaction Products- substances at the end of a reaction Coefficient- the number of molecules in the reaction. Subscript- refers to the number of atoms of an element in a molecule.

Types of chemical reactions • Synthesis reaction- when one or more elements create a complex molecule • 2 H 2 + O 2 2 H 2 O • Decomposition reaction- When a complex molecule breaks into simpler substances. • 2 H 2 O 2 2 H 2 O + O 2 • Replacement reaction- When one element replaces another element in a molecule. • 2 Cu 2 O + C 4 Cu + CO 2

What is a Chemical Equation? • A chemical equation is a way that scientists represent a chemical reaction that has occurred. It shows the rearrangement of atoms in a chemical reaction. • It contains the chemical formulas of the substances involved in the • • reaction. An arrow is used to distinguish between the reactants and products, and can be understood as meaning “yields” or “makes”. Reactants are the substances broken apart or combined in a chemical reaction (what you start with!) and they are located on the left side of the arrow in a chemical equation. Products are new substances formed in a chemical reaction (what you end with!) and they are located on the right side of the arrow in a chemical equation. Ex: C + O 2 CO 2 • This equation says “carbon reacts with oxygen to yield (make) carbon dioxide. ” • The arrow shows the direction of the reaction: reactants products.

What are common examples of Chemical Reactions? Type of chemical change Description New Substances formed Combustion A fuel burning in a furnace. Produces light, heat, and a new substance. Rusting A combination of a substance with oxygen A new substance is formed. Iron (Fe) produces rust. (Fe. O 2) Tarnishing A combination of a substance with a bright metal to produce a dark coating. Brass, copper and silver darken in color in the presence of air. Neutralization The formation of a neutral substance when an acid and base are combined. A Salt and water

What factors help to speed up or slow down a reaction? 1. Surface Area-By increasing the surface area of a substance chemists can speed up a reaction 2. Temperature- An increase in temperature can speed up a reaction. 3. Concentration- By increasing the concentration (the amount of substance), you can increase the speed of the reaction. 4. Add a catalyst- substance NOT involved in the reaction but used to speed up the reaction.

What is the Law of Conservation of Mass? • This Law states that during a chemical reaction matter is not created or destroyed. • This means the mass of the reactants is equal to the mass of the products.

What is the Law of Conservation of Mass? • When substances react with each other, many changes can take place, but in every case the total amount of matter afterward is the same as before. • Discovered by Lavoisier—French chemist • Law of Conservation of Mass: in a chemical rxn, mass (atoms) is neither created nor destroyed • Mass of reactants = mass of products • All atoms present in the reactants are also present in the products. There must be the same number of atoms in the products and reactants.
Mass Stays the SAME • The mass of the products must be the same as the mass of the reactants. • You do not magically gain or lose mass!!! • Example: If you have 2 grams of Na react with 1 gram of Cl to make Na. Cl, you know you must have 3 grams of Na. Cl in the products! • Na + Cl Na. Cl 2 g + 1 g 3 g

Number of Atoms Stays the SAME • You must have the SAME number of atoms of EACH element on both sides of the equation. • If you have 2 atoms of oxygen in the reactants, you must have 2 atoms of oxygen in the products. (You do not magically gain or lose atoms!!!) • This is NOT something someone made up; it’s how chemical reactions happen in nature!

How can I tell how many atoms of each element there are? • Coefficient: the “big” number written in front of a chemical formula that tells you how many molecules of that substance there are. • Ex: 5 H 2 O = 5 molecules of water • The subscript tells you how many atoms of each element there are. • The coefficient times the subscript tells you how many total atoms of that element are present. • 5 H 2 O = 10 atoms of Hydrogen, 5 atoms of Oxygen • If there is no coefficient, then there is only one molecule of that substance!

What is the Law of Conservation of Mass? 2 Mg + O 2 2 Mg. O Mg Mg O O Mg. O

What does it mean to Balance a Chemical Equation? • Sometimes we have to “balance” a chemical equation to make sure that we have the same number of atoms of each element on both sides of the equation. To do this, we change the number of molecules by changing the coefficients (NEVER THE SUBSCRIPTS!!!) until we have the same number of atoms of each element on both sides. When we do this, we are saying how many molecules of each substance must be present before the reaction will take place (remember: this is not something scientists made up; this is how the reactions happen in nature!).

How do I know if an Equation is Balanced? 1. Check the number of atoms of each element on both sides of the equation (reactants and products). 2. If the number of atoms of each element is the SAME on both sides, then the equation is balanced. Example: H 2 + O 2 H 2 O : Not Balanced 2 H 2 + O 2 2 H 2 O : Balanced https: //www. youtube. com/watch? v=zmdx. Mlb 88 Fs

Classifying Chemical Reactions • There are four types of chemical reactions • In chemical reactions substances may: • Break apart into simpler substances • Be combined to make more complex substances • Exchange parts • Synthesis Reactions • Occurs when two or more substances combine to make a more complex substance(s) • Examples: 2 H 2 + O 2 2 H 2 O 4 Fe + 3 O 2 2 Fe 2 O 3

Classifying Chemical Reactions • Decomposition Reactions • Occurs when more complex substances break down into simpler substances • Examples: H 2 O 2 H 2 + O 2 Ba. O Ba + O • Replacement Reactions • This occurs when one element replaces another in a reaction • There are two types of replacement reactions: Single Replacement and Double Replacement

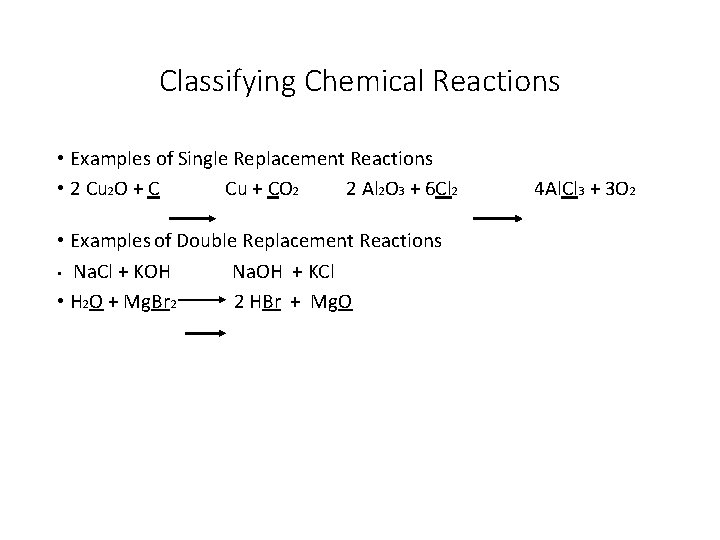
Classifying Chemical Reactions • Examples of Single Replacement Reactions • 2 Cu 2 O + C Cu + CO 2 2 Al 2 O 3 + 6 Cl 2 • Examples of Double Replacement Reactions • Na. Cl + KOH Na. OH + KCl • H 2 O + Mg. Br 2 2 HBr + Mg. O 4 Al. Cl 3 + 3 O 2

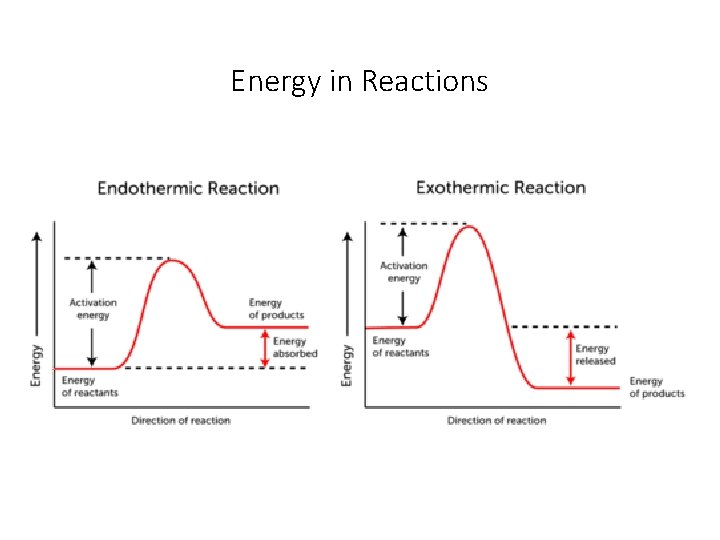
Energy in Reactions • All reactions need an energy input to get started. This is called the activation energy of the reaction.

Energy in Reactions • There are two types of chemical reactions---Exothermic and Endothermic • Both require energy to get started but only one needs energy to keep going • Exothermic reactions • • The products have less energy than the reactants No energy required to keep going Heat is given off Examples-----Burning wood, oil, gasoline We can use the heat that is given off to help keep us warm. The energy given off from the burning of gasoline helps to drive our cars. • Endothermic Reactions • Need additional heat to keep going • Products have more energy than the reactants

Energy in Reactions

Rates of Chemical Reactions • Chemical reactions do not all happen at the same rate. • Reactions like explosions or burning gunpowder happen very fast • Other reactions rusting of metal happen very slowly • For reactions to happen faster---more reactant particles need to get together faster with more energy. • To slow a reaction---the opposite needs to occur • Chemical reactions can be controlled by manipulating the factors that control the rates of reactions such as concentration, temperature, surface area and the use of catalysts and inhibitors • Surface Area • This is a direct relationship---the more surface area the faster the reaction will occur • When a solid reacts, only the particles on the surface are reacting

Rates of Chemical Reactions • By increasing the surface area the rate of the reaction will increase • Be careful though, this can be very dangerous • Temperature • By increasing the temperature the particles of reactants move faster. Faster moving particles increase the rate of reactions in two ways-1) they come into contact with each other more often and 2) faster moving particles have more energy • Lowering the temperature has the opposite effect • Concentration • The higher the concentration the faster the reaction will occur. • The higher the concentration the more likely reactant particles will collide leading to a faster reaction.

Rates of Chemical Reactions • Lowering the concentration will slow the rate of the reaction • Catalysts • Increase the rate of a reaction by lowering the activation energy required to start a reaction • Catalysts themselves are not permanently altered by a reaction, therefore they are not considered reactants. • • Inhibitors Decrease the rate of a chemical reaction Most inhibitors work by preventing reactants from coming together Typically inhibitors combine with one reactant, either temporarily or permanently. Food preservatives used to keep food from spoiling are a type of inhibitor.

Review • What is a chemical reaction? • A change in matter that produces one or more substances.

Review What are three signs a chemical reaction is taking place? • A color change • Bubbles form • A precipitate forms.

Review • What are three types of chemical reactions? • Combustion • Oxidation (Rusting) • Tarnishing • Neutralization

Review • List three ways You can speed up a chemical reaction. • Increase the temperature • Increase the surface area • Increase the concentration. • Add a catalyst.