CHEMICAL REACTIONS Chapter 11 Describing Chemical Change To

CHEMICAL REACTIONS Chapter 11

Describing Chemical Change: ■ To understand chemical reactions we need to describe them in writing. ■ Reactants vs. Products ■ Reactants = those substances that are reacting or changing ■ Products = those substances that are being formed from the reactants. ■ Reactants Products Yields

Combination (Synthesis) Reactions Two or more substances combine to form a new compound. A + X AX q. Must only have one product!!

Decomposition Reactions A single compound undergoes a reaction that produces two or more simpler substances AX A + X • Must only have one reactant!!

Single Replacement Reactions A + BX AX + B BX + Y BY + X • Must have an element reacting with a compound • MUST use activity series… more on this next week

Double Replacement Reactions The ions of two compounds exchange places in an aqueous solution to form two new compounds. AX + BY AY + BX • In order for this reaction to work, you must have a liquid, gas, or solid as a product. If not, it is not a reaction. More on this next week

Combustion Reactions • The burning of natural gas, wood, gasoline • Oxygen gas must be in the reactants. Carbon dioxide and water will be in the products. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(g)

Describing a Chemical Change: ■ Remember: All chemical reactions must follow the law of conservation of mass ■ What is this law?

Describing Chemical Change: ■ Skeleton Equation: – Does not indicate relative mole amounts – Shows only the formulas of the reactants and the products – Example: ■ Fe + O 2 Fe 2 O 3 – Not Balanced!! – Can also be written as a word equation: ■ Iron plus oxygen yields Iron (III) oxide

Describing a Chemical Change ■ You can also indicate the state of each substance by using symbols to represent solid (s), liquid (l), gas (g) and aqueous (aq) Example: Fe(s) + O 2 (g) Fe 2 O 3(s)

Balancing Chemical Equations ■ Balanced equations usually contain coefficients ■ Coefficients = numbers placed in front of the symbol/formula for each substance – Represents the number of moles ■ Coefficients are always whole numbers and in the lowest whole number ratio
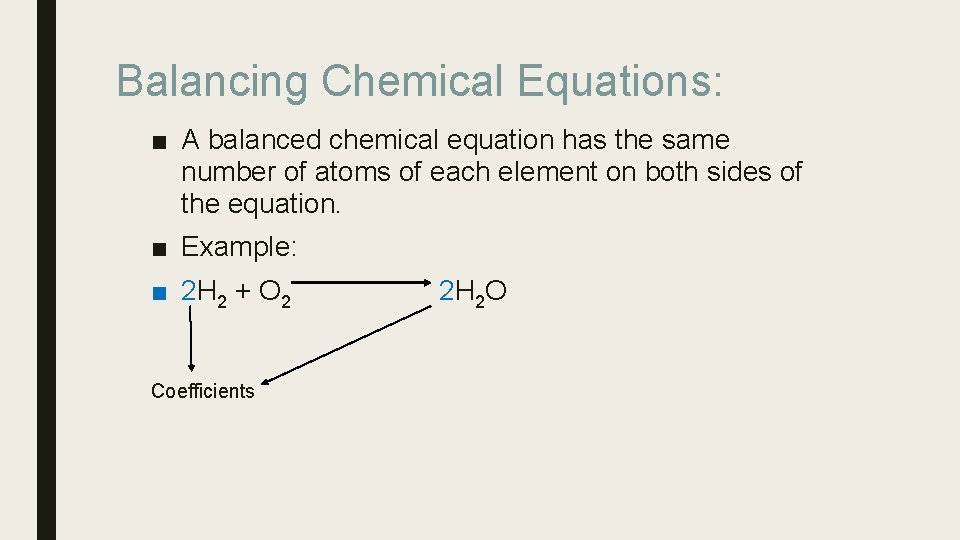
Balancing Chemical Equations: ■ A balanced chemical equation has the same number of atoms of each element on both sides of the equation. ■ Example: ■ 2 H 2 + O 2 Coefficients 2 H 2 O

Balancing Chemical Equations: ■ Some helpful tips: ■ 1. Solve the more complicated compounds first. Leave hydrogen and oxygen for the end. ■ 2. Polyatomic ions present on both sides of the equation count as one unit. ■ 3. When an element is present in 2 different spots on the same side, balance last.
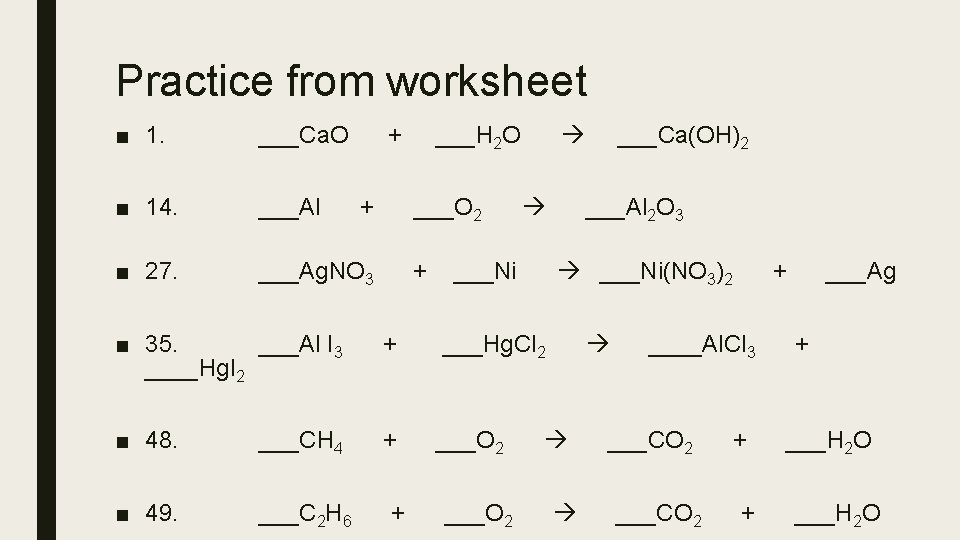
Practice from worksheet ■ 1. ___Ca. O ■ 14. ___Al ■ 27. ___Ag. NO 3 + + ___H 2 O ___O 2 + ___Ni(NO 3)2 + ___Hg. Cl 2 ■ 48. ___CH 4 + ___O 2 ■ 49. ___C 2 H 6 ___O 2 ___Al 2 O 3 ■ 35. ___Al I 3 ____Hg. I 2 + ___Ca(OH)2 + ____Al. Cl 3 ___CO 2 + + ___Ag + ___H 2 O
- Slides: 14