Chemical reactions can be classified into 4 types

Chemical reactions can be classified into 4 types: 1. 2. 3. 4. Synthesis Reactions Decomposition reactions Single Displacement Reactions Double Displacement Reactions

Synthesis Reaction: two or more elements unite to form one compound C + O 2 CO 2 2 H 2 + O 2 2 H 2 O 2 K + Cl 2 2 KCl

Decomposition Reaction: the breakdown of 1 substance into 2 or more other substances 2 H 2 O 2 H 2 + O 2 Ba. Cl 2 Ba + Cl 2 Mg. Cl 2 Mg + Cl 2

What do you notice about synthesis and decomposition reactions?

Single Displacement Reaction: 1 element replaces another in a compound Be. F 2 + Mg Mg. F 2 + Be 2 KBr + Cl 2 2 KCl + Br 2 2 Na. Br + Cl 2 2 Na. Cl + Br 2
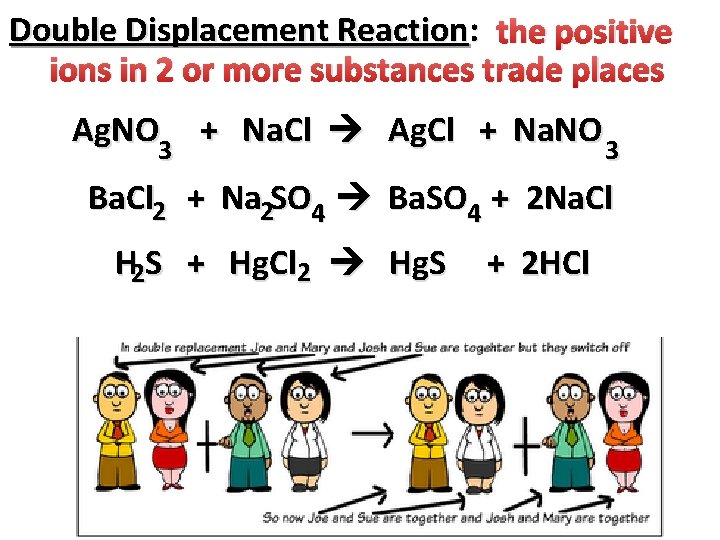
Double Displacement Reaction: the positive ions in 2 or more substances trade places Ag. NO 3 + Na. Cl Ag. Cl + Na. NO 3 Ba. Cl 2 + Na 2 SO 4 Ba. SO 4 + 2 Na. Cl H 2 S + Hg. Cl 2 Hg. S + 2 HCl
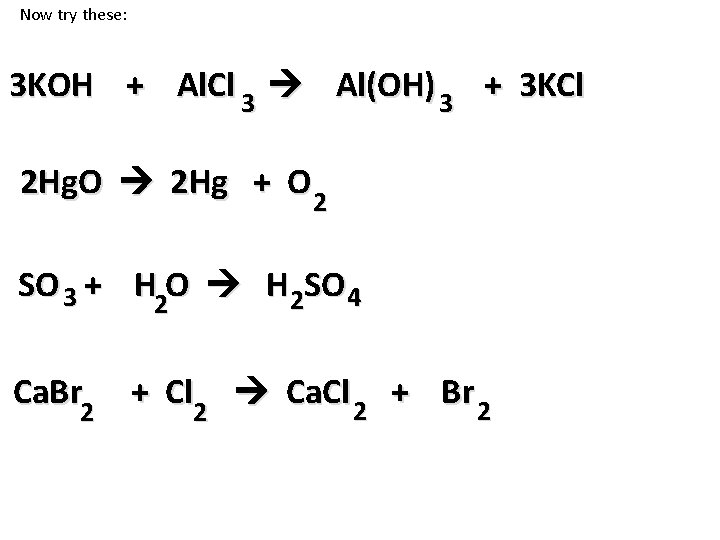
Now try these: 3 KOH + Al. Cl 3 Al(OH) 3 + 3 KCl 2 Hg. O 2 Hg + O 2 SO 3 + H 2 O H 2 SO 4 Ca. Br 2 + Cl 2 Ca. Cl 2 + Br 2
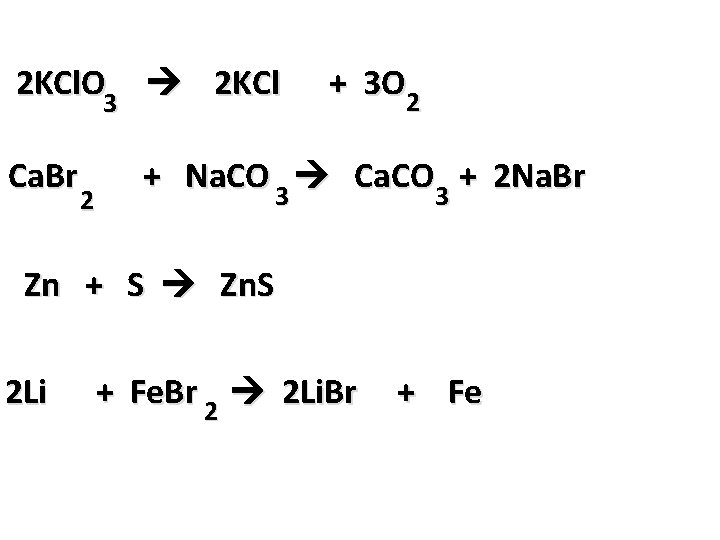
2 KCl. O 3 2 KCl Ca. Br 2 + 3 O 2 + Na. CO 3 Ca. CO 3 + 2 Na. Br Zn + S Zn. S 2 Li + Fe. Br 2 2 Li. Br + Fe
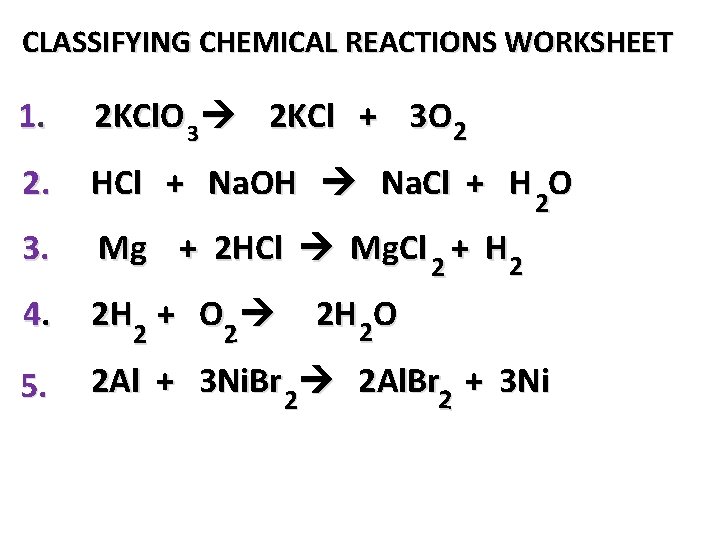
CLASSIFYING CHEMICAL REACTIONS WORKSHEET 1. 2 KCl. O 3 2 KCl + 3 O 2 2. HCl + Na. OH Na. Cl + H 2 O 3. Mg + 2 HCl Mg. Cl 2 + H 2 4. 2 H + O 2 2 5. 2 Al + 3 Ni. Br 2 2 Al. Br 2 + 3 Ni 2 2 H 2 O
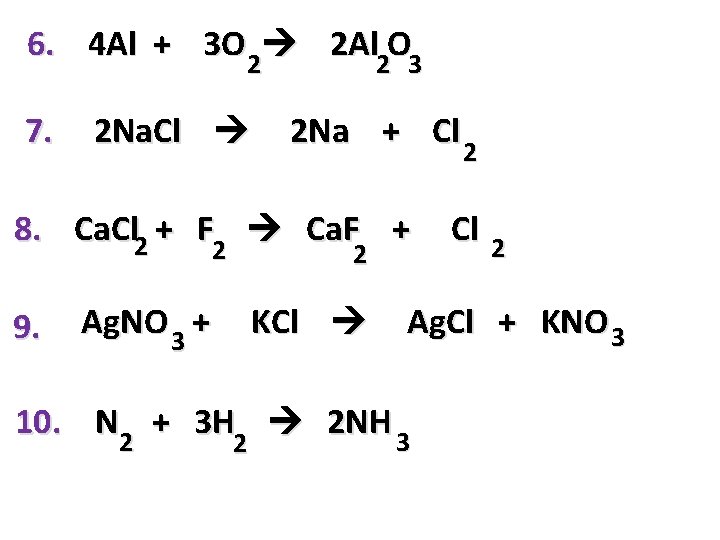
6. 4 Al + 3 O 2 2 Al 2 O 3 7. 2 Na. Cl 2 Na + Cl 2 2 8. Ca. Cl 2 + F Ca. F + 2 2 9. 2 Ag. NO 3 + KCl 2 Ag. Cl + KNO 3 10. N 2 + 3 H 2 NH 3 2 2 Cl
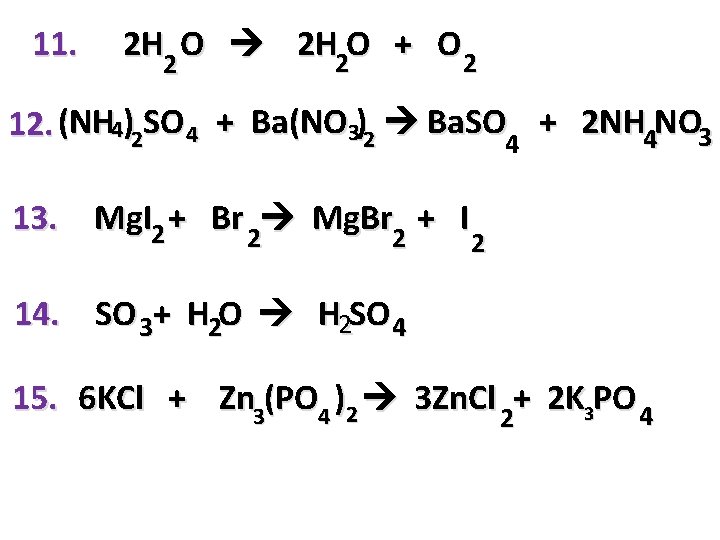
11. 2 H O 2 H 2 O + O 2 2 12. (NH 4)2 SO 4 + Ba(NO 3)2 Ba. SO 4 + 2 NH 4 NO 3 13. Mg. I 2 + Br 2 Mg. Br + I 2 2 14. SO 3+ H 2 O H 2 SO 4 15. 6 KCl + Zn 3(PO 4 )2 3 Zn. Cl 2+ 2 K 3 PO 4
- Slides: 11