CHEMICAL REACTIONS Balancing Equations PHYSICAL OR CHEMICAL CHANGE

CHEMICAL REACTIONS Balancing Equations

PHYSICAL OR CHEMICAL CHANGE? Physical changes affect only physical properties such as shape, size or state of matter Chemical changes produce new substances with properties different from any component of the substance The process that produces a chemical change is called a chemical reaction

CHEMICAL EQUATIONS The substances that you start with and react with one another are called REACTANTS The substances that are produced after the reaction are called PRODUCTS

CHEMICAL EQUATIONS Shorthand way to show a reaction Chemical equations use chemical symbols to explain chemical reactions Equations tell you the reactants, products, and physical state of each substance present

USING CHEMICAL NAMES Many household items have common names Baking Soda Vinegar Common names are not typically used in chemical equations Sodium Bicarbonate Acetic Acid

THE VINEGAR AND BAKING SODA REACTION… Acetic Acid + Sodium Bicarbonate Sodium Acetate + Water + Carbon Dioxide
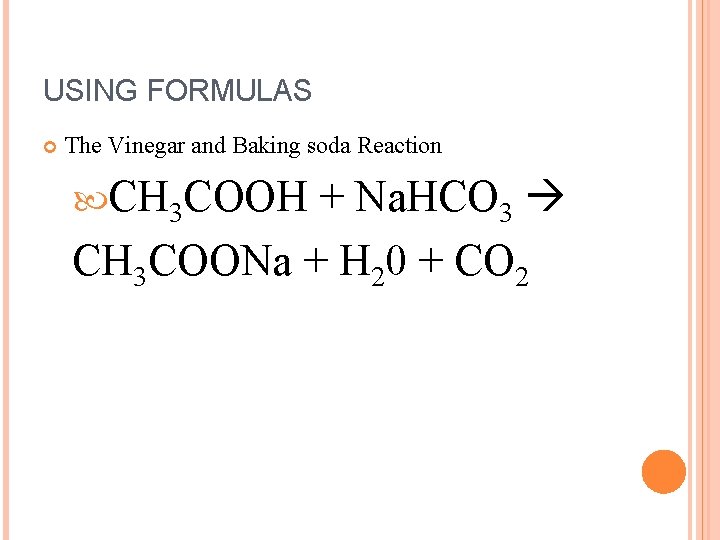
USING FORMULAS The Vinegar and Baking soda Reaction + Na. HCO 3 CH 3 COONa + H 20 + CO 2 CH 3 COOH

SUBSCRIPTS Those little numbers that are below the symbols If there is a number written, then there are exactly that many number of that atom If no number is written, then there is one atom present YOU CANNOT CHANGE A SUBSCRIPT!!! NEVER!!!! EVER!!!

USING A SUPERSCRIPT Silver + Hydrogen Sulfide Silver Chloride + Hydrogen Ag + H 2 S Ag 2 S + H 2 Is this equation balanced? What can we do to fix it?
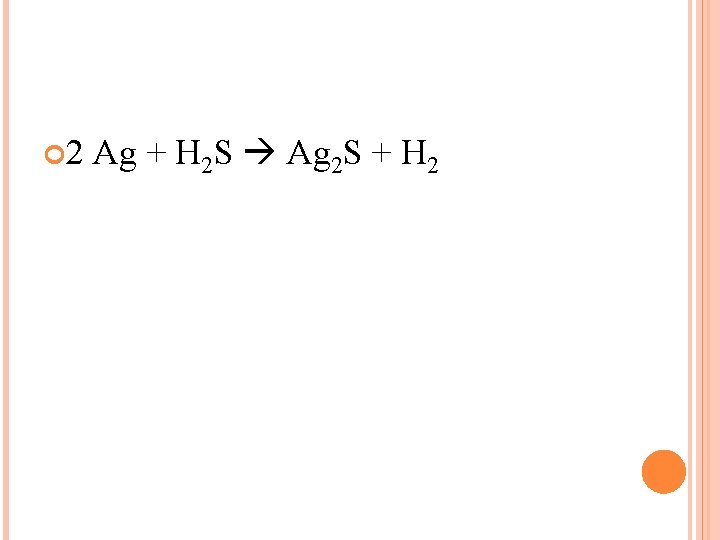
2 Ag + H 2 S Ag 2 S + H 2

BALANCE THESE EQUATIONS Na + Al. Cl 3 Na. Cl + Al KBr + Cl 2 KCl + Br 2 H 2 O 2 H 20 + O 2

TEAM BALANCING ACTIVITY Balance ALL of the given equations using the materials provided DO NOT write on the cards You may only change the COEFFICENTS Allow me to check each equation before moving on to the next equation
- Slides: 12