Chemical Reactions Balancing and Classifying Chemical Equations Chemical

Chemical Reactions Balancing and Classifying Chemical Equations

Chemical Reactions n Chemical reactions occur when a chemical change occurs

Evidence of a Chemical Reaction Color change n Formation of a gas n Odor change n Formation of a precipitate n Temperature change n p. H change n

Why do chemical reactions occur? n n Chemical reactions occur because the products are more stable than the reactants. All substances react in an effort to become more stable.

Common Symbols in Chemical Equations
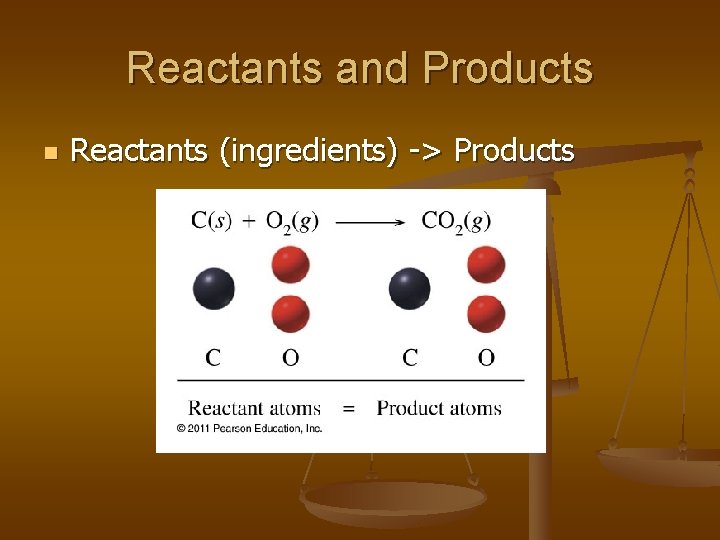
Reactants and Products n Reactants (ingredients) -> Products

n n How many oxygen atoms are in the reactants? How many oxygen atoms are in the products?

Balancing Equations

Balancing Equations n Law of Conservation of Matter (or mass): n In a chemical reaction, matter can be neither created nor destroyed. n In a chemical reaction, the amount of reactants equal the amount of products.

Balancing Equations Paraphrase: n Law of Conservation of Atoms: n n The number of atoms of each type of element must be the same on each side of the equation.

Balancing Equations n Hydrogen n H 2 n n n + oxygen + O 2 water H 2 O Hydrogen and oxygen are diatomic elements. Their subscripts cannot be changed. The subscripts on water cannot be changed.

Balancing Equation n H 2 n + O 2 H 2 O Count the atoms on each side. n Reactant side: 2 atoms H and 2 atoms O n Product side: 2 atoms H and 1 atom O

Balancing Equations n H 2 n n + O 2 H 2 O If the subscripts cannot be altered, how can the atoms be made equal? Adjust the number of molecules by changing the coefficients.

Balancing Equations n H 2 + O 2 2 H 2 O Reactants: 2 atoms of H and 2 atoms of O n Products: 4 atoms of H and 2 atoms of O n H is no longer balanced! n

Balancing Equations n 2 H 2 n n n + O 2 2 H 2 O Reactant side: 4 atoms of H and 2 atoms of O Product side: 4 atoms of H and 2 atoms of O It’s Balanced!

Balancing Equations n 2 H 2 + O 2 2 H 2 O

Balancing Equations n Balancing n Count hints: all atoms and polyatomic ions occurring on each side of -> n Balance the metals first. n Balance the polyatomic ions next. n Balance the other atoms. n Save the oxygen and hydrogen until the end.
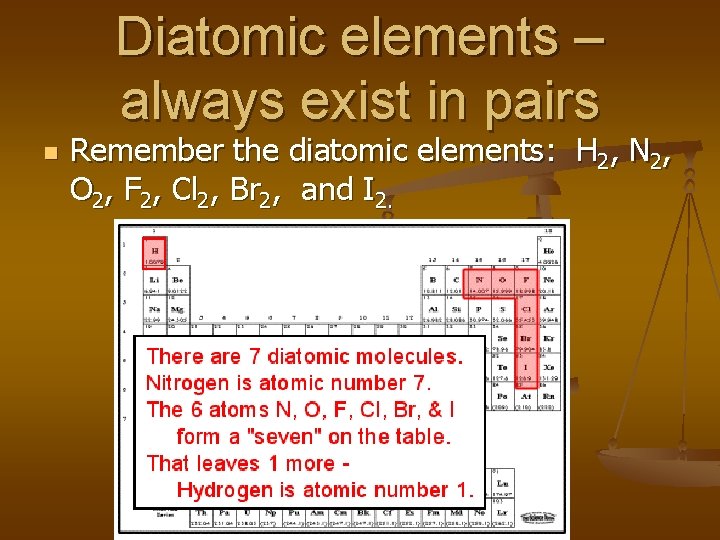
Diatomic elements – always exist in pairs n Remember the diatomic elements: H 2, N 2, O 2, F 2, Cl 2, Br 2, and I 2.

Types of Reactions n 1. Synthesis (or Addition or Combination) Reactions A+B AB Ø 2 or more substances combine to form a new compound. (A and B are elements or compounds) Ø small molecules join to form chains polymerization Fe + O 2 Fe 2 O 3

Types of Reactions n 2. Decomposition Reactions AB A+B Ø compounds are broken apart into smaller substances Example: Electrolysis of water 2 H 2 O 2 H 2 + O 2

Types of Reactions n Ø Ø Single – Replacement Reaction n. AX + B BX + A One element replaces a similar element in a compound Fe + Cu. Cl 2 Fe. Cl 2 + Cu Ø Look at the Metals activity series

Types of Reactions n Ø Double – Replacement Reaction AX + BY AY + BX Ions from 2 compounds in solution exchange places to produce 2 new compounds. Ø One of the compounds is usually … Ø A precipitate Ø A gas that bubbles off Ø A molecular compound like water

Types of Reactions n Combustion Reactions n These use O 2 as a reactant n One product contains O , usually H 2 O ______________________________________________________________ Importance of Good Air Supply 2 CH 4 + 4 O 2 2 CO 2 + 4 H 2 O 2 CH 4 + 3 O 2 2 CO + 4 H 2 O 2 CH 4 + 2 O 2 2 C + 4 H 2 O
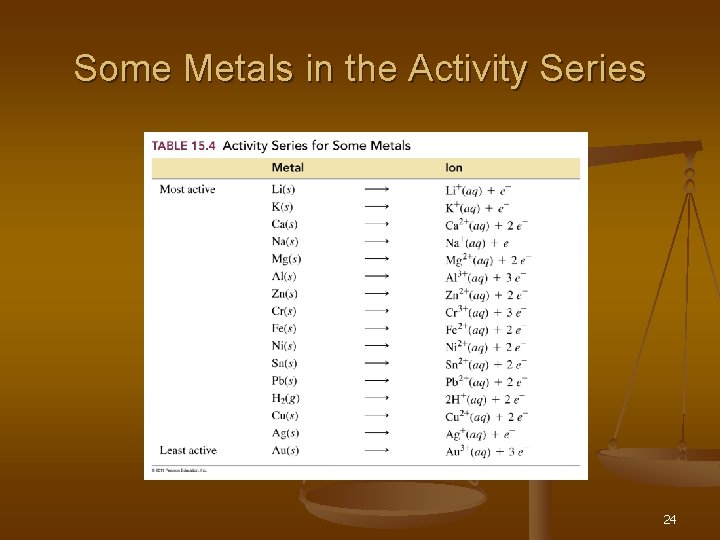
Some Metals in the Activity Series 24
- Slides: 24