Chemical Reactions Aqueous Solubility of Ionic Compounds Not

Chemical Reactions
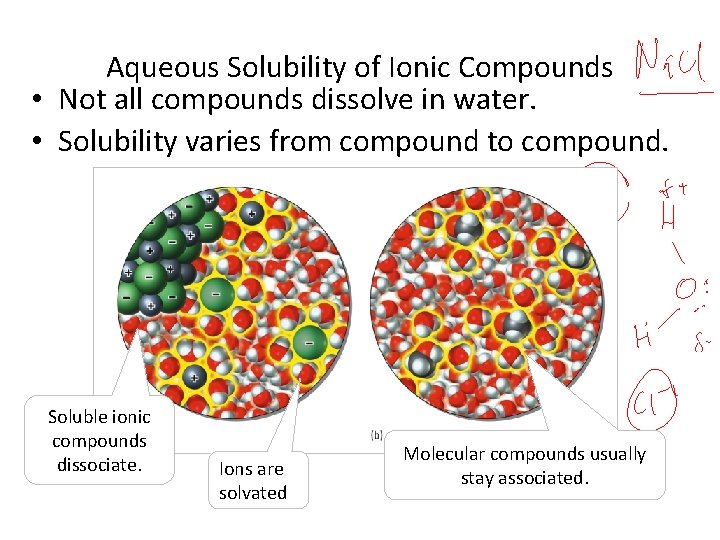
Aqueous Solubility of Ionic Compounds • Not all compounds dissolve in water. • Solubility varies from compound to compound. Soluble ionic compounds dissociate. Ions are solvated Molecular compounds usually stay associated.

ALL Soluble Ionic Compounds • Ammonium and group 1 A (Na+, K+… and NH 4+ salts) • Nitrates (NO 3 -) • Acetates (CH 3 COO-) • Chlorates (Cl. O 3 -) • Perchlorates (Cl. O 4 -) MOST • Chlorides, bromides and iodides § (not: Ag. X, Hg 2 X 2, and Pb. X 2 ; X = Cl-, Br-, I-). • Sulfates (SO 42 -) § (not: Ca. SO 4, Sr. SO 4, Ba. SO 4, and Pb. SO 4)

Insoluble Ionic Compounds Insoluble Salts • Phosphates (PO 43 -) • Carbonates (CO 32 -) • Oxalates (C 2 O 42 -) • Oxides (O 2 -) • Sulfides (S 2 -) Cd. S Sb 2 S 3 Pb. S § Mg. S, Ca. S & Ba. S are slightly soluble. • Hydroxides (OH-) § Sr(OH)2, Ba(OH)2 & Ca(OH)2 are slightly soluble. Remember ALL group 1 A & NH 4+ salts are soluble (NH 4)2 S
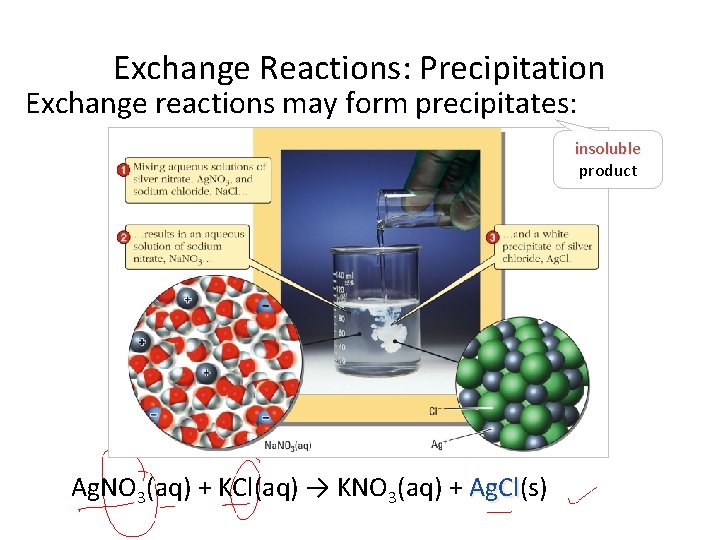
Exchange Reactions: Precipitation Exchange reactions may form precipitates: insoluble product Ag. NO 3(aq) + KCl(aq) → KNO 3(aq) + Ag. Cl(s) Ag. Cl

Precipitation Reactions A precipitate forms whenever a “reaction product” is insoluble. You must know the solubility rules: Pb(NO 3)2(aq) + 2 Li. I(aq) → Pbl 2(s) + 2 Li. NO 3(aq) Insoluble I- = usually soluble (Pb 2+ is an exception) Soluble NO 3 - = soluble Grp 1 A = soluble An aqeuous solution of sodium sulfate is added dropwise to an aqueous barium nitrate solution. Will a precipitate form?

Precipitation Reactions Na 2 SO 4(aq) + Ba(NO 3)2(aq) → precipitate? Possible products? The positive and negative ions can exchange partners: • Na. NO 3 • Ba. SO 4 Is either “product” insoluble?

Net Ionic Equations Soluble ionic compounds fully dissociate in water: Ag. NO 3(aq) → Ag+(aq) + NO 3 -(aq) KCl(aq) → K+(aq) + Cl-(aq) On mixing: Ag+(aq) + NO 3 -(aq) + K+(aq) + Cl-(aq) → Ag. Cl(s) + K+(aq) + NO 3 -(aq) K+ and NO 3 - appear on both sides § NO 3 - and K+ are spectator ions. § They are not directly involved in the reaction. Net ionic equation: Ag+(aq) + Cl-(aq) → Ag. Cl(s)

Net Ionic Equations General rules: 1. Write a balanced equation. 2. Dissociate soluble compounds. 3. Write the complete ionic equation. 4. Cancel ions appearing on both sides (spectators). 5. Check the charges are balanced.

Net Ionic Equations Aqueous solutions of (NH 4)2 S and Hg(NO 3)2 react to give Hg. S and NH 4 NO 3. (a) Write the overall balanced equation (b) Name each compound (c) Write the net ionic equation (d) What type of reaction is this? (NH 4)2 S(aq) + Hg(NO 3)2(aq) → Hg. S + NH 4 NO 3 Not balanced (NH 4)2 S(aq) + Hg(NO 3)2(aq) → Hg. S + 2 NH 4 NO 3 Balanced
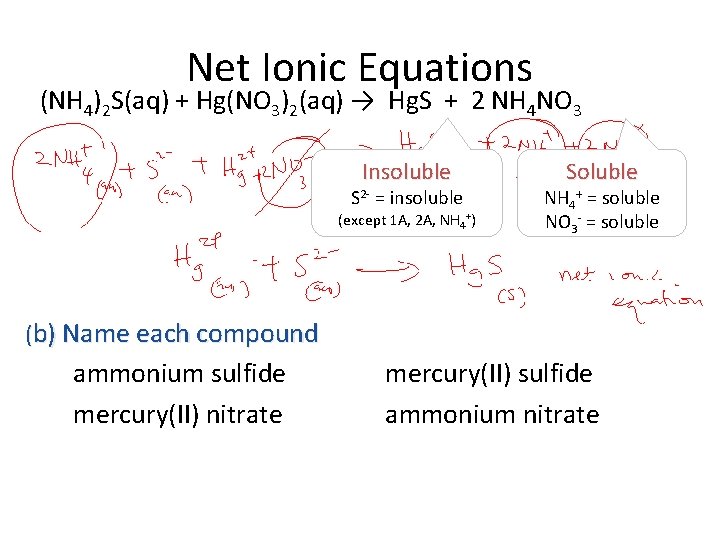
Net Ionic Equations (NH 4)2 S(aq) + Hg(NO 3)2(aq) → Hg. S + 2 NH 4 NO 3 Insoluble S 2 - = insoluble (except 1 A, 2 A, NH 4+) Soluble NH 4+ = soluble NO 3 - = soluble (b) Name each compound ammonium sulfide mercury(II) nitrate mercury(II) sulfide ammonium nitrate

Net Ionic Equations (c) Write a net ionic equation (NH 4)2 S(aq) + Hg(NO 3)2(aq) → Hg. S(s) + 2 NH 4 NO 3(aq) 2 NH 4+(aq) + S 2 -(aq) + Hg 2+(aq) + 2 NO 3 -(aq) → Hg. S(s) + 2 NH 4+(aq) + 2 NO 3 -(aq) S 2 -(aq) + Hg 2+(aq) → Hg. S(s) (d) Type of reaction? Exchange reaction Precipitation reaction

Acids Increase the concentration of H+ ions in water. • Protons (H+) always combine with water to form H 3 O+ (hydronium ion). ion • Sour tasting. • Change the color of pigments (indicators) § Litmus, phenolphthalein… Strong acids dissociate (>99%) in water (strong electrolytes). Weak acids partially dissociate (weak electrolytes). H 3 O+
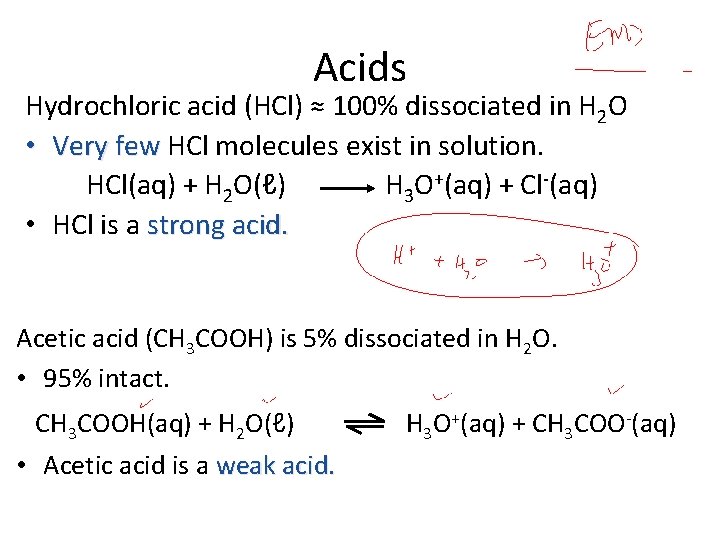
Acids Hydrochloric acid (HCl) ≈ 100% dissociated in H 2 O • Very few HCl molecules exist in solution. HCl(aq) + H 2 O(ℓ) H 3 O+(aq) + Cl-(aq) • HCl is a strong acid. Acetic acid (CH 3 COOH) is 5% dissociated in H 2 O. • 95% intact. CH 3 COOH(aq) + H 2 O(ℓ) • Acetic acid is a weak acid. H 3 O+(aq) + CH 3 COO-(aq)
Bases Increase the concentration of OH- (hydroxide ion) in water. Bases: • • Counteract an acid (neutralize an acid). Change an indicator’s color (phenolphthalein…). Have a bitter taste. Feel slippery. Bases can be “strong” or “weak”. H 2 O Na. OH(s) Na+(aq) + OH-(aq) strong NH 3(aq) + H 2 O(ℓ) weak NH 4+(aq) + OH-(aq)
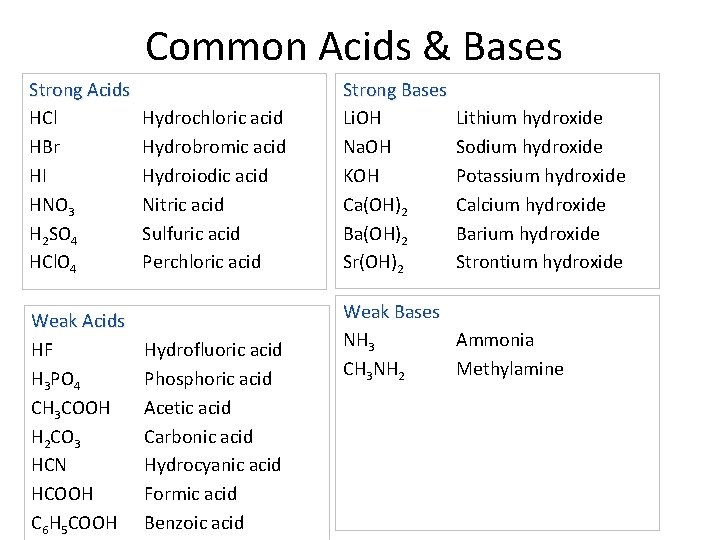
Common Acids & Bases Strong Acids HCl HBr HI HNO 3 H 2 SO 4 HCl. O 4 Weak Acids HF H 3 PO 4 CH 3 COOH H 2 CO 3 HCN HCOOH C 6 H 5 COOH Hydrochloric acid Hydrobromic acid Hydroiodic acid Nitric acid Sulfuric acid Perchloric acid Hydrofluoric acid Phosphoric acid Acetic acid Carbonic acid Hydrocyanic acid Formic acid Benzoic acid Strong Bases Li. OH Na. OH KOH Ca(OH)2 Ba(OH)2 Sr(OH)2 Lithium hydroxide Sodium hydroxide Potassium hydroxide Calcium hydroxide Barium hydroxide Strontium hydroxide Weak Bases NH 3 Ammonia CH 3 NH 2 Methylamine
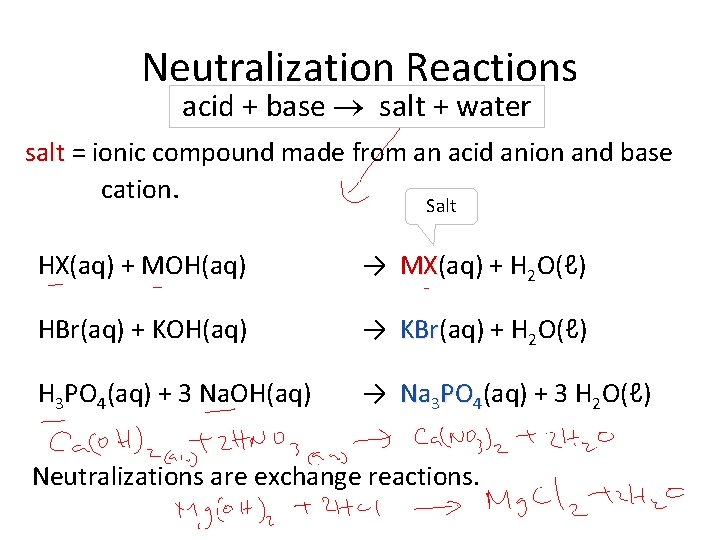
Neutralization Reactions acid + base salt + water salt = ionic compound made from an acid anion and base cation. Salt HX(aq) + MOH(aq) → MX(aq) + H 2 O(ℓ) MX HBr(aq) + KOH(aq) → KBr(aq) + H 2 O(ℓ) KBr H 3 PO 4(aq) + 3 Na. OH(aq) → Na 3 PO 4(aq) + 3 H 2 O(ℓ) Neutralizations are exchange reactions.
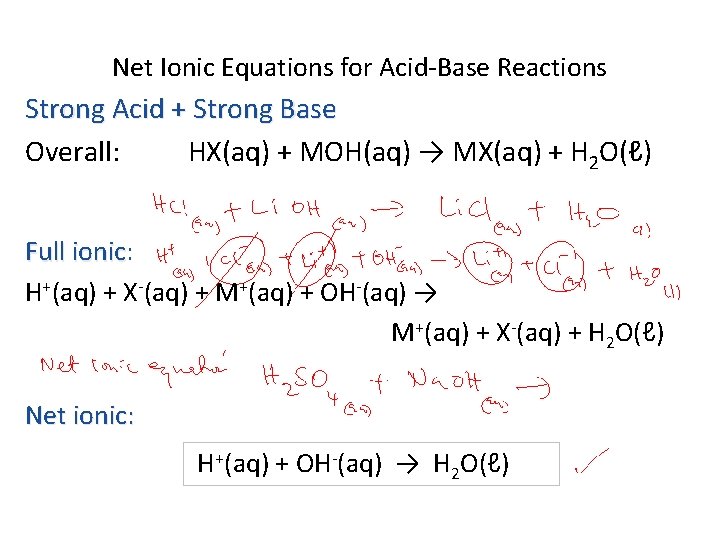
Net Ionic Equations for Acid-Base Reactions Strong Acid + Strong Base Overall: HX(aq) + MOH(aq) → MX(aq) + H 2 O(ℓ) Full ionic: ionic H+(aq) + X-(aq) + M+(aq) + OH-(aq) → M+(aq) + X-(aq) + H 2 O(ℓ) Net ionic: H+(aq) + OH-(aq) → H 2 O(ℓ)

Net Ionic Equations for Acid-Base Reactions Weak Acid + Strong Base Similar: HA(aq) + MOH(aq) → MA(aq) + H 2 O(ℓ) but the weak-acid remains (mostly) undissociated: Full ionic HA(aq) + M+(aq) + OH-(aq) → M+(aq) + A-(aq) + H 2 O(ℓ) Net ionic: HA(aq) + OH-(aq) → A-(aq) + H 2 O(ℓ)
Gas-Forming Exchange Reactions Exchange reactions can often form a molecular gas: Metal carbonate/acid exchange Ca. CO 3(s) + 2 HCl(aq) → Ca. Cl 2(aq) + H 2 CO 3(aq) Tums® H 2 CO 3(aq) → H 2 O(ℓ) + CO 2(g) Carbonic acid (unstable) often written combined: Ca. CO 3(s) + 2 HCl(aq) → Ca. Cl 2(aq) + H 2 O(ℓ) + CO 2(g) Net ionic: ionic Ca. CO 3(s) + 2 H+(aq) → Ca 2+(aq) + H 2 O(ℓ) + CO 2(g)

Gas-Forming Exchange Reactions Metal hydrogen carbonate/acid exchange Na. HCO 3(aq) + HCl(aq)→ Na. Cl(aq) + H 2 O(ℓ) + CO 2(g) Alka-Seltzer ® Net ionic HCO 3 -(aq) + H+(aq) → H 2 O(ℓ) + CO 2(g)
Gas-Forming Exchange Reactions Metal sulfite/acid exchange Sulfurous acid (unstable) Similar: Na 2 SO 3(aq) + 2 HCl(aq) → 2 Na. Cl(aq) + H 2 SO 3(aq) → H 2 O(ℓ) + SO 2(g) or Na 2 SO 3(aq) + 2 HCl(aq) → 2 Na. Cl(aq) + H 2 O(ℓ) +SO 2(g) Net ionic: SO 32 -(aq) + 2 H+(aq) → H 2 O(ℓ) + SO 2(g)
Gas-Forming Exchange Reactions Metal sulfide/acid exchange Na 2 S(aq) + 2 HCl(aq) → 2 Na. Cl(aq) + H 2 S(g) Net ionic: S 2 -(aq) + 2 H+(aq) → H 2 S(g)

Oxidation-Reduction Reactions Oxidation Originally: add oxygen. 2 Mg(s) + O 2(g) → 2 Mg. O(s) 2 CO(g) + O 2(g) → 2 CO 2(g) Mg and CO are oxidized O 2 is the oxidizing agent for both. Compound causing the oxidation Mg(s) burning in air

Oxidation-Reduction Reactions Reduction • Originally: reduce ore to metal. • reverse of oxidation. H 2 → H 2 O (oxidation) Cu. O = ox. agent Cu. O(s) + H 2(g) → Cu(s) + H 2 O Sn. O 2(s) + 2 C(s) → Sn(s) + 2 CO(g) Cu. O and Sn. O 2 are reduced H 2 and C are the reducing agents Compound causing the reduction C → CO (oxidation) Sn. O 2 = ox. agent

Oxidation-Reduction Reactions In all cases: • If a species is oxidized, another must be reduced. • Oxidationreduction = redox. Ox redox • Redox reactions move e-. Copper wire… +2 e- 2 Ag+(aq) + Cu(s) → 2 Ag(s) + Cu 2+(aq) -2 e- Here: …in Ag. NO 3 • Ag+ is reduced. • Reduction = gain of e- (so, loss of e- = oxidation)

Redox Reactions & Electron Transfer Loss of electrons is oxidation Gain of electrons is reduction Leo says ger Oxidation is loss Reduction is gain Oil rig
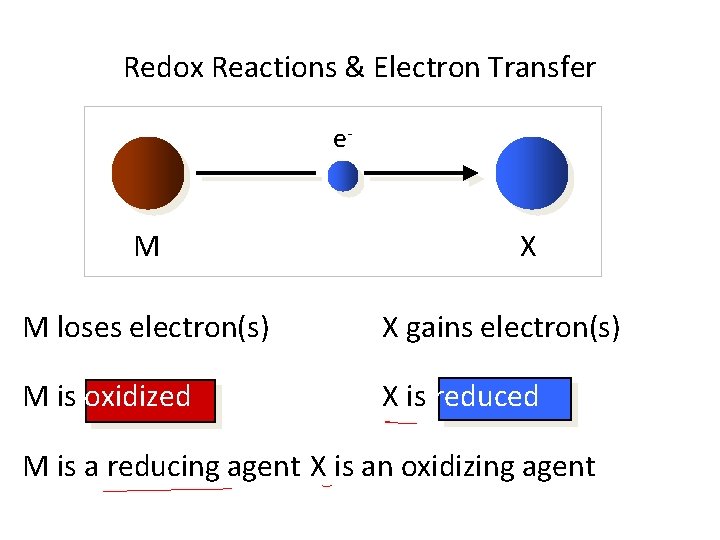
Redox Reactions & Electron Transfer e- M X M loses electron(s) X gains electron(s) M is oxidized X is reduced M is a reducing agent X is an oxidizing agent

Common Oxidizing & Reducing Agents Oxidizing Agent O 2 (oxygen) H 2 O 2 (hydrogen peroxide) F 2, Cl 2, Br 2, I 2 (halogen) HNO 3 (nitric acid) Cr 2 O 7 - (dichromate ion) Mn. O 4 - (permanganate ion) Reaction Product O 2 - (oxide ion) H 2 O(ℓ) F-, Cl-, Br-, I- (halide ion) Nitrogen oxides (NO, NO 2…) Cr 3+ (chromium(III) ion) Mn 2+ (manganese(II) ion) Reducing Agent H 2 (hydrogen) C M (metal: Na, K, Fe …) Reaction Product H+ or H 2 O CO and CO 2 Mn+ (Na+, K+, Fe 3+…)

Oxidation Numbers & Redox Reactions Oxidation number (Ox) Compares the charge of an uncombined atom with its relative charge in a compound. Rules: § Pure element: Ox = 0. § Monatomic ion: Ox = charge of ion. § Sum of Ox of all atoms = compound charge. § Ox is often constant for certain elements…

Oxidation Numbers & Redox Reactions Element Ox Exceptions F − 1 None O − 2 Metal peroxides (MO 2) = -1 Includes H 2 O 2 Halogen oxides (OXn) (varies) Cl, Br, I − 1 Interhalogens (“strongest” = -1; Cl > Br >I) H +1 Metal hydrides (e. g. Na. H) = -1 Determine the oxidation numbers for P 2 O 5. Not MO 2 or OXn Ox(O) = -2 P 2 O 5 2 Ox(P) + 5 Ox(O) = 0 Charge 2 Ox(P) + 5(-2) = 0 Ox(P) = 10/2 = +5

Oxidation Numbers & Redox Reactions Find ox. numbers for all elements in SO 32 - and H 2 PO 4 SO 32 - Ox(O) = -2 (not MO 2, not bonded to halogen) Ox(S) + 3 Ox(O) = -2 Ion charge Ox(S) + 3(-2) = -2 Ox(S) = +4 H 2 PO 4 - Ox(O) = -2; -2 Ox(H) = +1 2 Ox(H) + Ox(P) + 4 Ox(O) = -1 2(+1) + Ox(P) + 4(-2) = -1 Ox(P) = +5
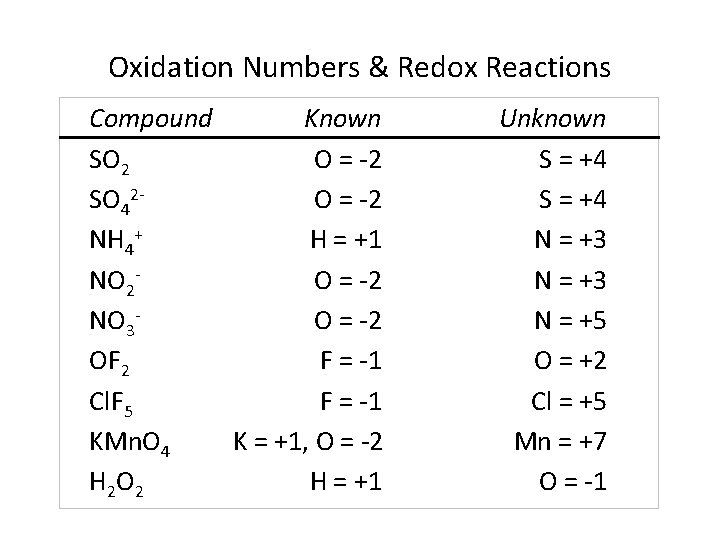
Oxidation Numbers & Redox Reactions Compound Known SO 2 O = -2 SO 42 O = -2 NH 4+ H = +1 NO 2 O = -2 NO 3 O = -2 OF 2 F = -1 Cl. F 5 F = -1 KMn. O 4 K = +1, O = -2 H 2 O 2 H = +1 Unknown S = +4 N = +3 N = +5 O = +2 Cl = +5 Mn = +7 O = -1

Oxidation Numbers & Redox Reactions Ox always change during redox reactions: Increase Ox = Oxidation Decrease Ox = Reduction It’s a redox reaction if: or: element → compound → element S 8(s) + 8 O 2(g) → 8 SO 2(g) Ox(S) : 0 → +4 Ox(O) : 0 → -2 (oxidized: lost e- ; increased Ox) (reduced: gained e- ; decreased Ox)

Oxidation Numbers & Redox Reactions A more complex example: Cu(s)+4 H+(aq)+2 NO 3 -(aq) → Cu 2+(aq)+2 NO 2(g)+2 H 2 O(ℓ) 0 +1 +5 -2 +2 Cu is oxidized (Ox ↑; loss of e-). H is unchanged. O is unchanged. N is reduced (Ox ↓; gain of e-). +4 -2 +1 -2

Oxidation Numbers & Redox Reactions Exchange reactions are not redox – no change in oxidation state occurs. +2 +1 -1 +1 +2 -1 Pb(NO 3)2(aq) + 2 Kl(aq) 2 KNO 3(aq) + Pbl 2(s) NO 3 -: Ox(O) = -2; Ox(N) = +5

Displacement Reactions + A + XZ Redox: Fe(s) + Cu. SO 4(aq) 0 +2 Cu(s) + 2 Ag. NO 3(aq) 0 +1 AZ X → Fe. SO 4(aq) + Cu(s) +2 0 → Cu(NO 3)2(aq) + 2 Ag(s) +2 0 Not all metals can displace another from its salts: Cu(s) + Zn. SO 4(aq) no reaction An activity series was developed…
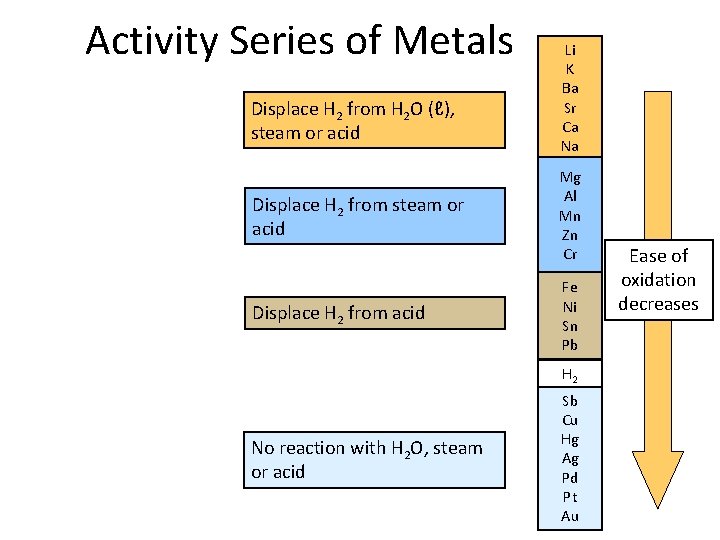
Activity Series of Metals Displace H 2 from H 2 O (ℓ), steam or acid Displace H 2 from acid Li K Ba Sr Ca Na Mg Al Mn Zn Cr Fe Ni Sn Pb H 2 No reaction with H 2 O, steam or acid Sb Cu Hg Ag Pd Pt Au Ease of oxidation decreases

Displacement Reactions Powerful reducing agents at the top. Higher elements displace lower ones: Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Mg(s) + 2 HCl(aq) → Mg. Cl 2(aq) + H 2(g) Metals at the bottom are unreactive. • Coinage metals • Their ions are powerful oxidizing agents. Li K : : Na Mg : Zn Cr Fe : Pb H 2 Sb Cu : Ag : Au
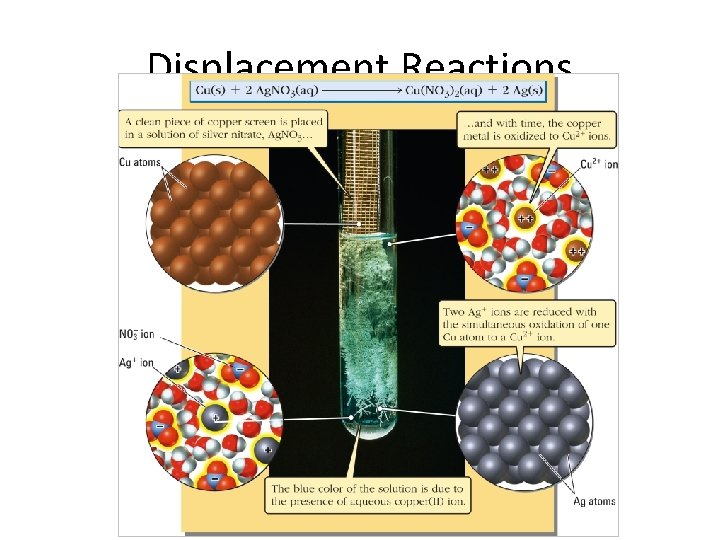
Displacement Reactions

Displacement Reactions 2 Na(s) + 2 H 2 O(ℓ) → 2 Na. OH(aq) + H 2(g)
- Slides: 41