Chemical Reactions and Law of Conservation of Mass











- Slides: 13
Chemical Reactions and Law of Conservation of Mass

Chemical reactions are everywhere! • Chemical Reactions: the process by which the atoms of one or more substances are rearranged to form different substances.

Chemical Equations Reactants Products • Reactants: starting substances • Products: substances formed during the reaction • means “yield” or “react to produce”

Law of Conservation of Mass is neither created nor destroyed during chemical or physical reactions. Antoine Lavoisier Total mass of reactants = Total mass of products

The Law of conservation of mass states that matter cannot be created or destroyed in any chemical reaction The atoms in the reactants are rearranged to form new compounds, but none of the atoms disappear, and no new atoms are formed. H H O Reactants O Products

Because of this law • When we write chemical equations, we have to make sure that they are balanced – In other words when we finish there has to be the same number of atoms on the left side as the right side

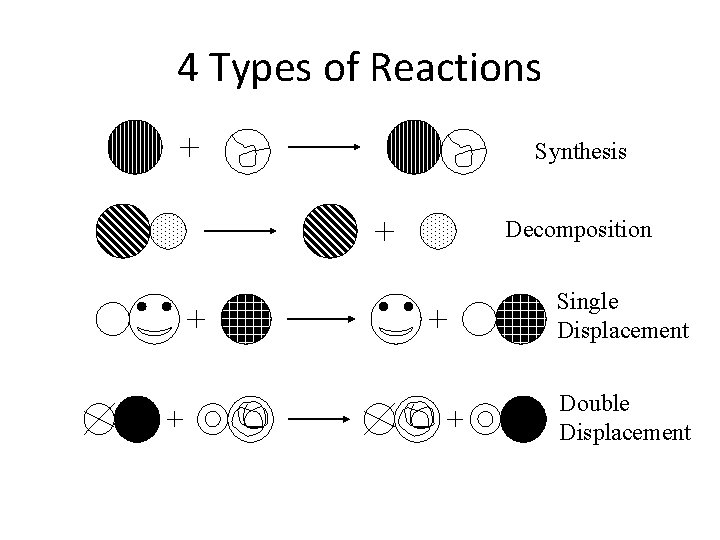
4 main types of chemical reactions • • Synthesis Decomposition Single Displacement Double Displacement

Synthesis Reaction A new substance forms!!! • A + B AB • Ex. metal + oxygen metal oxide • 2 Mg (s) + O 2 (g) 2 Mg. O (s)

Decomposition Reaction A compound is broken down (decomposed) AB A + B Ex. salt metal + nonmetal 2 Na. Cl (s) 2 Na (s) + Cl 2 (g)

Single-Displacement Reaction • An element in a compound is replaced by another element A + BC B + AC or A + BC C + BA Ex. metal + acid hydrogen + salt • Zn (s) + 2 HCl (aq) H 2 (g) + Zn. Cl 2 (aq) Ex. metal + salt • Fe (s) + Cu. SO 4 (aq) Cu (s) + Fe. SO 4 (aq)

Double-Displacement Reaction • Elements in two or more compounds all switch around AB + CD AD + CB Ex. acid + base salt + water • HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l)

Can you Classify the following picture reactions? 1 2 3 4

4 Types of Reactions Synthesis Decomposition Single Displacement Double Displacement