Chemical Reactions and Equations Chemical Changes Balancing Chemical

Chemical Reactions and Equations Chemical Changes Balancing Chemical Equations 1

Physical Properties l l l l color melting point boiling point electrical conductivity specific heat density state (solid, liquid, or gas) 2

Physical Changes in physical properties l melting l boiling lcondensation No change occurs in the identity of the substance Example: Ice , rain, and steam are all water 3

Chemical Change l. Atoms in the reactants are rearranged to form one or more different substances (products) l. Old bonds are broken; new bonds form Examples: Fe and O 2 form rust (Fe 2 O 3) Ag and S form tarnish ( Ag 2 S) 4

Check-In Classify each of the following as a 1) physical change or 2) chemical change A. ____ a burning candle B. ____ melting ice C. ____ toasting a marshmallow D. ____ cutting a pizza E. ____ polishing silver 5
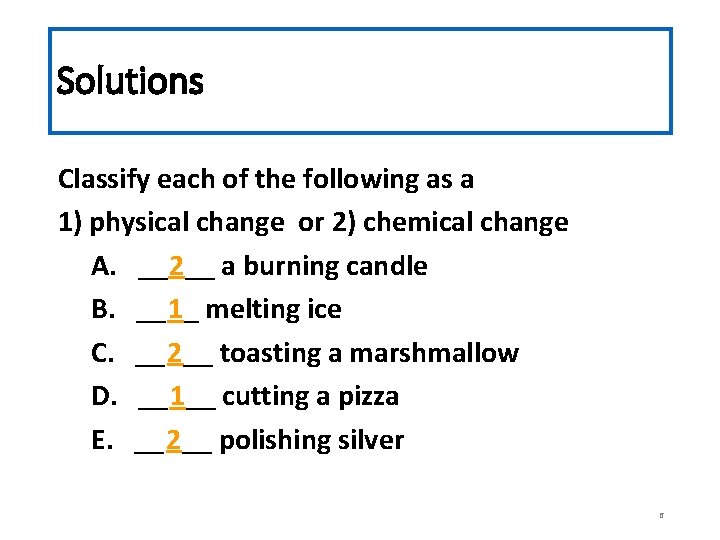
Solutions Classify each of the following as a 1) physical change or 2) chemical change A. __2__ a burning candle B. __1_ melting ice C. __2__ toasting a marshmallow D. __1__ cutting a pizza E. __2__ polishing silver 6

Chemical Reaction A process in which at least one new substance is produced as a result of chemical change. 7
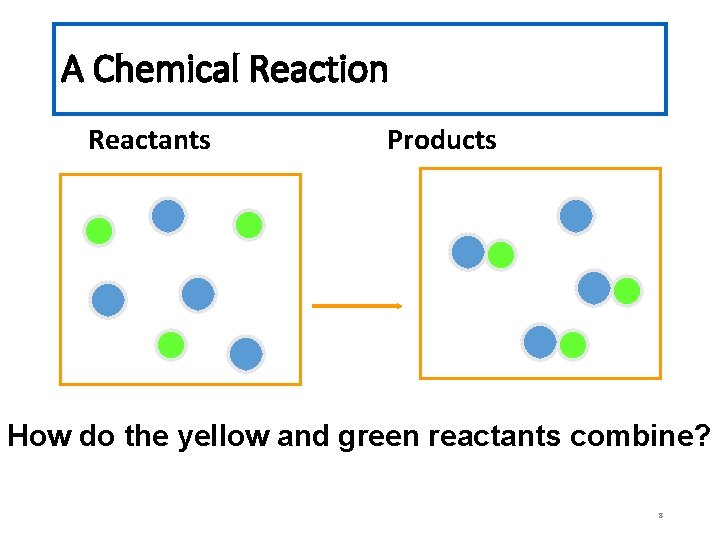
A Chemical Reaction Reactants Products How do the yellow and green reactants combine? 8
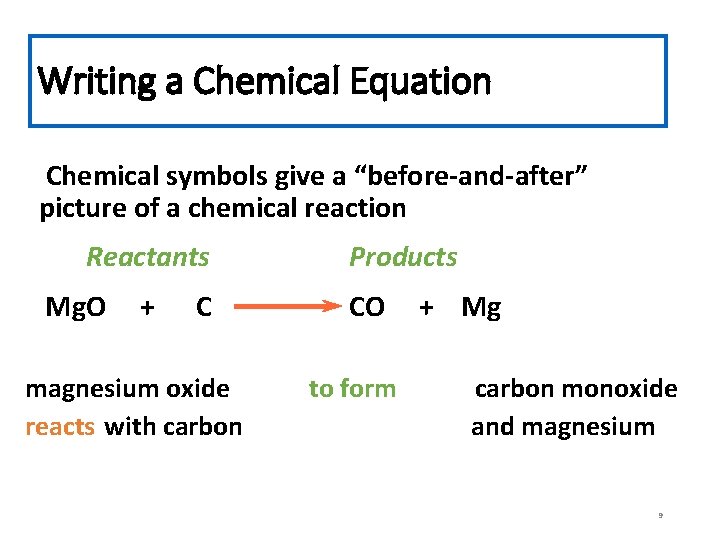
Writing a Chemical Equation Chemical symbols give a “before-and-after” picture of a chemical reaction Reactants Mg. O + C magnesium oxide reacts with carbon Products CO to form + Mg carbon monoxide and magnesium 9
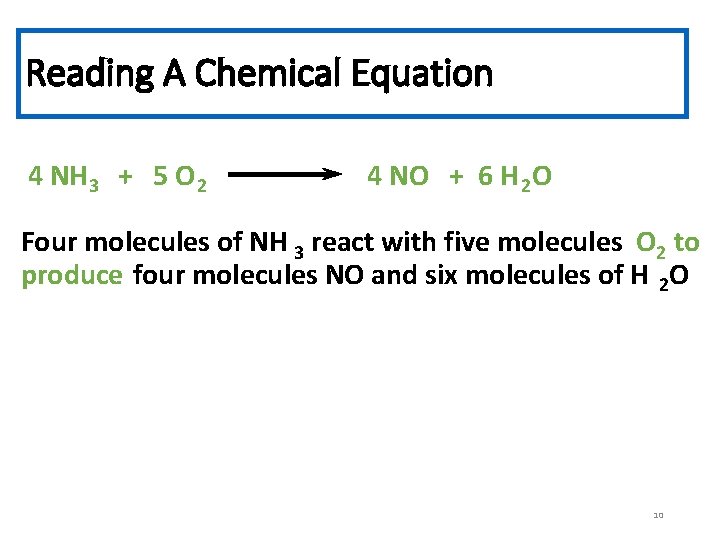
Reading A Chemical Equation 4 NH 3 + 5 O 2 4 NO + 6 H 2 O Four molecules of NH 3 react with five molecules O 2 to produce four molecules NO and six molecules of H 2 O 10

A Balanced Chemical Equation Same numbers of each type of atom on each side of the equation Al + S Al 2 S 3 2 Al + 3 S Al 2 S 3 Not Balanced 11
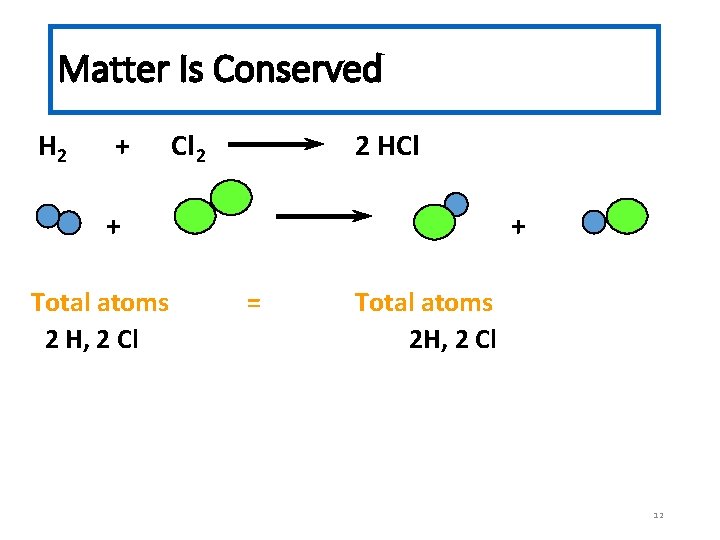
Matter Is Conserved H 2 + Cl 2 2 HCl + Total atoms 2 H, 2 Cl + = Total atoms 2 H, 2 Cl 12

Stop Notes • https: //www. youtube. com/watch? v=Fof. Pjj 7 v 414 13

Law of Conservation of Mass In any ordinary chemical reaction, matter is not created nor destroyed 14

Balance Equations with Coefficients in front of formulas balance each type of atom 4 NH 3 + 5 O 2 4 NO + 6 H 2 O 4 N = 4 N 12 H = 12 H 10 O = 10 O 15
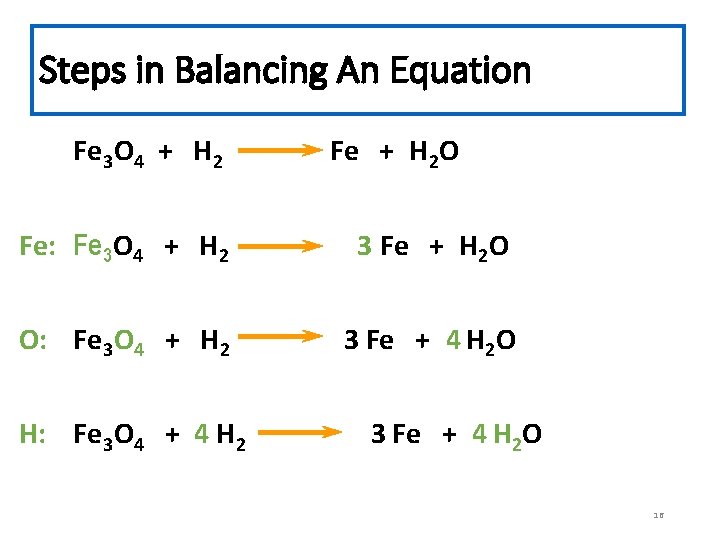
Steps in Balancing An Equation Fe 3 O 4 + H 2 Fe + H 2 O Fe: Fe 3 O 4 + H 2 3 Fe + H 2 O O: Fe 3 O 4 + H 2 3 Fe + 4 H 2 O H: Fe 3 O 4 + 4 H 2 3 Fe + 4 H 2 O 16

Check-In Fe 3 O 4 + 4 H 2 3 Fe + 4 H 2 O 1. Number of H atoms in 4 H 2 O A) 2 B) 4 C) 8 2. Number of O atoms in 4 H 2 O A) 2 B) 4 C) 8 3. Number of Fe atoms in Fe 3 O 4 A) 1 B) 3 C) 4 17

Solutions Fe 3 O 4 + 4 H 2 3 Fe + 4 H 2 O 1. Number of H atoms in 4 H 2 O C) 8 2. Number of O atoms in 4 H 2 O B) 4 3. Number of Fe atoms in Fe 3 O 4 B) 3 Lecture. PLUS Timberlake 18

Check-In Balance each equation. The coefficients for each equation are read from left to right A. Mg B. Al + + N 2 Mg 3 N 2 Cl 2 Al. Cl 3 19
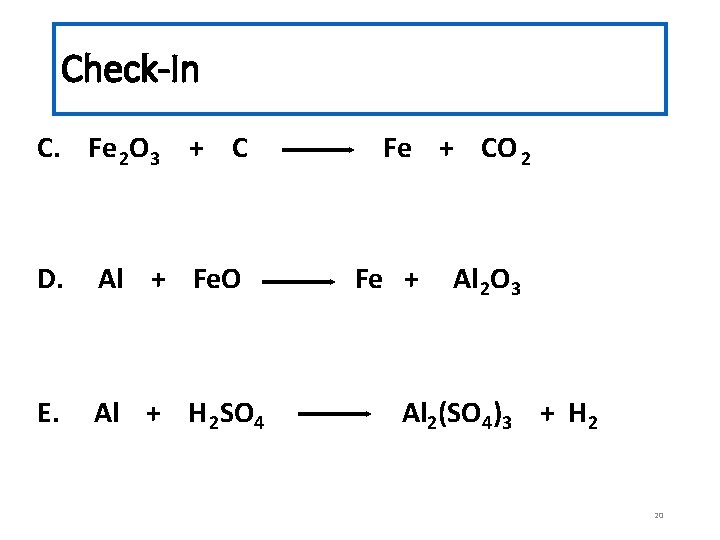
Check-In C. Fe 2 O 3 + C D. Al + Fe. O E. Al + H 2 SO 4 Fe + CO 2 Fe + Al 2 O 3 Al 2(SO 4)3 + H 2 20
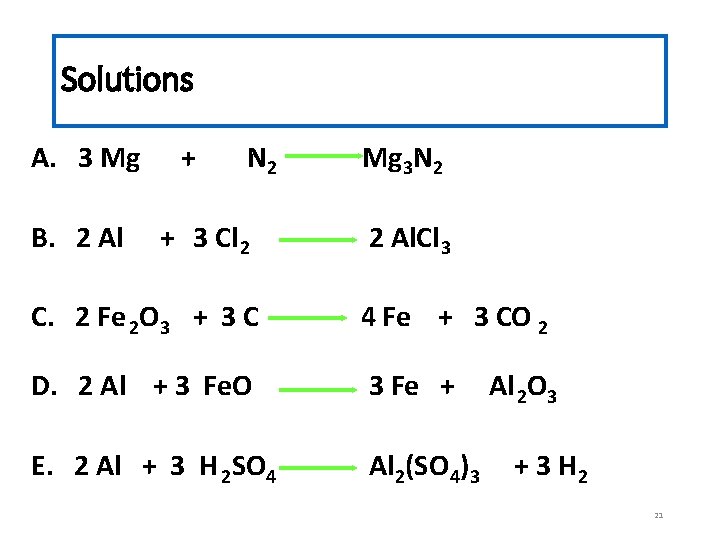
Solutions A. 3 Mg B. 2 Al + N 2 + 3 Cl 2 Mg 3 N 2 2 Al. Cl 3 C. 2 Fe 2 O 3 + 3 C 4 Fe + 3 CO 2 D. 2 Al + 3 Fe. O 3 Fe + E. 2 Al + 3 H 2 SO 4 Al 2(SO 4)3 Al 2 O 3 + 3 H 2 21
- Slides: 21