Chemical Reactions and Energy Chemical Reactions The book
Chemical Reactions and Energy

Chemical Reactions • The book has some really stupid ways of talking about chemical reactions that are complex like they change substances, or they can possibly produce gas or change color, or that they rearrange atoms • Basically a chemical reaction has something to do with moving electrons…PHYSICAL CHANGES DO NOT INVOLVE ELECTRONS AT ALL

Exothermic vs Endothermic Energy Curves to Explain Using Butane As Ex for both • Exothermic: Endothermic: • It takes energy to form bonds, breaking them releases energy • Remember reactants, products, EA

Classifying Reaction Types • Synthesis: Combine separate atoms to make a combined compound such as what is done to make polymers • Decomposition: Opposite of synthesis • Combusion: Same result from burning every time • Single and Double Displacement: When the atoms in the compounds seemingly swap or switch atoms they are connected/bonded too
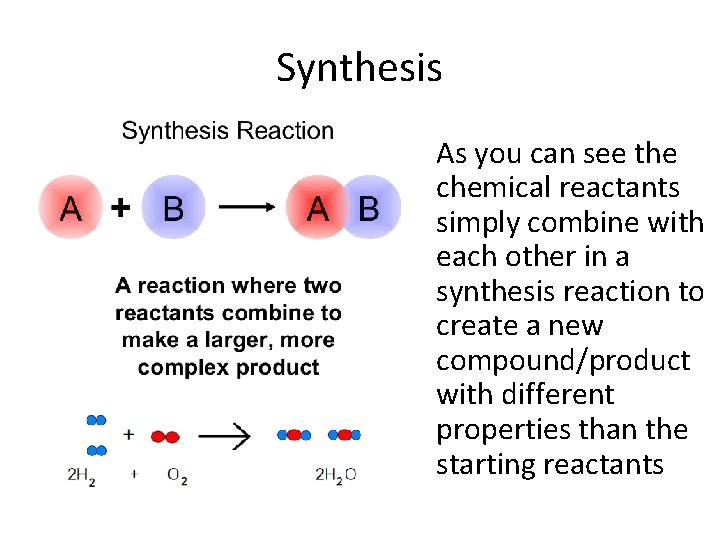
Synthesis • As you can see the chemical reactants simply combine with each other in a synthesis reaction to create a new compound/product with different properties than the starting reactants
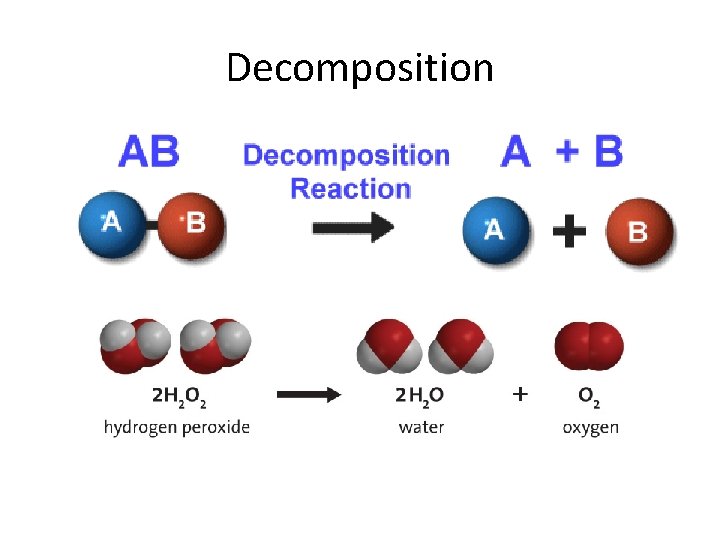
Decomposition
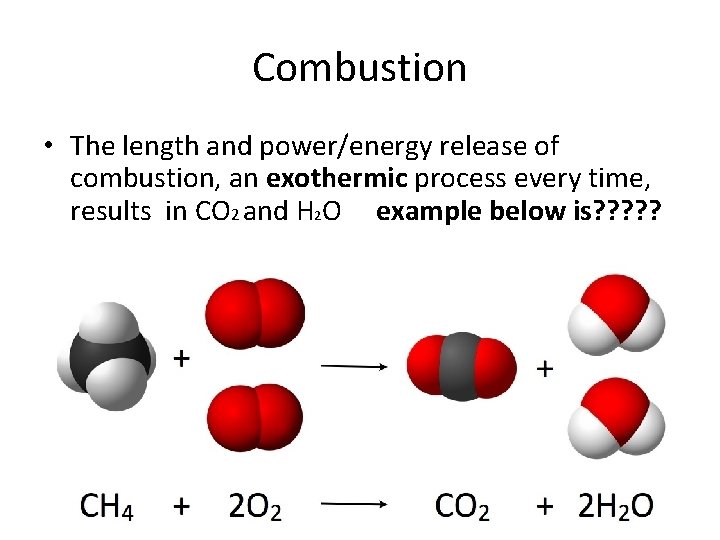
Combustion • The length and power/energy release of combustion, an exothermic process every time, results in CO 2 and H 2 O example below is? ? ?
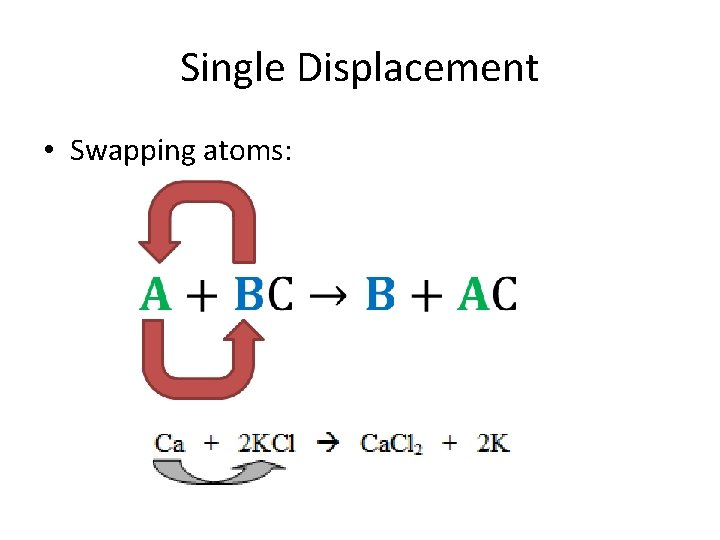
Single Displacement • Swapping atoms:
Single Displacement • Or what ever analogy you can find to help… :
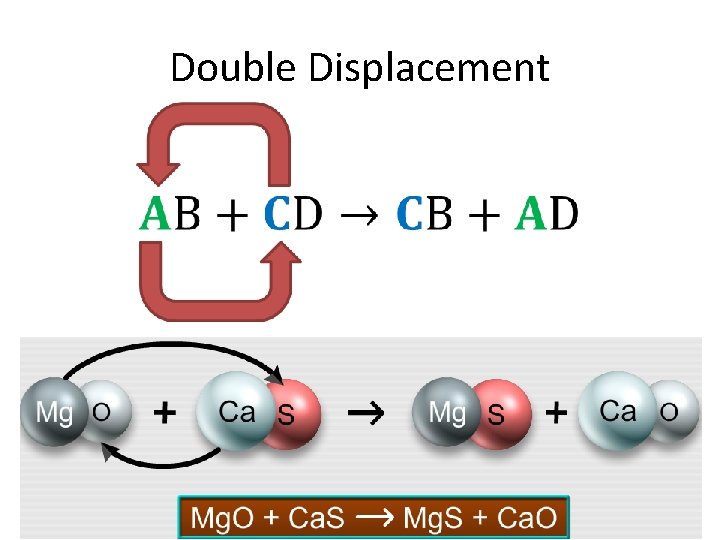
Double Displacement

Redox Reactions • Involve reactions with oxygen and are special types of reactions • Reduction either something that is gaining electrons or atoms. Electrons only matter if it is a metal reacting with oxygen. • Oxidation is the thing that is losing electrons or atoms. Electrons only matter if it is a metal reacting with oxygen.

Redox Reactions • When potassium reacts with oxygen, Potassium loses electrons so it gets oxidized while the oxygen gains electrons getting reduced: Same with iron as well and oxygen: • When burn something such as gas, the carbon and hydrogen go to carbon dioxide and water: Thus the carbon and hydrogen are oxidized while the oxygen is reduced. Think who gives and who takes electrons.

Summarizing Redox again • https: //www. youtube. com/watch? v=RX 6 rheefl. M Don’t look at the oxidation number rules

So think logically • Oxygen oxidizes stuff while it does that the oxygen gets equally effected, this means it gets reduced in most, many cases. • Oxygen does not have to be the only thing that gets reduced though, anything that takes the electrons or atoms and makes a large number of things small is getting reduced in size thus the word reduced ex of many to few with bonds:

Balancing Chemical Equations • THIS IS WHERE KNOWING MOLS MATTERS again I will let you know how to calculate any gram weights but you must be able to use the ratio to be able to convert at least once to find limiting reagents/reactants • Basic Balancing Examples: Next slide

Balancing Equations Basic

Using Molar Mass assuming its to the total of one mol to Calculate Grams in Equations and Limiting Reagents • Worksheet/ do first few together

After we balance our reactions… • Then our reactions occur at different rates and take some time, they do not starts and magically finish without time passing. • THIS IS KNOWN AS REACTION RATE • Factors which effect this period of time: Temperature, Surface Area Exposure, Atom Size/Mass, Concentration, Pressure, and concentration of catalysts/enzymes
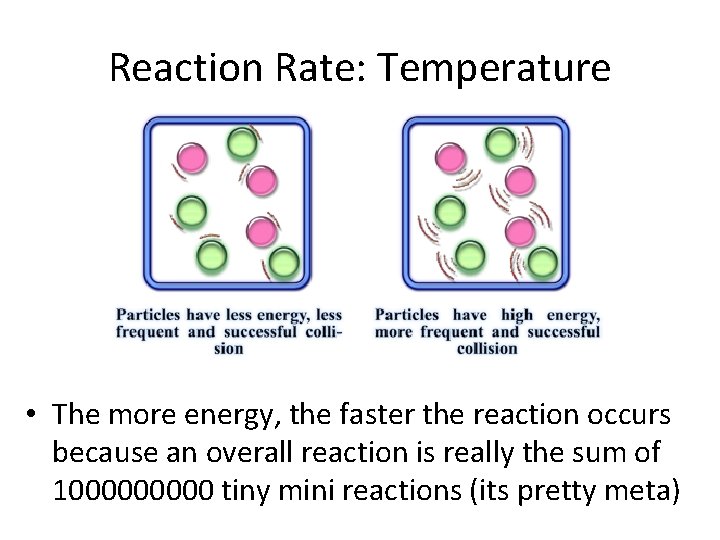
Reaction Rate: Temperature • The more energy, the faster the reaction occurs because an overall reaction is really the sum of 100000 tiny mini reactions (its pretty meta)
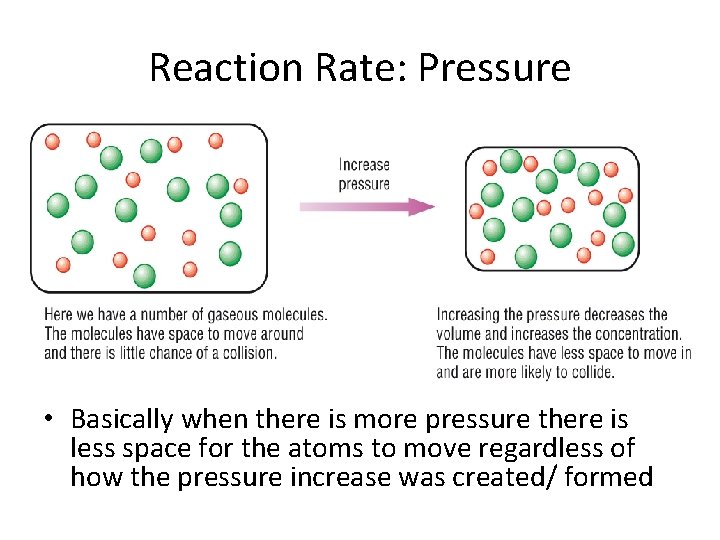
Reaction Rate: Pressure • Basically when there is more pressure there is less space for the atoms to move regardless of how the pressure increase was created/ formed

Reaction Rate: Mass remember KMT • Mass larger atom starts and stops_____ than one that is smaller • However GEOMETRY MATTERS in some cases atoms usually more MOLECULES that this pertains to but the overall structure allows exposure to possible collision with reactions

Reaction Rate: Surface Area • 2 solar panel example: you have two solar panels one is half as large as the other but only a third of the panel gets hit by decent light… More surface exposure means more possible bonds ie 0 vs about 40 in the bottom right example

Reaction Rate: Concentration • Again the more molecules there are, the less space for them to move without hitting each other • Flipside: They are more likely to interact with each other or bump into each other
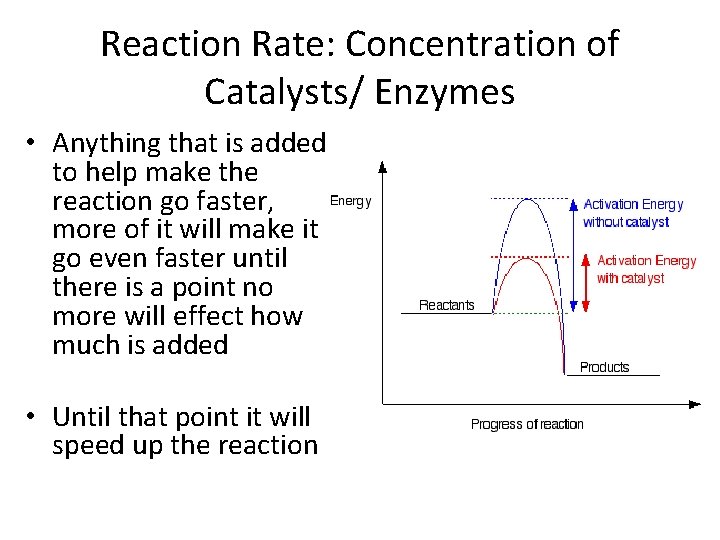
Reaction Rate: Concentration of Catalysts/ Enzymes • Anything that is added to help make the reaction go faster, more of it will make it go even faster until there is a point no more will effect how much is added • Until that point it will speed up the reaction

Why does this matter? • Back to the moonshiner, who is doing it legally that is, who is a master distiller and gets his money from a family business, a chemist who is mixing firework powders and needs them to set where naturally they set in 10 years, a pharmaceutical company making insulin but the way the chemicals naturally cleave without an enzyme additive but takes 30 days or longer than just minutes…. why does it matter? ? ?

Equilibrium Systems • So back when you were told how water is made (to be drawn below): • Probably had an arrow like -> but should really look like <-> or something that looks like two weird half pieces of the symbol

Equilibrium: What exactly is going on and what is it in a reaction? • Based off any of the things that could increase or decrease rate of reactions can be done not in just one way but in both ways to benefit both systems. Ie if increasing temperature increases rate in one direction decreasing the temperature favors the reaction in the other way: Forming of water from hydrogen and oxygen easier vs harder
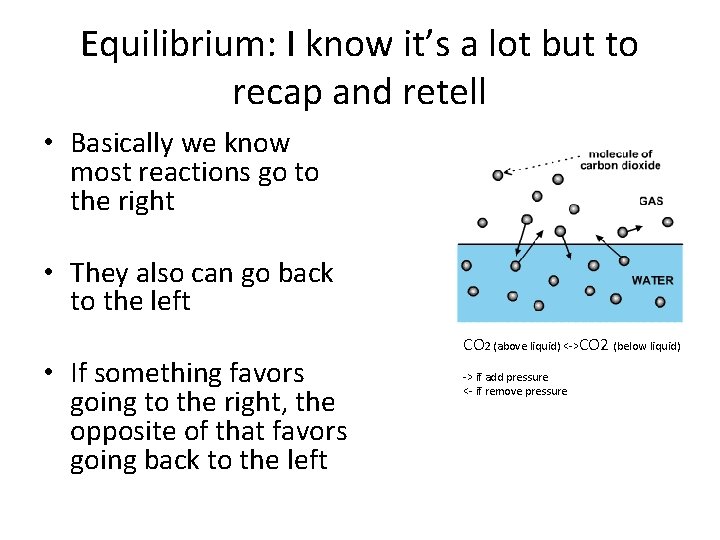
Equilibrium: I know it’s a lot but to recap and retell • Basically we know most reactions go to the right • They also can go back to the left • If something favors going to the right, the opposite of that favors going back to the left CO 2 (above liquid) <->CO 2 (below liquid) -> if add pressure <- if remove pressure

Equilibrium: When it actually is achieved • When all the forces driving a reaction <- equals all the forces going -> the equation is at chemical equilibrium • THIS DOES NOT MEAN NO REACTION IS OCCURRING IT MEANS FOR EVERY REACTION IN ONE DIRECTION, A DIFFERENT ONE IN THE OPPOSITE DIRECTION IS PRACTICALLY OCCURRING AT THE SAME TIME • Reactions try to self equalize over time and respond to minimal/small changes in a big way. Obviously more of one factor (pressure, concentration, exposure, etc. ) would increase the rate at which this occurs though

Equilibrium: Wrap Up • So basically there are two opposite reactions combating each other to go the other way, the on the left going right is usually the winner but when any bit of the opposite reaction going from right to left is occurring, it acts like a brake slowing down the rate of the reaction going from left to right • In other words there is no just one way in which an equation favors, rather, the reactions that work the fastest are experiencing the least amount of resistance in reacting the other way (or experience less of a brake-like force) which may be wanted if need something to occur carefully and also slowly aswell
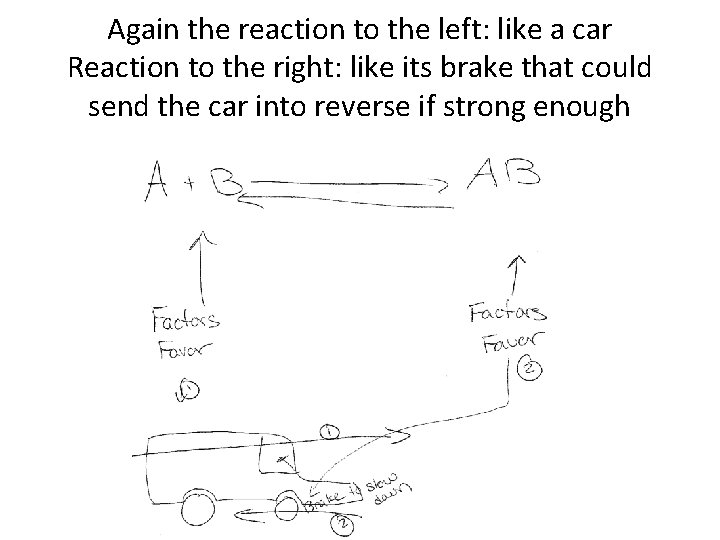
Again the reaction to the left: like a car Reaction to the right: like its brake that could send the car into reverse if strong enough
- Slides: 32