CHEMICAL REACTION ENGINEERING REACTOR DESIGN PROJECT Caitlin Boyd

CHEMICAL REACTION ENGINEERING: REACTOR DESIGN PROJECT Caitlin Boyd Katherine Ross April 23, 2008
OVERVIEW Elements of Reactor Design Reaction of 1 -butene to maleic anhydride Preliminary Plug Flow Reactor Design Inclusion of Energy Balance Optimization Process Optimized Reactor Conclusions

ELEMENTS OF REACTOR DESIGN Momentum Balance & Pressure Drop Reaction Mechanism Kinetics Conversion Production and Selectivity Energy Balance Thermodynamic Stability Optimization Assumptions

MOMENTUM BALANCE AND PRESSURE DROP The momentum balance accounts for the pressure change in the reactor. where Pressure drop cannot exceed 10% of initial pressure.

REACTION MECHANISM 1 -butene to maleic anhydride (1) C 4 H 8 (2) C 4 H 8 (3) C 4 H 8 (4) C 4 H 8 + 3 O 2 + 6 O 2 + O 2 C 4 H 2 O 3 + 3 H 2 O 4 CO 2 + 4 H 2 O 2 C 2 H 4 O C 4 H 6 O + H 2 O

KINETICS Preliminary Reaction Kinetics (1 Reaction) rm = k 1 * p. B where p. B is partial pressure of 1 -butene and k 1 = 3. 8075 x 105 *exp(-11569/T) [=] kmol/ kgcat-bar-s

KINETICS Kinetics for Multiple Reactions from Literature 1

CONVERSION OF REACTANT The goal conversion of 1 -butene found in literature was 90%. 1 This was used as a basis for all reactor models throughout the design process. X stands for conversion, FBT 0 is the initial flow of 1 butene and FBT is the outlet flow of 1 -butene.

PRODUCTION AND SELECTIVITY Goal Production: 40, 000 metric tons/ year Selectivity of maleic anhydride, the desired product, was found by the following equation: Selectivity
![ENERGY BALANCE where [=] k. J/ kgcat-s o This accounts for non- isothermal behavior ENERGY BALANCE where [=] k. J/ kgcat-s o This accounts for non- isothermal behavior](http://slidetodoc.com/presentation_image_h/8a84bfc4043675f4df68947718cf9a53/image-10.jpg)
ENERGY BALANCE where [=] k. J/ kgcat-s o This accounts for non- isothermal behavior in the reactor and allows for the optimization of the reactor temperature.

THERMODYNAMIC STABILITY The reactor gain was analyzed to determine whether the reactor was thermodynamically stable. The gain analysis involves raising the coolant fluid temperature one degree and finding the how much the hotspot temperature changes. A gain less than two indicates a thermodynamically stable reactor.

OPTIMIZATION Throughout the reactor design project this semester each memo submission involved a new aspect of the reactor: o o Volume Pressure Drop Multiple Reactions Energy Balance The final challenge was to optimize a reactor in both Polymath and Aspen that would include these aspects.

INITIAL REACTOR ASSUMPTIONS 90% conversion of 1 - butene Phosphorous and vanadium oxide catalyst 2 Inlet pressure of 2. 2 bar Reactor at 400 o. C Catalyst bulk density of 1000 kgcat/ m 3 Void fraction: 0. 45
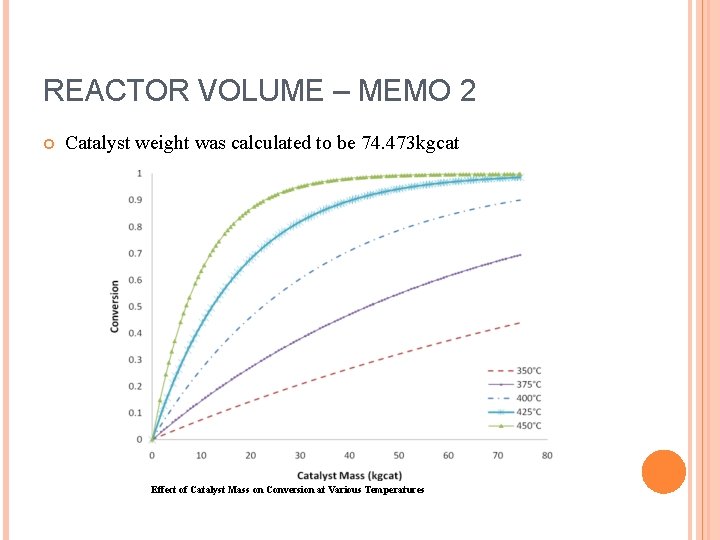
REACTOR VOLUME – MEMO 2 Catalyst weight was calculated to be 74. 473 kgcat Effect of Catalyst Mass on Conversion at Various Temperatures
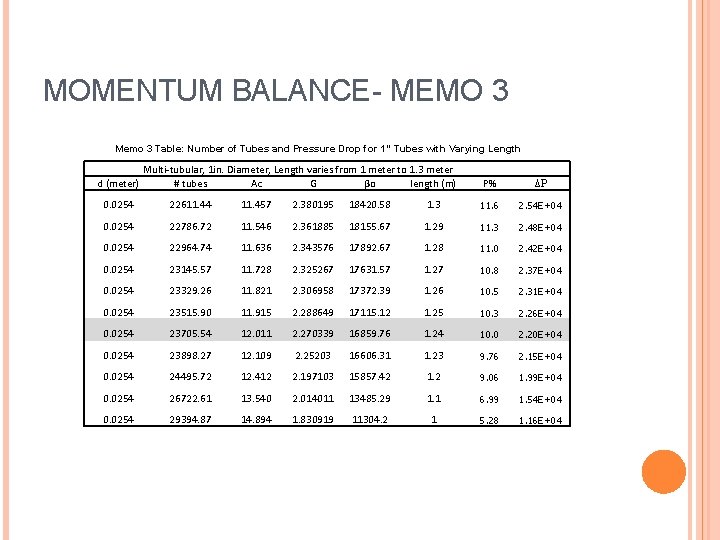
MOMENTUM BALANCE- MEMO 3 Memo 3 Table: Number of Tubes and Pressure Drop for 1” Tubes with Varying Length d (meter) Multi-tubular, 1 in. Diameter, Length varies from 1 meter to 1. 3 meter # tubes Ac G βo length (m) P% ΔP 0. 0254 22611. 44 11. 457 2. 380195 18420. 58 1. 3 11. 6 2. 54 E+04 0. 0254 22786. 72 11. 546 2. 361885 18155. 67 1. 29 11. 3 2. 48 E+04 0. 0254 22964. 74 11. 636 2. 343576 17892. 67 1. 28 11. 0 2. 42 E+04 0. 0254 23145. 57 11. 728 2. 325267 17631. 57 1. 27 10. 8 2. 37 E+04 0. 0254 23329. 26 11. 821 2. 306958 17372. 39 1. 26 10. 5 2. 31 E+04 0. 0254 23515. 90 11. 915 2. 288649 17115. 12 1. 25 10. 3 2. 26 E+04 0. 0254 23705. 54 12. 011 2. 270339 16859. 76 1. 24 10. 0 2. 20 E+04 0. 0254 23898. 27 12. 109 2. 25203 16606. 31 1. 23 9. 76 2. 15 E+04 0. 0254 24495. 72 12. 412 2. 197103 15857. 42 1. 2 9. 06 1. 99 E+04 0. 0254 26722. 61 13. 540 2. 014011 13485. 29 1. 1 6. 99 1. 54 E+04 0. 0254 29394. 87 14. 894 1. 830919 11304. 2 1 5. 28 1. 16 E+04
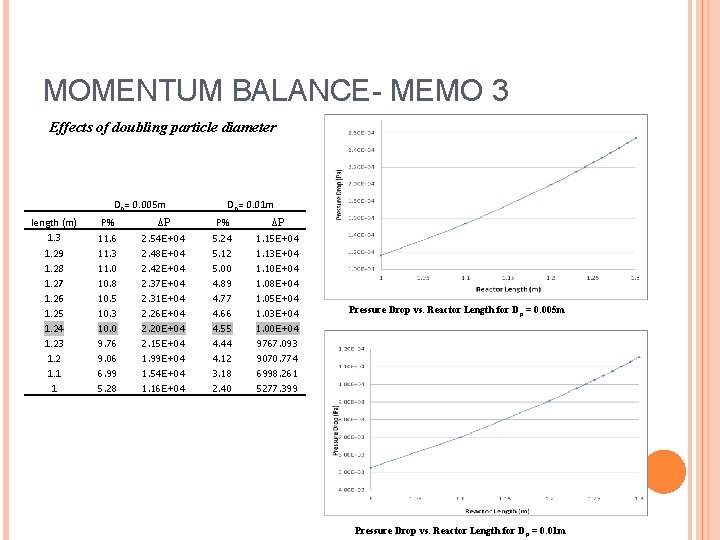
MOMENTUM BALANCE- MEMO 3 Effects of doubling particle diameter Dp= 0. 005 m length (m) 1. 3 1. 29 1. 28 1. 27 1. 26 1. 25 1. 24 1. 23 1. 2 1. 1 1 Dp= 0. 01 m P% ΔP 11. 6 2. 54 E+04 5. 24 1. 15 E+04 11. 3 11. 0 10. 8 10. 5 10. 3 10. 0 9. 76 9. 06 6. 99 5. 28 2. 48 E+04 2. 42 E+04 2. 37 E+04 2. 31 E+04 2. 26 E+04 2. 20 E+04 2. 15 E+04 1. 99 E+04 1. 54 E+04 1. 16 E+04 5. 12 5. 00 4. 89 4. 77 4. 66 4. 55 4. 44 4. 12 3. 18 2. 40 1. 13 E+04 1. 10 E+04 1. 08 E+04 1. 05 E+04 1. 03 E+04 1. 00 E+04 9767. 093 9070. 774 6998. 261 5277. 399 Pressure Drop vs. Reactor Length for D p = 0. 005 m Pressure Drop vs. Reactor Length for D p = 0. 01 m
MULTIPLE REACTIONS- MEMO 4 Assumptions Isothermal reactor at 623 K Target conversion: 90% Particle diameter: 0. 005 m Bulk density: 1, 000 kgcat/m 3 Inlet pressure: 2. 2 bar Void Fraction 0. 4 http: //www. bartek. ca/images/chemical. jpg Reactions (1) C 4 H 8 (2) C 4 H 8 (3) C 4 H 8 (4) C 4 H 8 + 3 O 2 + 6 O 2 + O 2 C 4 H 2 O 3 + 3 H 2 O 4 CO 2 + 4 H 2 O 2 C 2 H 4 O C 4 H 6 O + H 2 O
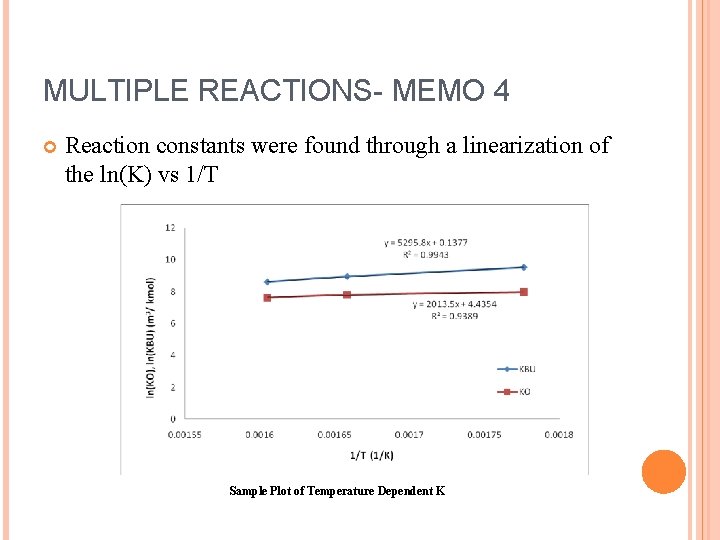
MULTIPLE REACTIONS- MEMO 4 Reaction constants were found through a linearization of the ln(K) vs 1/T Sample Plot of Temperature Dependent K
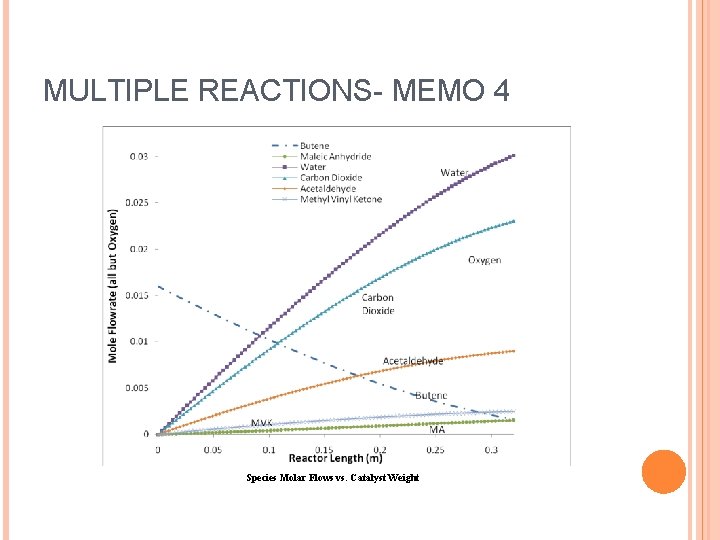
MULTIPLE REACTIONS- MEMO 4 Species Molar Flows vs. Catalyst Weight
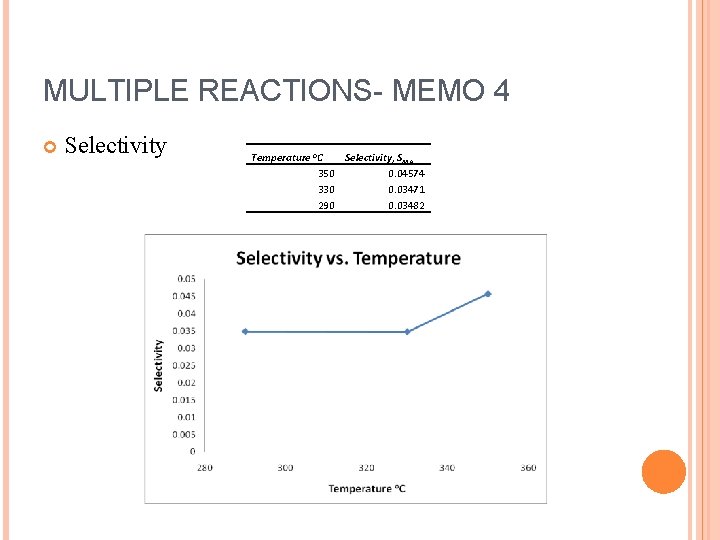
MULTIPLE REACTIONS- MEMO 4 Selectivity Temperature o. C Selectivity, SMA 350 0. 04574 330 0. 03471 290 0. 03482

ENERGY BALANCE- MEMO 5 New assumptions • • Inlet temperature: 563 K Target conversion: 90%3 Inlet Pressure = 220, 000 Pa 15 Bulk density = 1000 kgcat/ m 3 rxtr 15 Dp = 5 x 10 -3 m Φ = 0. 45 U = 0. 227 k. J/ m 2 -s-K Coolant temperature: 558 K E. B. used to locate and control reactor hotspot
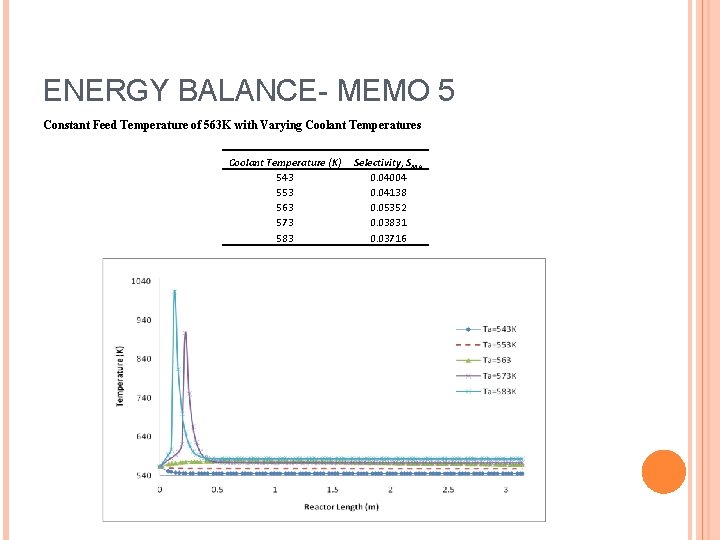
ENERGY BALANCE- MEMO 5 Constant Feed Temperature of 563 K with Varying Coolant Temperatures Coolant Temperature (K) 543 553 563 573 583 Selectivity, SMA 0. 04004 0. 04138 0. 05352 0. 03831 0. 03716
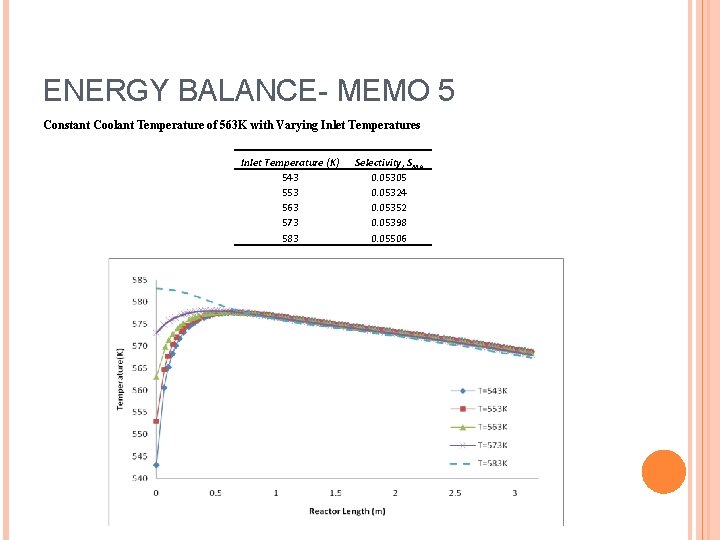
ENERGY BALANCE- MEMO 5 Constant Coolant Temperature of 563 K with Varying Inlet Temperatures Inlet Temperature (K) 543 553 563 573 583 Selectivity, SMA 0. 05305 0. 05324 0. 05352 0. 05398 0. 05506
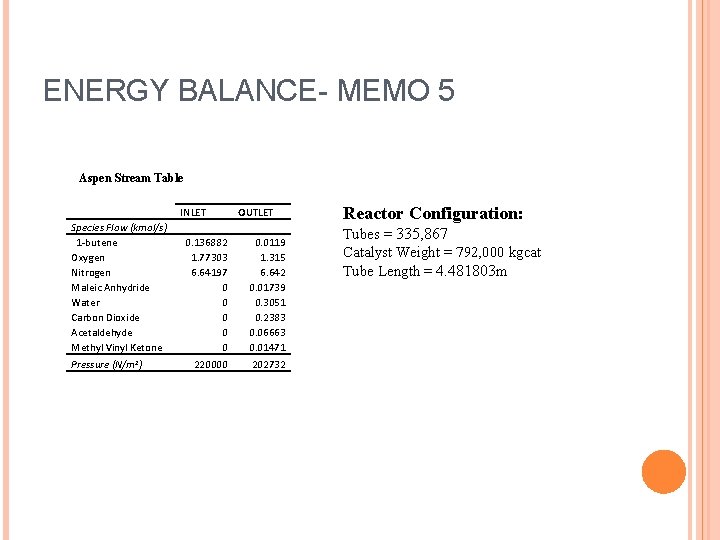
ENERGY BALANCE- MEMO 5 Aspen Stream Table Species Flow (kmol/s) 1 -butene Oxygen Nitrogen Maleic Anhydride Water Carbon Dioxide Acetaldehyde Methyl Vinyl Ketone Pressure (N/m 2) INLET OUTLET 0. 136882 0. 0119 1. 77303 1. 315 6. 64197 6. 642 0 0. 01739 0 0. 3051 0 0. 2383 0 0. 06663 0 0. 01471 220000 202732 Reactor Configuration: Tubes = 335, 867 Catalyst Weight = 792, 000 kgcat Tube Length = 4. 481803 m
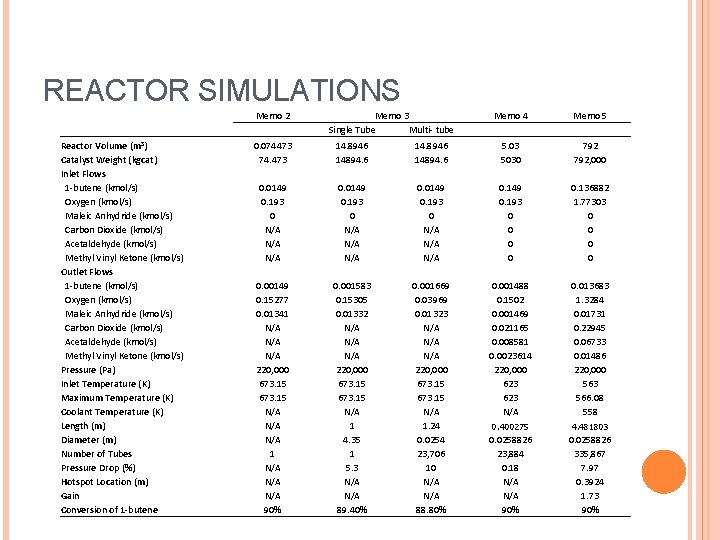
REACTOR SIMULATIONS Reactor Volume (m 3) Catalyst Weight (kgcat) Inlet Flows 1 -butene (kmol/s) Oxygen (kmol/s) Maleic Anhydride (kmol/s) Carbon Dioxide (kmol/s) Acetaldehyde (kmol/s) Methyl Vinyl Ketone (kmol/s) Outlet Flows 1 -butene (kmol/s) Oxygen (kmol/s) Maleic Anhydride (kmol/s) Carbon Dioxide (kmol/s) Acetaldehyde (kmol/s) Methyl Vinyl Ketone (kmol/s) Pressure (Pa) Inlet Temperature (K) Maximum Temperature (K) Coolant Temperature (K) Length (m) Diameter (m) Number of Tubes Pressure Drop (%) Hotspot Location (m) Gain Conversion of 1 -butene Memo 2 Memo 3 Single Tube Multi- tube Memo 4 Memo 5 0. 074473 74. 473 14. 8946 14894. 6 5. 03 5030 792, 000 0. 0149 0. 193 0 N/A N/A N/A 0. 0149 0. 193 0 N/A N/A 0. 149 0. 193 0 0 0. 136882 1. 77303 0 0 0. 00149 0. 15277 0. 01341 N/A N/A 220, 000 673. 15 N/A N/A 1 N/A N/A 90% 0. 001583 0. 15305 0. 01332 N/A N/A 220, 000 673. 15 N/A 1 4. 35 1 5. 3 N/A 89. 40% 0. 001669 0. 03969 0. 01323 N/A N/A 220, 000 673. 15 N/A 1. 24 0. 0254 23, 706 10 N/A 88. 80% 0. 001488 0. 1502 0. 001469 0. 021165 0. 008581 0. 0023614 220, 000 623 N/A 0. 013683 1. 3284 0. 01731 0. 22945 0. 06733 0. 01486 220, 000 563 566. 08 558 0. 400275 4. 481803 0. 0258826 23, 884 0. 18 N/A 90% 0. 0258826 335, 867 7. 97 0. 3924 1. 73 90%

OPTIMIZED REACTOR An inlet temperature of 563 K, a coolant temperature of 558 K and an inlet pressure 2. 4 bar produce a gain under two. Coolant temperature (K) Inlet Temperature (K) Hotspot Temperature (K) Gain 559 558 563 568. 593818 566. 657912 1. 935906 557 563 564. 86977 1. 788142 Other conditions gave a thermodynamically unstable reactor. With these conditions the reactor volume and catalyst weight were changed to give a 90% conversion and optimal selectivity of maleic anhydride.
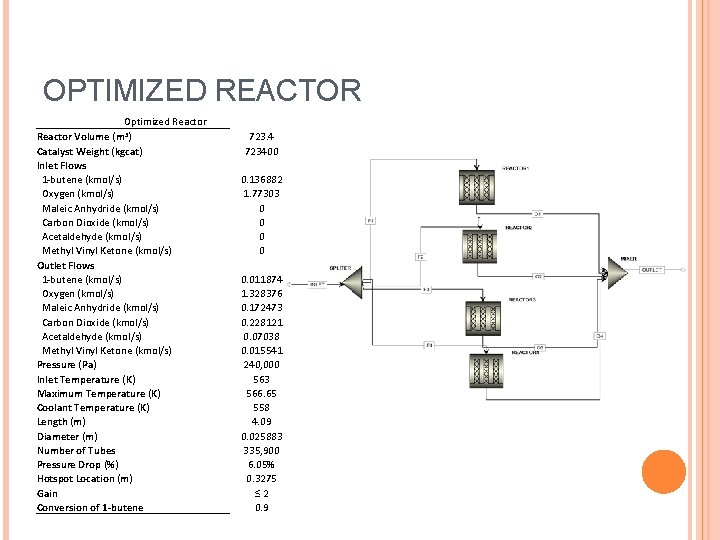
OPTIMIZED REACTOR Optimized Reactor Volume (m 3) Catalyst Weight (kgcat) Inlet Flows 1 -butene (kmol/s) Oxygen (kmol/s) Maleic Anhydride (kmol/s) Carbon Dioxide (kmol/s) Acetaldehyde (kmol/s) Methyl Vinyl Ketone (kmol/s) Outlet Flows 1 -butene (kmol/s) Oxygen (kmol/s) Maleic Anhydride (kmol/s) Carbon Dioxide (kmol/s) Acetaldehyde (kmol/s) Methyl Vinyl Ketone (kmol/s) Pressure (Pa) Inlet Temperature (K) Maximum Temperature (K) Coolant Temperature (K) Length (m) Diameter (m) Number of Tubes Pressure Drop (%) Hotspot Location (m) Gain Conversion of 1 -butene 723. 4 723400 0. 136882 1. 77303 0 0 0. 011874 1. 328376 0. 172473 0. 228121 0. 07038 0. 015541 240, 000 563 566. 65 558 4. 09 0. 025883 335, 900 6. 05% 0. 3275 ≤ 2 0. 9

CONCLUSIONS Overall the selectivity from the reaction scheme is not optimal for producing maleic anhydride When the reaction temperature is above 563 K the reaction becomes a runaway The reactor is too large to be cost effective After 1983 nothing was published because it was found that butane was a better feedstock

REFERENCES 1 Cavani, F. , Trifiro, F. ; Oxidation of 1 -Butene and Butadiene to Maleic Anhydride. Industrial Engineering Chemical Product Research and Development. 1983. Vol 22. No. 4, 570 -577 2 Varma, R. L. ; Saraf, D. N. ; Journal of Catalysis; [online] 1978, 55, 351 -272
- Slides: 29