Chemical Reaction Engineering CRE is the field that















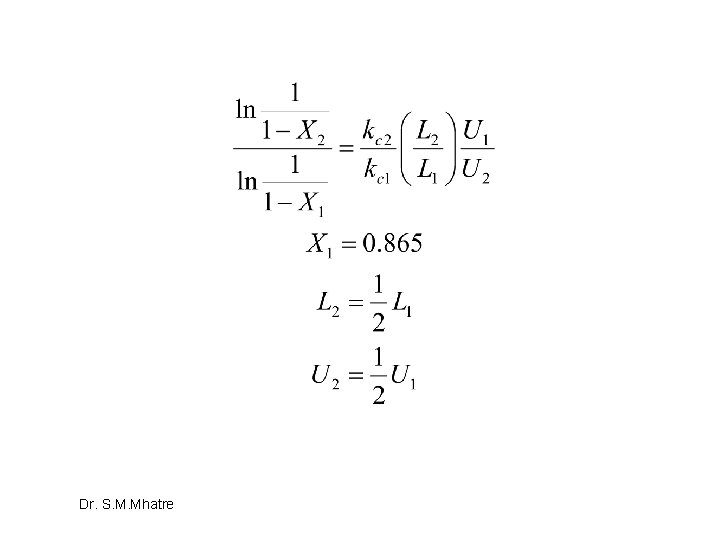
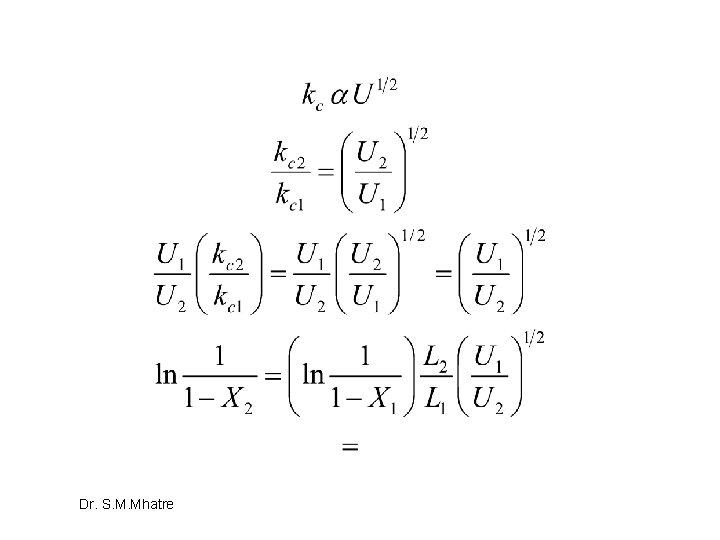




- Slides: 29

Chemical Reaction Engineering (CRE) is the field that studies the rates and mechanisms of chemical reactions and the design of the reactors in which they take place. External Diffusion Effect on Heterogeneous reactions: Mass transfer fundamentals Dr. S. M. Mhatre

Contents: 1. Seven Steps in a Catalytic Reaction 2. External Diffusion Across a Stagnant Film 3. Relative Rates of Diffusion and Reaction 4. Mass Transfer in a Packed Bed of Catalyst Particles 5. Shrinking Core Model Dr. S. M. Mhatre

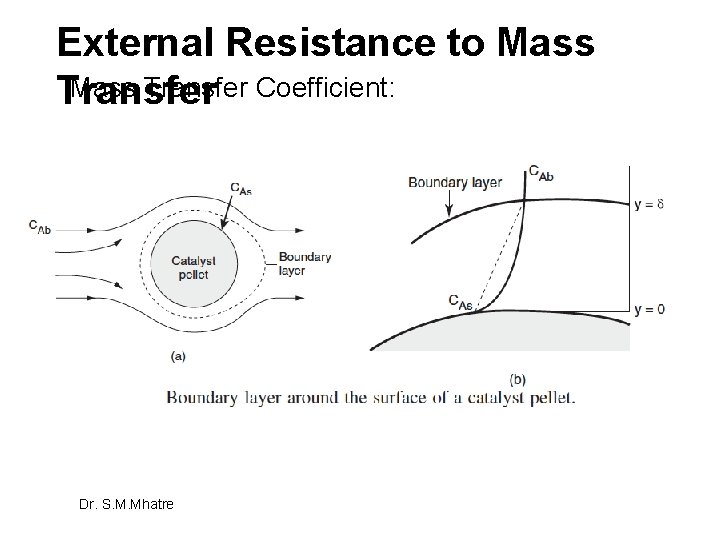
Steps in a Catalytic Reaction � 1. Mass transfer (diffusion) of the reactant(s) from the bulk fluid to the external surface of the catalyst pellet � 2. Diffusion of the reactant from the pore mouth through the catalyst pores to the immediate vicinity of the internal catalytic surface � 3. Adsorption of reactant A onto the catalytic surface � 4. Reaction on the surface of the catalyst � 5. Desorption of the products from the surface � 6. Diffusion of the products from the interior of the pellet to the pore mouth at the external surface � 7. Mass transfer of the products from the external pellet surface to the bulk fluid We shall now focus on steps 1, 2, 6, and 7. Because the reaction below does not occur in the bulk phase (only at the surface, at z = delta). Dr. S. M. Mhatre

Binary Diffusion �Diffusion is the spontaneous intermingling or mixing of atoms or molecules by random thermal motion. Mass transfer is any process in which diffusion plays a role. �The molar flux is just the molar flow rate, FA, divided by the cross sectional area, AC, normal to the flow. WA = FA/AC �Molar flux of A , WA (moles/time/area) with respect to fixed coordinate system WA = J A + B A JA = diffusional flux of A with respect to bulk motion, i. e. molar average velocity BA = flux of A resulting from bulk flow Dr. S. M. Mhatre

Derive WA =JA +y. A(WA +WB) WA = JA + BA BA =CAU Binary System, Multiply and divide by Dr. S. M. Mhatre

Molar Flux at different conditions: 1. For equal molar counter diffusion 2. Diffusion through a stagnant film, WB = 0 3. For dilute concentration Dr. S. M. Mhatre

Dr. S. M. Mhatre

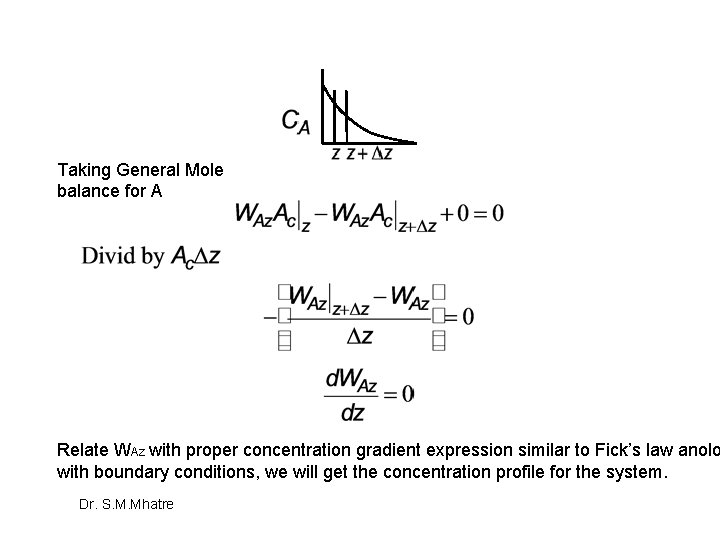
Modeling Diffusion Without Reaction In developing mathematical models for chemically reacting systems in which diffusion effects are important, the first steps are: Step 1: Perform a differential mole balance on a particular species A. Step 2: Substitute for FAZ in terms of WAZ. Step 3: Replace W, by the appropriate expression for the concentration gradient Step 4: State the boundary conditions. Step 5: Solve for the concentration profile. Step 6: Solve for the molar flux Dr. S. M. Mhatre

Dr. S. M. Mhatre

External Resistance to Mass Transfer Coefficient: Transfer Dr. S. M. Mhatre

Taking General Mole balance for A Relate WAZ with proper concentration gradient expression similar to Fick’s law anolo with boundary conditions, we will get the concentration profile for the system. Dr. S. M. Mhatre

Dr. S. M. Mhatre

Dr. S. M. Mhatre

Dr. S. M. Mhatre

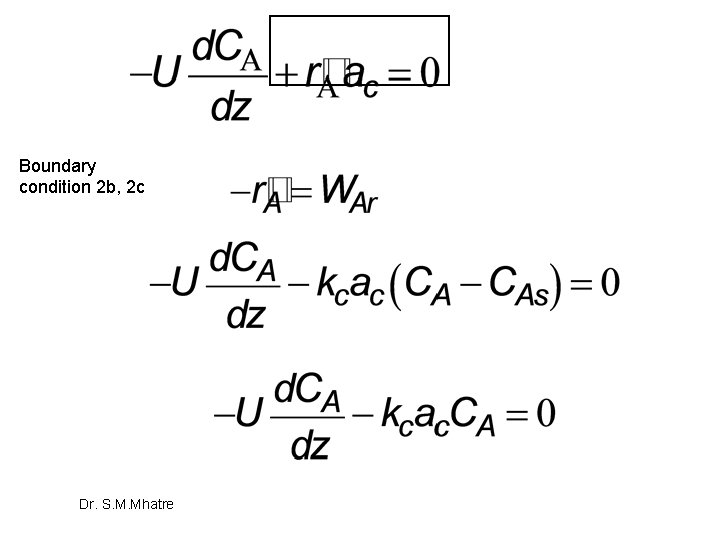
Rapid reaction on Catalyst surface The rate of reaction at surface of catalyst is given by, As per boundary conditions 2 b and 2 c We need to eliminate CAs. as it is not easily measurable compare to bulk concentration, so Dr. S. M. Mhatre

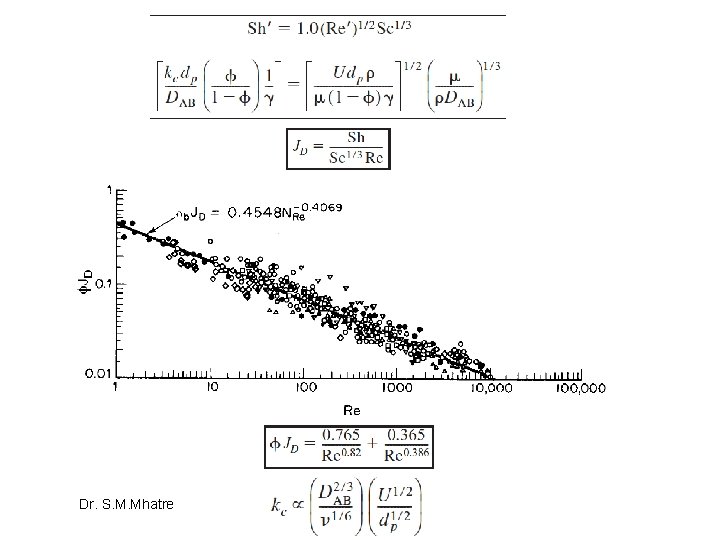
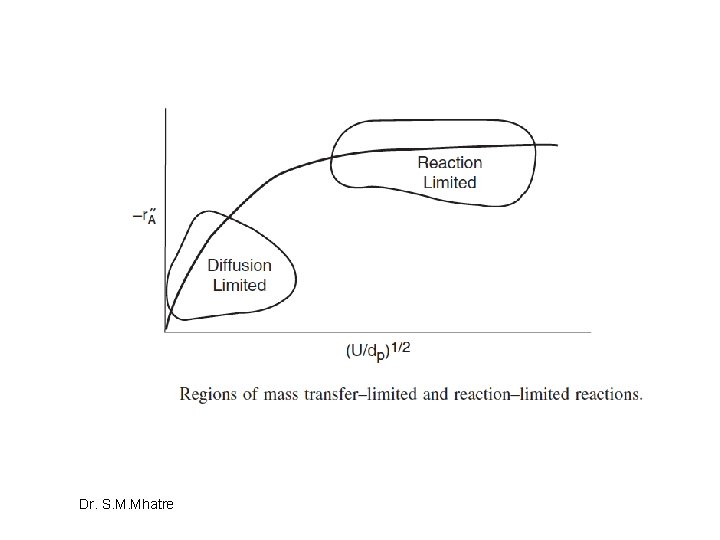
One will often find the flux to or from the surface as written in terms of an effective transport coefficient keff: where Case 1: rapid reaction, diffusion limits Case 2: Slow reaction, reaction limits Dr. S. M. Mhatre

Dr. S. M. Mhatre

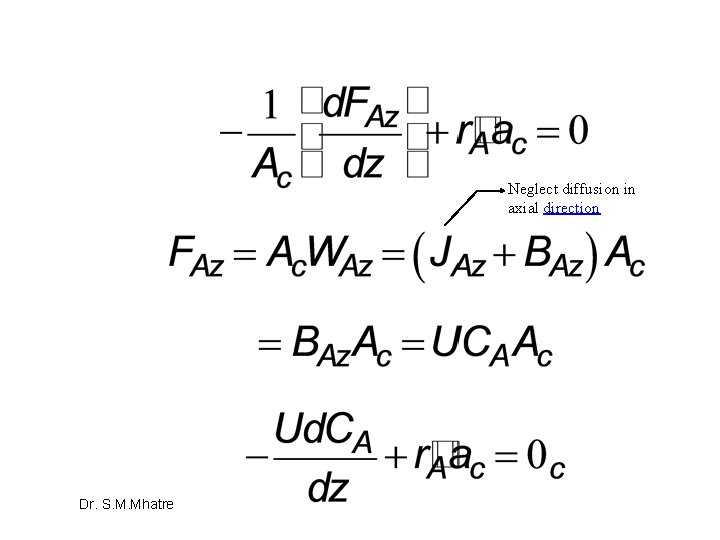
Mass transfer limited reaction in packed bed Dr. S. M. Mhatre

Neglect diffusion in axial direction Dr. S. M. Mhatre

Boundary condition 2 b, 2 c Dr. S. M. Mhatre

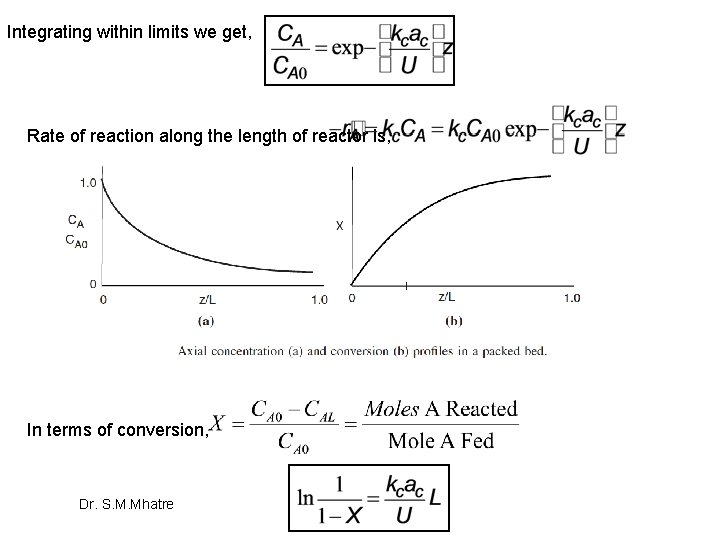
Integrating within limits we get, Rate of reaction along the length of reactor is, In terms of conversion, Dr. S. M. Mhatre

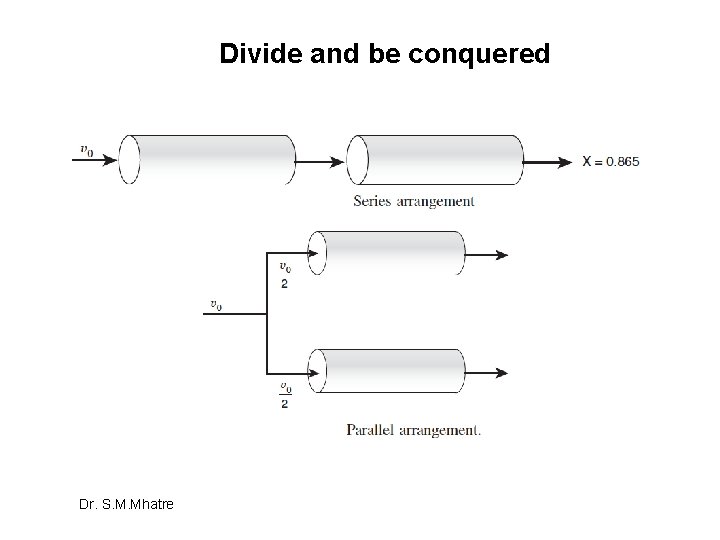
Divide and be conquered Dr. S. M. Mhatre

Suffix 1 –undivided system, suffix 2 divided system Dr. S. M. Mhatre
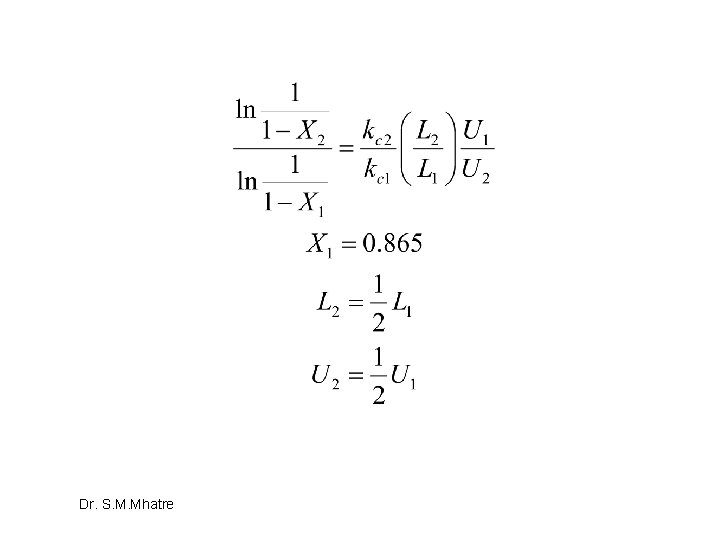
Dr. S. M. Mhatre
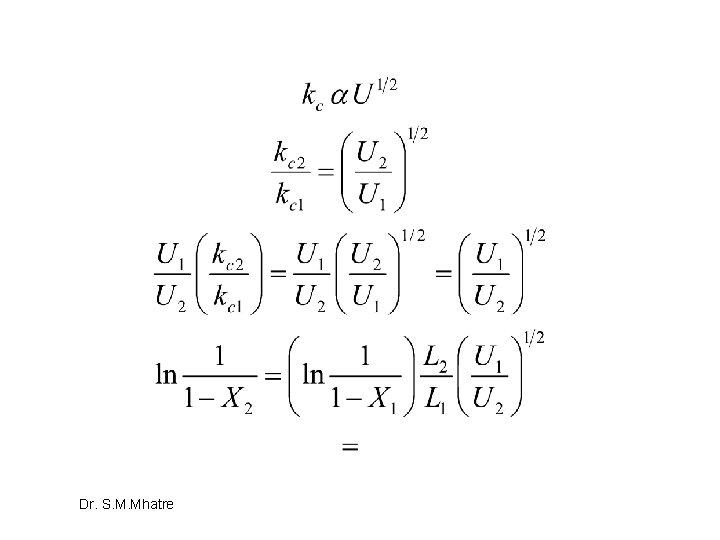
Dr. S. M. Mhatre

Hence, Division is Bad idea in terms of conversion Dr. S. M. Mhatre

The same reaction as that in previous Example is being carried out In the same two reactors in series. A new engineer suggests that the rate of reaction could be increased by a factor of 210 by increasing the reaction temperature from 400 0 C to 500°C, reasoning that the reaction rare doubles for every 10°C increase lo temperature. Another engineer arrives on the scene and beratcs the new engineer with quotations from Chapter 3 concerning the rule of thumb. She points out that it is valid only for a specific activation energy within a specific temperature range. She then suggests that he go ahead with the proposed temperature increase but that the rate of reaction could be increased by a factor of 23. What do you Dr. S. M. Mhatre think? Who is correct

Shrinking Core model The shrinking core model is used to describe situations in which solid particles are being consumed either by dissolution or reaction and, as a result, the amount of the material being consumed is "shrinking. " Dr. S. M. Mhatre

Case of catalyst regeneration In progress…………. . Dr. S. M. Mhatre