Chemical Re actions Enzymes Chemical Reactions and Enzymes










- Slides: 14

Chemical Re actions & Enzymes

Chemical Reactions and Enzymes As we have seen living things are made of chemical compounds (Biomolecules), but more importantly chemistry isn’t just what life is made of, chemistry is also what life does.

Chemical Reactions 0 Chemical reaction – is a process in which one or more chemicals change into another chemical. 0 The chemicals (elements or compounds) that enter into a chemical reaction are the reactants. 0 The chemicals (elements or compounds) produced by a chemical reaction are known as products.

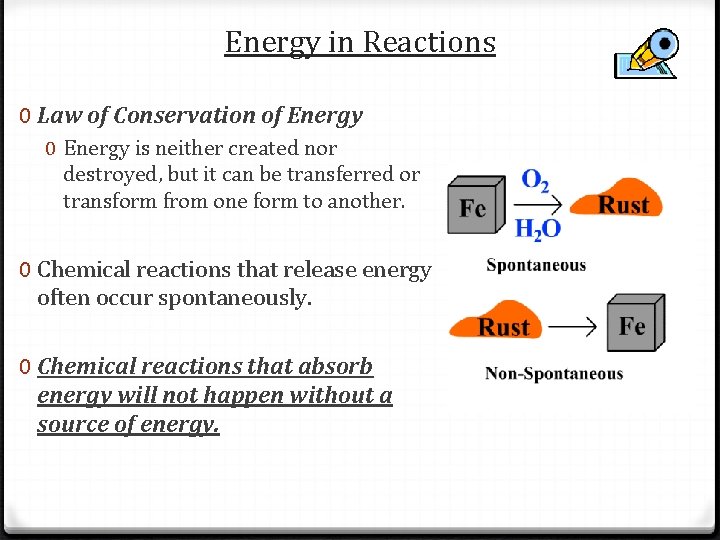
Energy in Reactions 0 Law of Conservation of Energy 0 Energy is neither created nor destroyed, but it can be transferred or transform from one form to another. 0 Chemical reactions that release energy often occur spontaneously. 0 Chemical reactions that absorb energy will not happen without a source of energy.

Activation Energy 0 The minimum energy that must be input to a chemical system in order for a chemical reaction to occur. 0 Non-Spontaneous Reaction 0 It absorbs energy 0 Its Activation Energy is high 0 Always requires energy to start 0 Spontaneous Reaction 0 It releases energy 0 Its Activation Energy is low 0 It happens quickly

Enzymes 0 Some chemical reactions are too slow or have activation energies that are too high. 0 Catalyst works by lowering a reaction’s activation energy, but with out being changed by the chemical reaction. 0 These reactions require catalysts or substances that speed up the rate of a chemical reaction.

Enzymes 0 Enzymes – are proteins that act as biological catalysts. 0 Enzymes speed up chemical reactions that take place in cells. 0 Enzymes are very specific, generally catalyzing only one type of chemical reactions. 0 Lock and Key concept 0 One Enzyme can catalyze or speed up multiple reactions 0 Enzymes are recyclable

Enzymes 0 For a chemical reaction to occur, the reactants must collide with enough energy so that existing bonds will be broken and new bonds will be formed. 0 Enzymes provide a place where reactants can be brought together to react. 0 The reactants of an enzyme – catalyzed reactions are known as substrates.

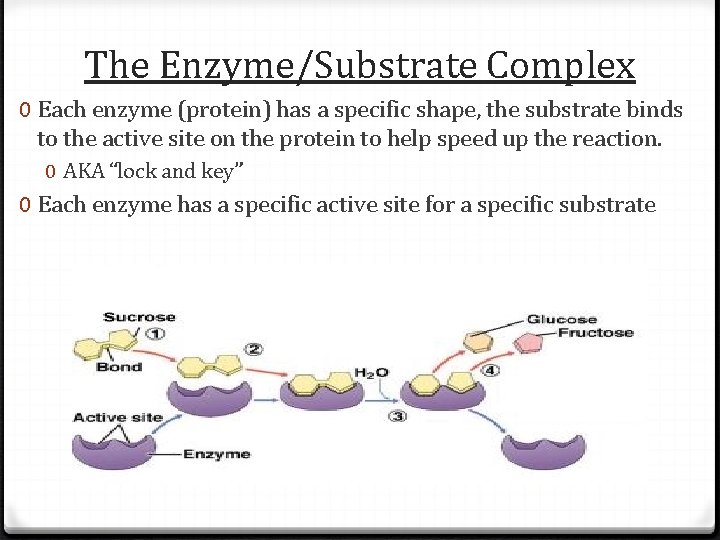
The Enzyme/Substrate Complex 0 Each enzyme (protein) has a specific shape, the substrate binds to the active site on the protein to help speed up the reaction. 0 AKA “lock and key” 0 Each enzyme has a specific active site for a specific substrate

Disrupting Enzyme Activity 0 The activity of enzymes can be affected by different factors. 0 These Factors can be 0 Denature 0 Reversible inhibition 0 Irreversible inhibition 0 Inhibitor 0 a thing that blocks someone or something.

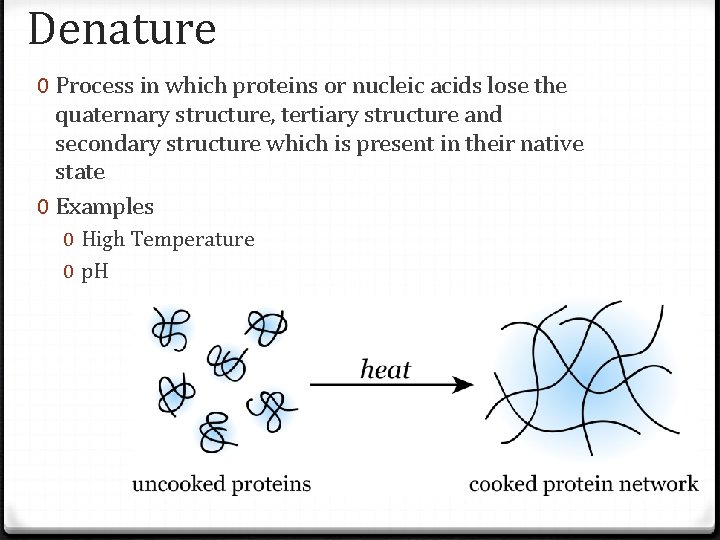
Denature 0 Process in which proteins or nucleic acids lose the quaternary structure, tertiary structure and secondary structure which is present in their native state 0 Examples 0 High Temperature 0 p. H

Reversible Inhibition 0 Reversible inhibitors can bind to enzymes through weak noncovalent interactions. 0 Inhibitors can be easily removed 0 Enzyme and inhibitor complex can be dissociated (separated)

Types of Reversible Inhibition 0 Competitive Inhibition 0 Inhibitor competes with substrate to bind to the enzyme. 0 Uncompetitive Inhibition 0 Inhibitor binds to the enzyme with its substrate 0 Non-Competitive Inhibition 0 Inhibitor binds to enzyme at any place with or without a substrate

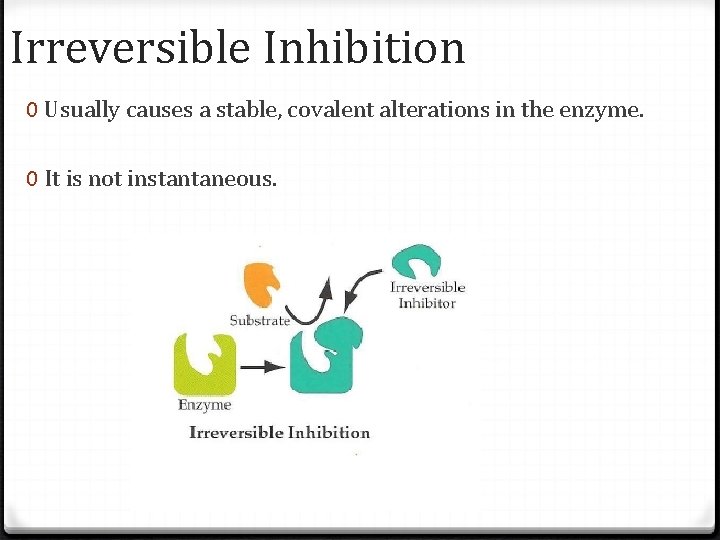
Irreversible Inhibition 0 Usually causes a stable, covalent alterations in the enzyme. 0 It is not instantaneous.