CHEMICAL PHYSICAL PROPERTIES CHANGES 101019 OBJECTIVE Define and

CHEMICAL & PHYSICAL PROPERTIES & CHANGES 10/10/19 OBJECTIVE: Define and distinguish chemical and physical properties and changes TASK: Journal Entry: Contrast how you separate elements from compounds (like water electrolysis) with how you separate parts or mixtures (like mixture lab). Elements from Compounds Mixtures

DISCUSSION QUESTIONS • How are the videos/demo similar? • How are the videos/demo different?

CARD SORT • Sort the cards into categories based on whatever criteria makes sense to you. • I will not tell you how many categories there should be. • Sort your cards on the whiteboard and write your categories for each column. • Be prepared to share with the class.

CARD SORT • Sort your cards into categories on your whiteboard:

1. PHYSICAL PROPERTIES A. Definition –a characteristic of a pure substance that can be observed without changing it into another substance B. Ex: density, color, texture, conductivity, malleability (will it bend, ) boiling point, melting point

2. CHEMICAL PROPERTIES A. Definition- a characteristic of a pure substance that describes its ability to change into a different substance B. Ex: flammability, corrosiveness, reactivity

3. PHYSICAL CHANGES A. Definition – a change in a substance after which the substance remains the same B. Used to separate mixtures C. Ex: pounding, tearing, cutting, dissolving, evaporating, melting, boiling, pulling

4. CHEMICAL CHANGES A. Definition – a change in a substance which results in new substance with new properties B. Also known as a chemical reaction C. Ex: burning, digesting, fermenting, decomposing

5. CHEMICAL CHANGE EVIDENCE A. Precipitate forms immediately (solid) B. Gas is created (bubbles) – Not just released C. Spontaneous color change D. Energy change • Exothermic = releases energy • Endothermic = absorbs energy E. Odor produced (not released)

CARD SORT – ROUND 2 • Sort your cards into the following categories: Physical Properties Physical Changes Chemical Properties Chemical Changes
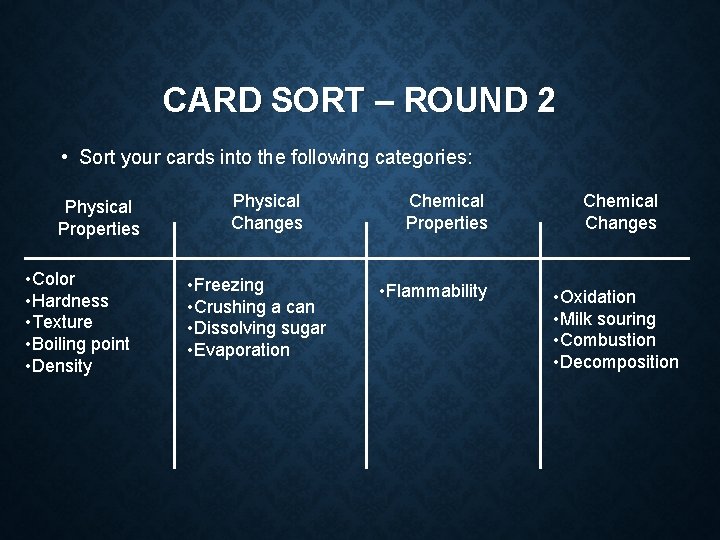
CARD SORT – ROUND 2 • Sort your cards into the following categories: Physical Properties • Color • Hardness • Texture • Boiling point • Density Physical Changes • Freezing • Crushing a can • Dissolving sugar • Evaporation Chemical Properties • Flammability Chemical Changes • Oxidation • Milk souring • Combustion • Decomposition

REVIEW • If a new substance is formed, what type of change has occurred? • Give an example of a physical property. • Chemical property. • Physical change. • Chemical change.
- Slides: 12