Chemical Nomenclature Ionic Bonds vs Covalent Bonds Ionic

Chemical Nomenclature

Ionic Bonds vs Covalent Bonds � Ionic Bonds › Metal + Non-Metal › Metal (CATION) always first followed by Non-metal (ANION) › Electrons are Transferred � Covalent Bonds (Sometimes called Molecular) › Non-metal + Non-metal › Electrons are Shared � We will talk more about how these bonds form, their properties, etc next semester
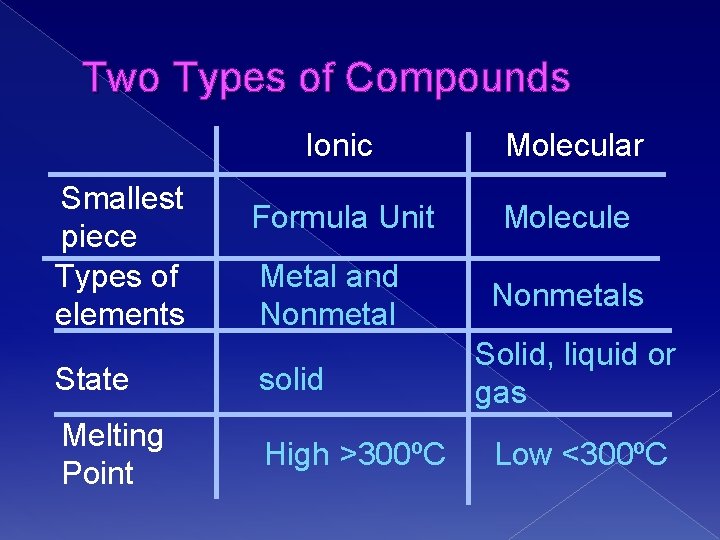
Two Types of Compounds Smallest piece Types of elements Ionic Molecular Formula Unit Molecule Metal and Nonmetal State solid Melting Point High >300ºC Nonmetals Solid, liquid or gas Low <300ºC

IONIC NOMENCLATURE Ionic Nomenclature Binary Compounds Ternary Compounds Ionic Compounds with Transition Metals

Binary Ionic Compounds � Binary = 2 elements total in the cmpd � Naming Binary Compounds › Take the cation (metal) and name it just like the element on the periodic table › Take the anion (non-metal) and change the ending of the element to –ide › Ex: Al 2 O 3 = Aluminum Oxide

Most commonly missed –ide changes � Oxygen = Oxide � Sulfur = Sulfide � Hydrogen = Hydride � Phosphorus = Phosphide � Nitrogen = Nitride � Silicon = Silicide � Selenium = Selenide � Carbon = Carbide

Practice These: � Na. Cl � Mg 3 N 2 � K 2 S � Li. H � Sr. Br 2 � Al. P

Writing Formulas for Binary Ionic Compounds Step 1 Step 2 Step 3 � Al+3 O 2 - • Cross charges and make subscripts from them (remove + or - ) Write elements together as compound � Al 2 O 3 • If necessary, simplify subscripts to lowest terms � • Write elements with their charges (M first, then NM) Step 3 not necessary
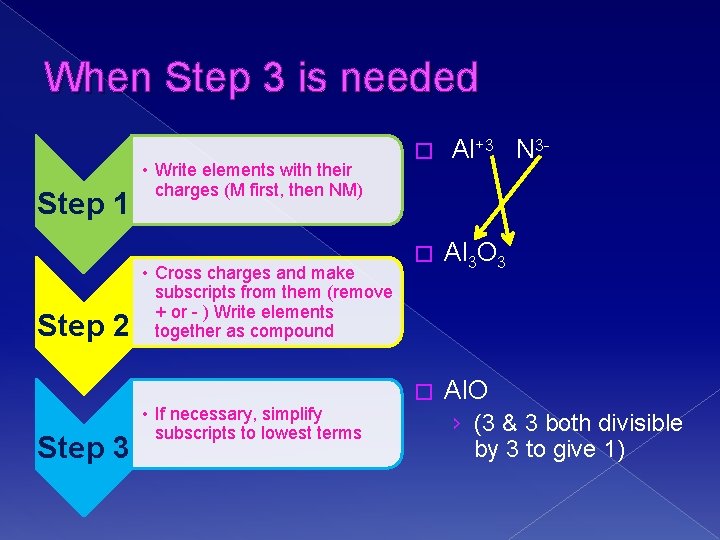
When Step 3 is needed Step 1 Step 2 Step 3 • Write elements with their charges (M first, then NM) • Cross charges and make subscripts from them (remove + or - ) Write elements together as compound • If necessary, simplify subscripts to lowest terms � Al+3 N 3 - � Al 3 O 3 � Al. O › (3 & 3 both divisible by 3 to give 1)
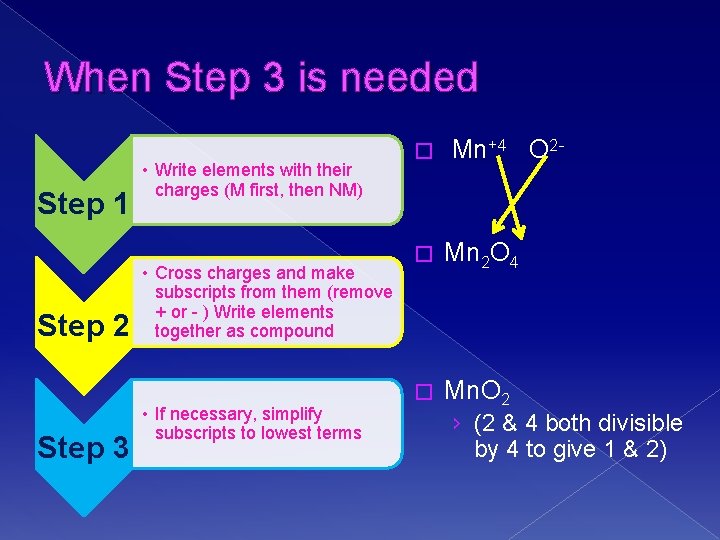
When Step 3 is needed Step 1 Step 2 Step 3 • Write elements with their charges (M first, then NM) • Cross charges and make subscripts from them (remove + or - ) Write elements together as compound • If necessary, simplify subscripts to lowest terms � Mn+4 O 2 - � Mn 2 O 4 � Mn. O 2 › (2 & 4 both divisible by 4 to give 1 & 2)

Ternary Ionic Compounds � Ternary = 3 or more elements in compound � Naming Ternary Ionic Compounds › Name like Binary (element name + --ide)EXCEPT when POLYATOMIC IONS are used as metals or non-metals › You will need to memorize the most common polyatomic Ions
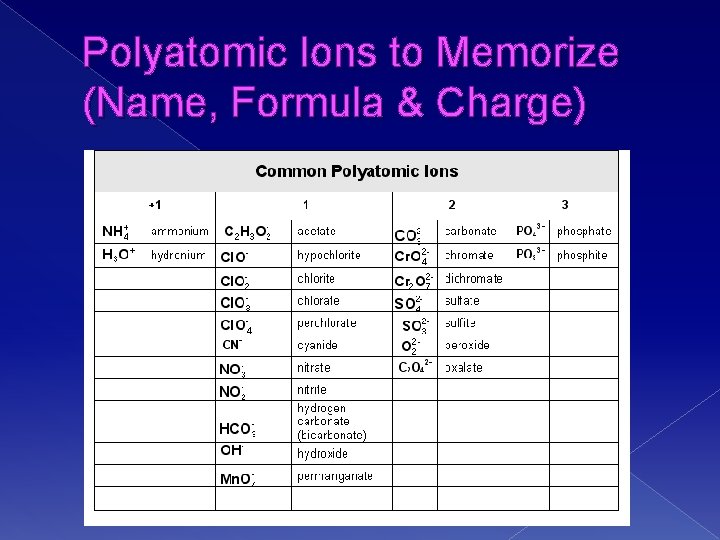
Polyatomic Ions to Memorize (Name, Formula & Charge)

Practice (it may help to put parentheses around polyatomic when you ID them) � NH 4 Cl � Mg(OH)2 � Na. NO 3 � Al. PO 4 � K 2 SO 4 � H 2 O 2

Formulas for Ternary Compounds (involve polyatomic ions) 1. Still… cation FIRST, anion SECOND Na+ OH- 2. Criss-cross the superscript charge to get the subscripts. Na 1 OH 1 3. Reduce the subscript to the least whole number ratio if needed. Na. OH

Ternary Compounds (Cont. ) 4. If polyatomic ion has a subscript in the formula greater than 1, you must put parentheses around the polyatomic ion and place the subscript outside the parenthesis. Al(OH)3

Practice Example: Barium nitrate 1. Write the symbols/formulas for the cation and anion, including CHARGES! 2. Cross Charges. 3. Simplify subscripts. Use parentheses if you need more than one of a polyatomic ion.

Practice Example: Ammonium sulfate 1. Write the formulas for the cation and anion, including CHARGES! 2. Cross Charges. 3. Simpifly subscripts. Use parentheses if you need more than one of a polyatomic ion.

Try these on your own… Magnesium carbonate � Strontium hydroxide � Aluminum phosphate �

Ionic Compounds with Transition Metals � Remember Transition Metals are called transition because they can have varying oxidation states!!!

Metals that form more than One Cation The name of metals with two or more positive ions (cations) use a Roman numeral to identify ionic charge. Lead Pb 2+ lead(II) Pb 4+ lead(IV) Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings 21

Naming Ionic Compounds with Transition Metals � Step 1: The easiest way to do this is to uncross the subscripts back to charges. › ALWAYS MOVE THE ANION’S CHARGE BACK FIRST! CHECK & MAKE SURE THE ANION CHARGE IS WHAT IT SHOULD BE! › Ex. Fe 3 N 2 uncrossed: Fe 2+ N 3� What we expect on N › Ex. Mn. O 2 uncrossed: Mn+2 O-1 � NOT WHAT WE EXPECT ON O! WE KNOW O should be a -2 which meant the subscripts simplified. So…. � REAL ANSWER: Mn. O 2 uncrossed: Mn+4 O 2 -

Naming Ionic Compounds with Transition Metals Step 2 -4 � Once you know the charges, now name the transition metal with Roman Numerals: I II IV V VI � � Ex: Fe+3 O 2› Iron (III) Oxide � Ex: Mn+4 O 2› Manganese (IV) Oxide

Writing Formulas for Ionic Compounds with Transition Metals � Just like Binary & Ternary Rules › Now the metal charge is given to you, you don’t need to remember it or look it up! › Don’t forget ONLY transition metals use roman numerals › Don’t forget about your polyatomics

Practice!!! Mn 2 O 7 Nb. Cl 5 Ti. P Pd(SO 4)2 Pt. F 2 Os 2 O 3 Ir(NO 3)4 Co(Cl. O)2 Fe 2 S 3 Au. I 3 Manganese (VII) Oxide Niobium (V) Chloride Titanium (III) Phosphide Palladium (IV) Sulfate Platinum (II) Fluoride Osmium (III) Oxide Iridium (IV) Nitrate Cobalt (II) Hypochloride Iron (III) Sulfide Gold (III) Iodide

Sometimes…. � You will see Transition metals named by their classical/traditional names. �I want you to be aware of the following, but you won’t be tested over them in this class
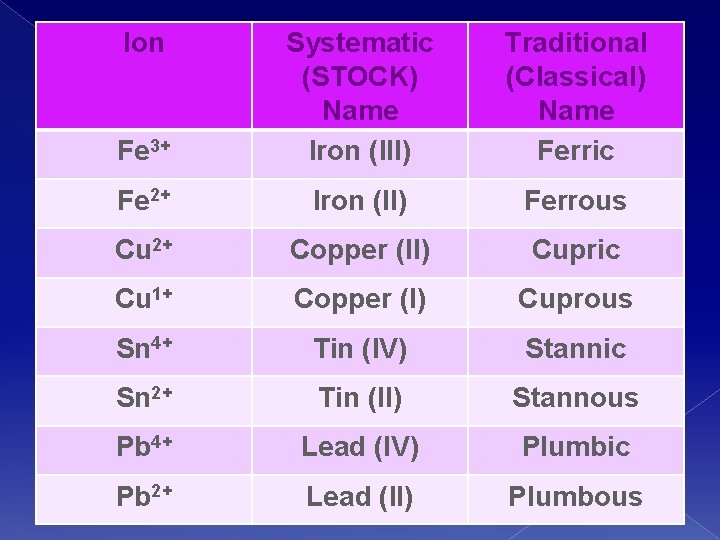
Ion Fe 3+ Systematic (STOCK) Name Iron (III) Traditional (Classical) Name Ferric Fe 2+ Iron (II) Ferrous Cu 2+ Copper (II) Cupric Cu 1+ Copper (I) Cuprous Sn 4+ Tin (IV) Stannic Sn 2+ Tin (II) Stannous Pb 4+ Lead (IV) Plumbic Pb 2+ Lead (II) Plumbous

Traditional System Name the metal first; use Latin names of metals. � In binary compounds the nonmetal takes the suffix –ide. (Like before) � The ion with the lower charge takes the suffix –ous � The ion with the higher charge takes the suffix – ic. �
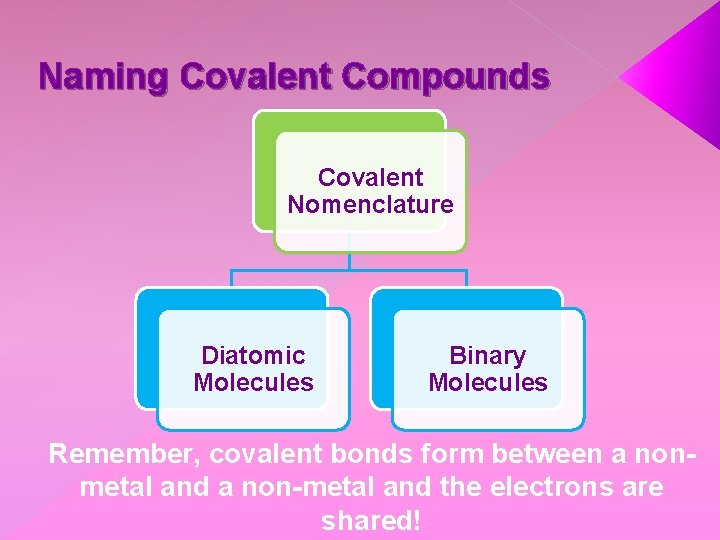
Naming Covalent Compounds Covalent Nomenclature Diatomic Molecules Binary Molecules Remember, covalent bonds form between a nonmetal and a non-metal and the electrons are shared!

Diatomic Molecules � When some elements are by themselves, with no other element, they pair up with other elements that are like them. � They are called diatomic molecules! � We name them just by the element name on the periodic table Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2

Practice Br 2 � H 2 � Oxygen � Chlorine � Bromine � Hydrogen � O 2 � Cl 2 � If these 7 elements are paired with other elements, they will NOT be named like this! They will be named like we talk about later!

Binary Covalent Compounds � � Covalent Compounds use Prefixes to name compounds ALWAYS use prefixes except: › Only use mono on the second element › If you have a diatomic molecule � 1 st: (prefixelementname) 2 nd: (prefixelementendinide) PREFIXES 1 -mono 2 -di 3 -tri 4 -tetra 5 -penta 6 -hexa 7 -hepta 8 -octa 9 -nona 10 -deca

Examples P 2 O 5 diphosphorous pentoxide OF 2 oxygen difluoride PBr 3 phosphorous tribromide

Practice! 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. CO 2 CO P 2 O 5 N 2 O Si. O 2 CBr 4 SO 2 PBr 5 ICl 3 NI 3 N 2 O 3 B 2 H 6 NBr 3 SCl 6 P 4 O 10 S 7 O 2 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. Carbon dioxide Carbon monoxide Diphosphorous pentaoxide Dinitrogen monoxide Silicon dioxide Carbon tetrabromide Sulfur dioxide Phosphorous pentabromide Iodine trichloride Nitrogen triiodide Dinitrogen trioxide Diboron hexahydride Nitrogen tribromide Sulfur hexachloride Tetraphosphorous decaoxide Heptasulfur dioxide

To Get Covalent Formulas, just go backwards….

In reverse to get Formulas! 1. Silicon tetrafluoride 2. Dinitrogen trisulfide 3. Sulfur trioxide 4. Dinitrogen tetroxide 5. Nitrogen monoxide 6. Nitrogen dioxide 7. Tetraphosphorous octaoxide 8. Tetraphosphorous nonasulfide 9. Diarsenic pentoxide 10. Phosphorous trichloride 11. Carbon tetrachloride 12. Dihydrogen monoxide 13. Selenium hexafluoride 14. Iodine heptafluoride 15. Tetraarsenic decoxide 16. Carbon disulfide 17. Oxygen difluoride 1. Si. F 4 2. N 2 S 3 3. SO 3 4. N 2 O 4 5. NO 6. NO 2 7. P 4 O 8 8. P 4 S 9 9. As 2 O 5 10. PCl 3 11. CCl 4 12. H 2 O 13. Se. Fl 6 14. IF 7 15. As 4 O 10 16. CS 2 17. OF 2

Naming Acids � Acid formulas usually begin with H � We name them according to how the ANION name ends! � Common Acids we use: › HCl: Hydrochloric Acid › HNO 3: Nitric Acid › H 2 SO 4: Sulfuric Acid
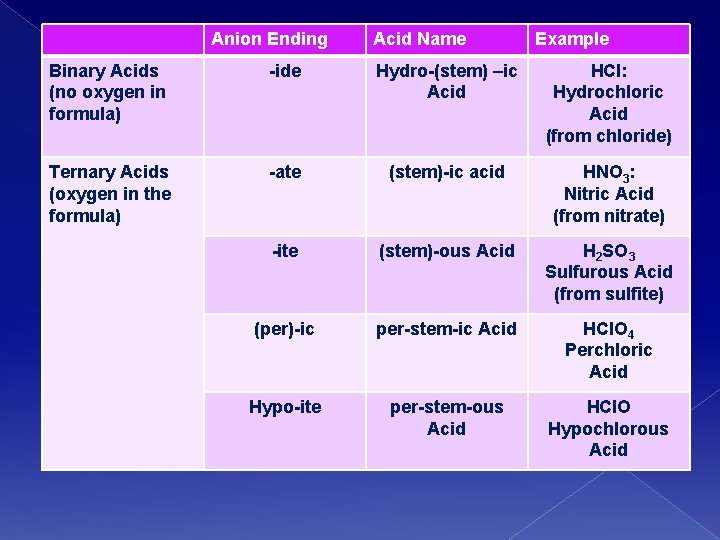
Anion Ending Acid Name Example Binary Acids (no oxygen in formula) -ide Hydro-(stem) –ic Acid HCl: Hydrochloric Acid (from chloride) Ternary Acids (oxygen in the formula) -ate (stem)-ic acid HNO 3: Nitric Acid (from nitrate) -ite (stem)-ous Acid H 2 SO 3 Sulfurous Acid (from sulfite) (per)-ic per-stem-ic Acid HCl. O 4 Perchloric Acid Hypo-ite per-stem-ous Acid HCl. O Hypochlorous Acid
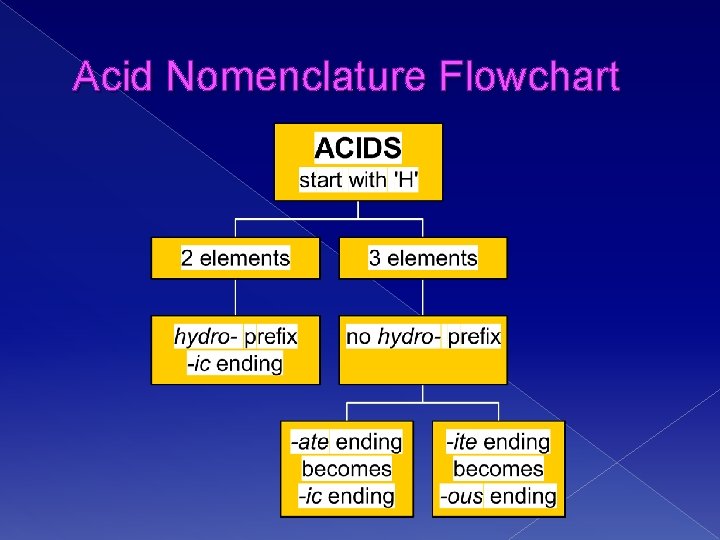
Acid Nomenclature Flowchart

Acid Nomenclature • HBr (aq) • No oxygen, -ide hydrobromic acid carbonic acid sulfurous acid • H 2 CO 3 • Has oxygen, -ate • H 2 SO 3 • Has oxygen, -ite

Acid Nomenclature • hydrofluoric acid • 2 elements H+ F- HF (aq) • sulfuric acid • 3 elements, -ic H+ SO 42 - H 2 SO 4 • nitrous acid • 3 elements, -ous H+ NO 2 - HNO 2

Name ‘Em! � HI (aq) � HCl � H 2 SO 3 � HNO 3 � HIO 4

Write the Formula! � Hydrobromic acid � Nitrous acid � Carbonic acid � Phosphoric acid � Hydrotelluric acid

Hydrates �Some compounds contain H 2 O in their › These compounds are called hydrates. structure. �This is different from (aq) because the H 2 O is part of the molecule (not just surrounding it). �The H 2 O can usually be removed if heated. �A dot separates water: e. g. Cu. SO 4 • 5 H 2 O is copper(II) sulfate pentahydrate. �Name the ionic part (the salt) just the way you would as before; a greek prefix indicates the # of H 2 O groups.

Hydrates (examples) �Na 2 SO 4 • 10 H 2 O �Ni. SO 4 • 6 H 2 O �sodium sulfate decahydrate nickel(II) sulfate hexahydrate carbonate monohydrate Na 2 CO 3 • H 2 O Ba. Cl 2 • 2 H 2 O �barium chloride dihydrate
Nomenclature Summary Flowchart
- Slides: 46