Chemical Nomenclature chemistry Ionic Nomenclature For cations Keeps















- Slides: 18

Chemical Nomenclature chemistry

Ionic Nomenclature �For cations ◦ Keeps its elemental name ◦ Examples: Na+ – sodium ion Al 3+ – aluminum ion � For anions ◦ Change the ending of the elemental name to -ide ◦ Examples: Cl is chlorine Cl– is chloride ion S is sulfur S 2– is sulfide ion

Ionic Nomenclature � Metal + Nonmetal � Name Cation first, then anion � Ca. Cl 2 : Calcium chloride � Mg. O: Magnesium oxide

Name the following compound, Ba. I 2. A. B. C. D. Barium iodide Baride iodine Barium iodine Baride iodide

Name this Compound, Ca. Br 2. A. B. C. D. Calcium bromine Carbon bromine Calcium bromide Carbon bromine

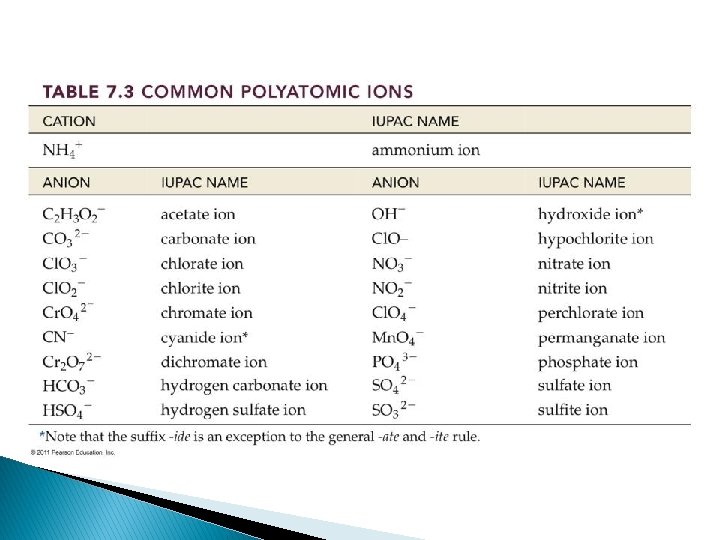
Polyatomic Ions � If Polyatomic is Cation, then list name & add anion name to the end ◦ Ex) (NH 4)Cl: Ammonium chloride � If polyatomic is anion, then name cation and list polyatomic name ◦ Ex) Mg (NO 3)2 Magnesium nitrate

� Look at handout for more info

Name this compound, Na. NO 3 A. B. C. Sodium nitrate Sodium nitrite Sodium nitrogen oxide

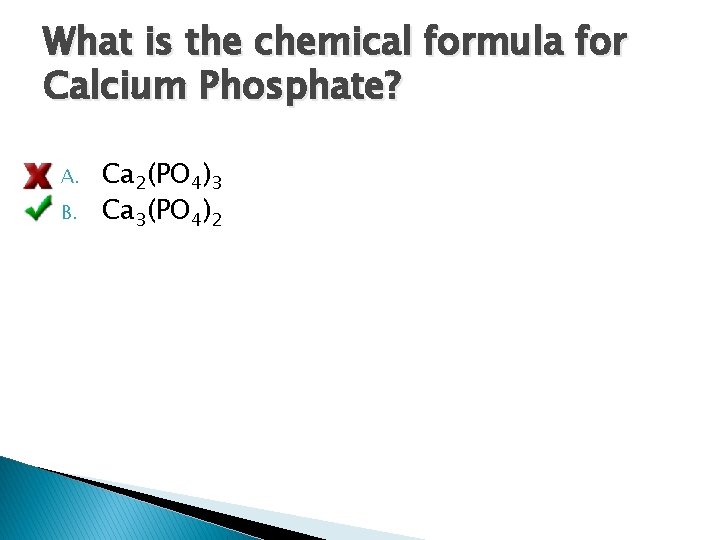
What is the chemical formula for Calcium Phosphate? A. B. Ca 2(PO 4)3 Ca 3(PO 4)2

Name Formula � barium fluoride � aluminum bromide � strontium phosphate

Write the formula for Potassium Nitride

Stock System (Roman numerals) � Some transition metals can have multiple charges ◦ A roman numeral is used to indicate charge � Ex) Chromium can be +2 (II) or +3 (II) ◦ Chromium (II) Sulfide = Cr+2 & S-2 = Cr. S ◦ Chromium (III) Sulfide = Cr+3 & S-2 = Cr 2 S 3

Write the formula for silver (I) hydroxide Name the compound Fe. N Write the formula for Copper (II) Oxide

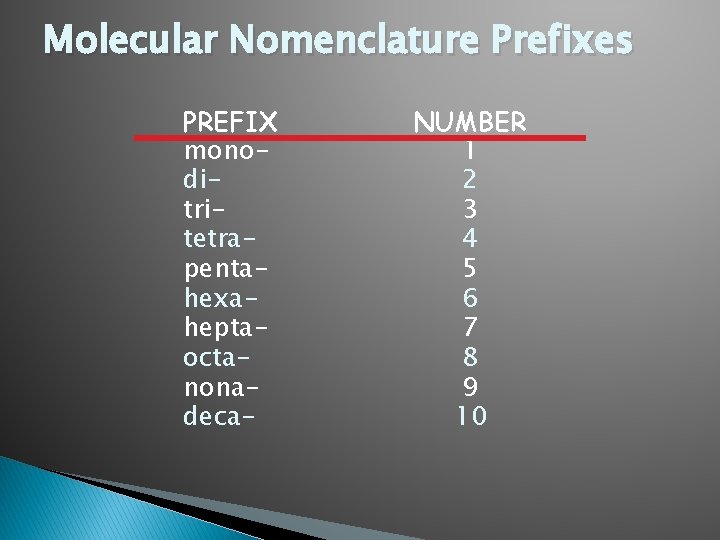
Molecular (Covalent) Nomenclature for two nonmetals � Prefix System (binary compounds) 1. Least electronegative atom comes first. 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. 3. Mono- is OPTIONAL on the SECOND element 4. Change the ending of the second element to -ide.

Molecular Nomenclature Prefixes PREFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10

Molecular Nomenclature: Examples • CCl 4 • carbon tetrachloride • N 2 O • dinitrogen monoxide • SF 6 • sulfur hexafluoride

More Molecular Examples • arsenic trichloride • As. Cl 3 • dinitrogen pentoxide • N 2 O 5 • tetraphosphorus decoxide • P 4 O 10

Name the compound CCl 4 What is the chemical formula for Sulfur Trioxide