CHEMICAL KINETICS Rate of Reaction Rate Laws Importance














- Slides: 22

CHEMICAL KINETICS Rate of Reaction Rate Laws

Importance of Reaction Rates Many reactions, though spontaneous, do not necessarily occur at a reasonable rate. The following reactions are thermodynamically possible but are extremely slow at room temperature conditions: C 6 H 12 O 6(s) + 6 O 2(g) 6 CO 2(g) + 6 H 2 O(g) diamond(s) + O 2(g) CO 2(g) Other spontaneous reactions occur rapidly and often explosively. Acid-base neutralization: Na. OH(aq) + HCl(aq) Na. Cl(aq) + H 2 O(l) Sodium metal on cold water: 2 Na(s) + 2 H 2 O(l) 2 Na. OH(aq) + H 2(g) (sodium is stored under oil) White phosphorus in air: phosphorus is stored under water) P 4(s) + 5 O 2(g) 2 P 2 O 5(s) (white While some reactions are beneficial if slowed down (ex. rusting, decomposition of materials, etc), others are more economical if sped up (ex. Industrial production of pharmaceuticals, chemicals, etc).

The rate of a reaction – how fast or slow a reactant is consumed or a product is formed. Example: Find the average rate of product increase, using data given.

If the reaction is A B (i. e. mole ratio of 1: 1), then: Average rate = ΔConcentration product = ΔT - ΔConcentrationreactant ΔT In other cases, the stoichiometry would indicate the relationship of the rates of the reactants and products. Example: A 2 2 A, [A 2] went from 0. 200 M to 0. 166 M in 8. 4 min. Find the average rate of A 2 used up and A formed in reaction.

1. Nature of reactants: ■ Simple ions in aqueous solution react extremely quickly. ■ Covalent compounds usually react slowly. 2. Phase of the reactants, surface area of solids Factors affectin g reactio n rates: ■ Reaction rates increase from chunk to powder form, to liquid, to gas. ■ Aqueous ionic reactions are almost instantaneous. ■ Reactions in liquid state and homogeneous gas reactions are much faster: Na. Cl(aq) + Ag. NO 3(aq) Ag. Cl(s) + Na. NO 3(aq) is instantaneous Na. Cl(s) + Ag. NO 3(aq) Reaction undetectable if any. 3. Concentration of reactants 4. Temperature 5. Catalyst 6. Other factors affecting reaction rates ■ higher pressure in gas reactions causes faster rates (same as concentration factor) ■ reactant and solvent substitutions may cause changes in reaction rates (intermolecular forces); ■ using UV light, electrical sparks (different photons) for initiation purposes.

How reaction rates are monitored and measured: By observing some change in a physical or chemical property as reaction progresses. Ø Ø Measure the formation of a gas by measuring changes in volume or pressure If a gas is released, measuring the change in mass of the reaction system Measure a change in p. H if acid or base are either formed or consumed during the reaction Monitoring changes in colour, if possible, using a spectrophotometer that measures the colour intensity of a solution Ø If there is a change in electrical conductivity of a solution this can be used to measure the rate Example: Explain what change you would monitor in the following reactions: 1. Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(aq) + H 2 O(l) + CO 2(g) 2. 5(COOH)2 + 2 Mn. O 4 -(aq) + 6 H+(aq) 10 CO 2(g) + 2 Mn 2+(aq) + 8 H 2 O(l) purple v. pale pink 3. (CH 3)3 CBr(aq) + H 2 O(l) (CH 3)3 COH(aq) + H+(aq) + Br-(aq)

■ AP practice questions

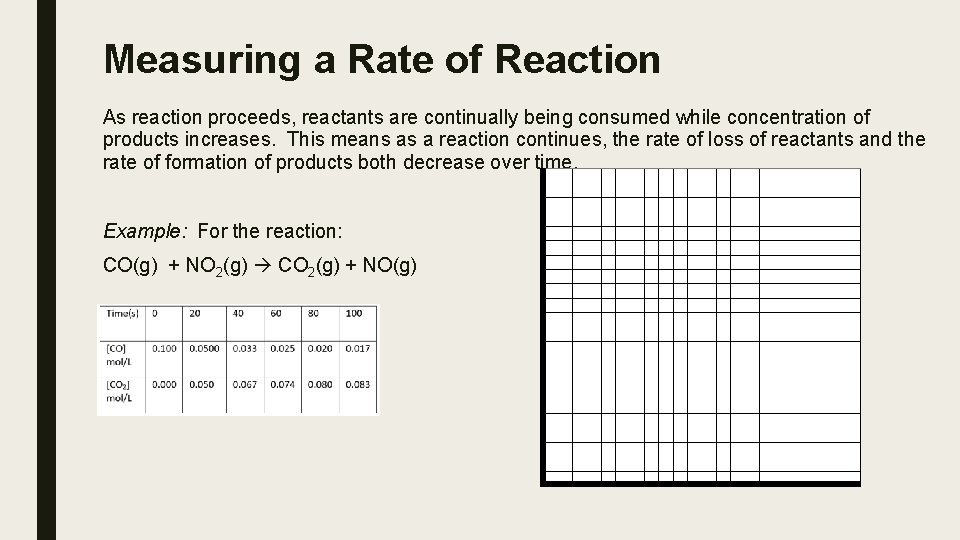
Measuring a Rate of Reaction As reaction proceeds, reactants are continually being consumed while concentration of products increases. This means as a reaction continues, the rate of loss of reactants and the rate of formation of products both decrease over time. Example: For the reaction: CO(g) + NO 2(g) CO 2(g) + NO(g)

■ Find the average rate from 0 s to 40 s: ■ The average rate is obtained by finding the slope between two points ■ The instantaneous rate is obtained by finding the tangent ■ Eventually, the rates become steady i. e. concentrations of reactants and products become equal. ■ The reaction has reached an equilibrium (balanced) state. Rateforward = Ratereverse ■ Reaction rates may be obtained using graphical methods: – by plotting Concentration vs Time graph and the tangents for instantaneous rates – by plotting log Concentration vs Time graph or Reciprocal Concentration vs Time (we will look at in 5. 3)

Introduction to Rate Law Types of Rate Laws Depending on the type of data that you can collect there are two different rate laws you can use to determine the rate of the reaction. Once you determine one rate, you will also know the other. Differential rate law: (also called the rate law) A rate law that expresses how the rate depends on concentration. This law is used when it is possible to determine experimentally the rate change with respect to concentration. Integrated rate law: A rate law that describes how the concentration depends on time. This law is used if you look at concentration change with respect to time. (We will look at this in 5. 3. )

Differential Rate Law In general: As concentration of reactants increases, the rate increases. Therefore, in general, the reaction rate is proportional to the concentration of reactants. for reaction: a. A + b. B products rate a [A]m[B]n or rate = k[A]m[B]n

Rate Law Equation: Shows the relationship between the concentrations of the reactants and the rate of the reaction. It is determined through experiment and cannot be determined by looking at the reaction equation. rate = k[A]m[B]n … etc. where: Ø k is the rate constant – It is constant under constant conditions but is dependent on temperature Ø [A], [B], etc. are the concentrations of reactants Ø m, n, etc. are the rate law exponents – They are determined experimentally – They do not change with temperature – The overall reaction order is m+n…

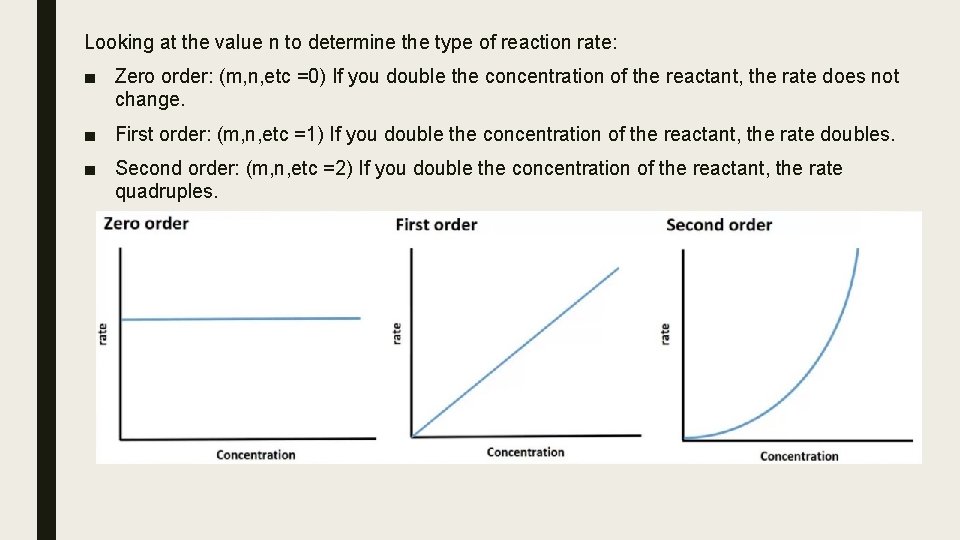
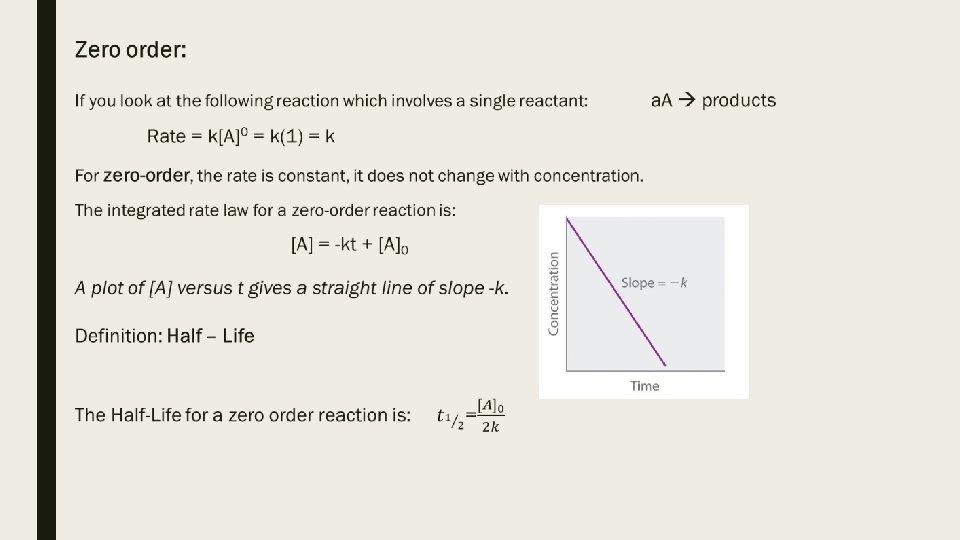
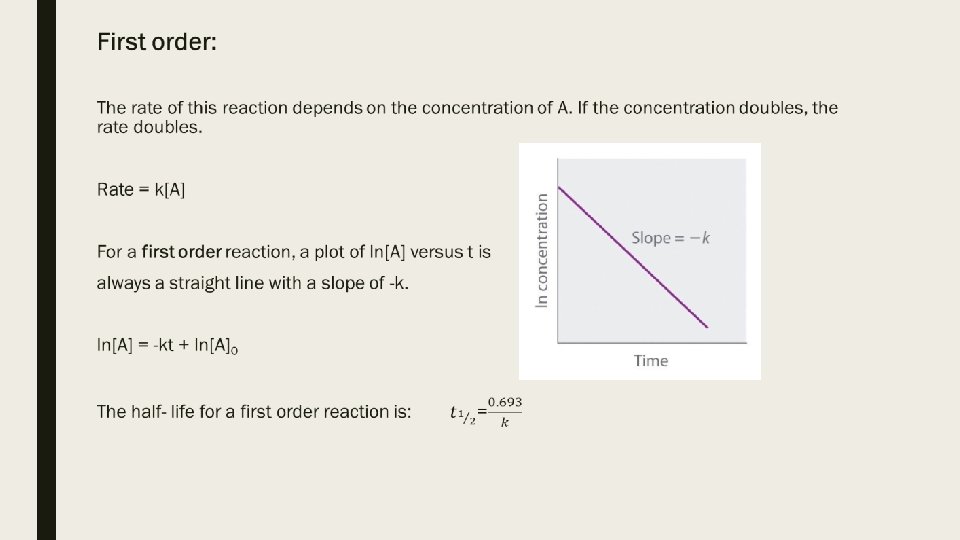
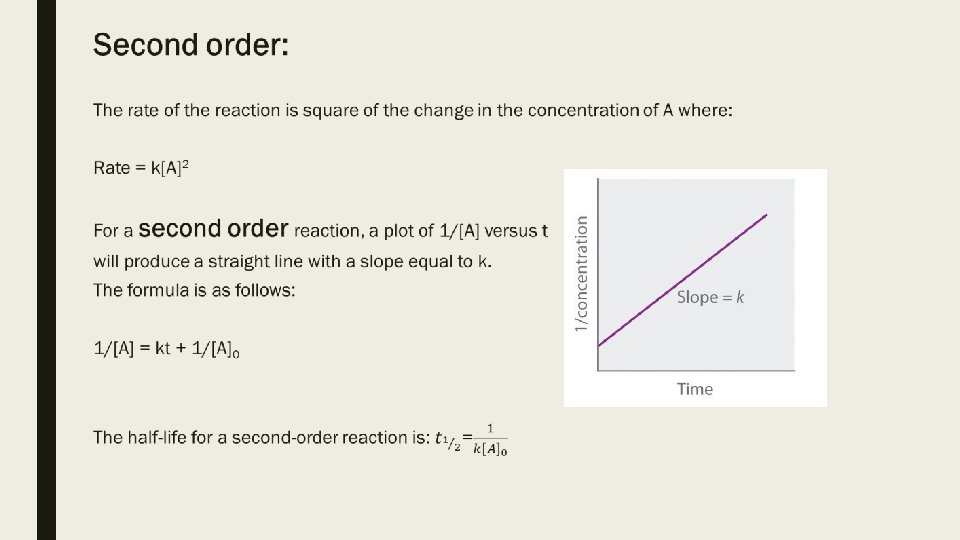
Looking at the value n to determine the type of reaction rate: ■ Zero order: (m, n, etc =0) If you double the concentration of the reactant, the rate does not change. ■ First order: (m, n, etc =1) If you double the concentration of the reactant, the rate doubles. ■ Second order: (m, n, etc =2) If you double the concentration of the reactant, the rate quadruples.

Normally in the dry lab format, the information to calculate the rate law is presented in a table. Step 1: Write the rate law expression. Step 2: Make a comparison of experiment 1 and 2 to determine the effect of [NO 2 -] on the initial rate. Step 3: Make a comparison of experiment 2 and 3 to determine the effect of [NH 4+] on the initial rate.

Step 4: Use any of the experiments to determine the rate constant k. Watch your units for k…they are important. Maybe: Step 5: Recheck you values with another experiment. Step 6: If required, provide the overall reaction order.

■ AP practice questions

Concentration Changes over Time Types of Rate Laws Depending on the type of data that you can collect there are two different rate laws you can use to determine the rate of the reaction. Once you determine one rate, you will also know the other. Differential rate law: (also called the rate law) A rate law that expresses how the rate depends on concentration. This law is used when it is possible to determine experimentally the rate change with respect to concentration. rate = k[A]m[B]n (this is what we studied in the last section) Integrated rate law: A rate law that describes how the concentration depends on time. This law is used if you look at concentration change with respect to time.



Note: First order reactions commonly show up in questions. Decaying, such as nuclear decaying, is first order. For example: The half-life for the radioactive decay of C-14 is 5730 years. a) Write the integrated rate law for this process. b) Write the differential rate law for this process. c) How long will it take for C-14 in a sample to be 25% of its original amount? d) What is the rate of loss of C-14 when 2. 5 g are present in a sample?


■ AP practice questions