Chemical Kinetics Khadijah Hanim Abdul Rahman Uni MAP

Chemical Kinetics Khadijah Hanim Abdul Rahman Uni. MAP ERT 108 – Physical Chemistry Semester II- 2010/2011 1

Subtopics �Experimental Chemical and Kinetics Reactions �First Order Reactions �Second Order Reactions �Reaction Rates and Reaction Mechanisms �Light Spectroscopy and Adsorption Chemistry (Experimental methods for fast reactions). ERT 108 – Physical Chemistry Semester II- 2010/2011 2

Experimental Chemical and Kinetics Reactions �Rates of chemical Reactions: Ø the rate of speed with which a reactant disappears or a product appears. Ø the rate at which the concentration of one of the reactants decreases or of one of the products increases with time. Ø typically, mol L-1 s-1. ERT 108 – Physical Chemistry Semester II- 2010/2011 3

The rates of reactions �Depends on composition and temperature of reaction mixture. (a) Definition of rate - as the slope of the tangent drawn to the curve showing the variation of conc with time ERT 108 – Physical Chemistry Semester II- 2010/2011 4
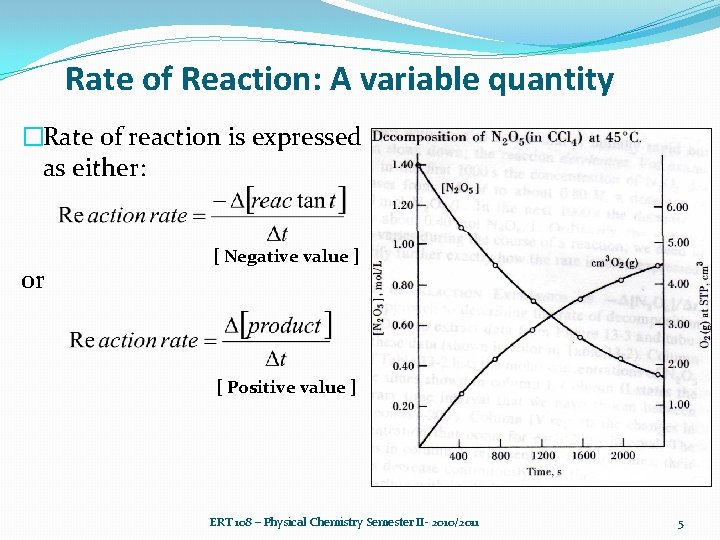
Rate of Reaction: A variable quantity �Rate of reaction is expressed as either: or [ Negative value ] [ Positive value ] ERT 108 – Physical Chemistry Semester II- 2010/2011 5

Consider a reaction: A + 2 B 3 C + D � Rate of consumption – (one of the reactants, A or B) at a given time is d[R]/dt, R is A/B. � Rate of formation – (products, C/D denotes as P) at a given time is d[P]/dt. � These rates are positive values. The +/- signindicate that conc is increasing/decreasing. ERT 108 – Physical Chemistry Semester II 2010/2011 6

�From the stoichiometry: �Rate of reaction is related to rates of change of concentration of products and reactants �Undesirability of having different rates to describe the same reaction- using the extent of reaction, ξ. �v. J is the stoichiometric coefficient for species J. �Rate of reaction, r ERT 108 – Physical Chemistry Semester II- 2010/2011 7
![�For homogenous reaction the V can be taken inside the differential, [J]=n /V J �For homogenous reaction the V can be taken inside the differential, [J]=n /V J](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-8.jpg)
�For homogenous reaction the V can be taken inside the differential, [J]=n /V J �For heterogeneous reaction, use the surface area (constant), A as substitution to V, σJ=n /A J �Common units for r = mol dm-3 s-1 or related units for homogenous reaction �For heterogeneous reaction= mol m-2 s-1 ERT 108 – Physical Chemistry Semester II- 2010/2011 8

�Example 1 �The rate of change of molar conc of CH 3 radicals in the reaction 2 CH 3(g) was reported as d[CH 3]/dt= -1. 2 mol dm-3 s-1 under particular conditions. What is (a) the rate of reaction (b) the rate of formation of CH 3? ERT 108 – Physical Chemistry Semester II- 2010/2011 9

Rate laws and rate constants �Rate of reaction is often proportional to conc of reactants raised to a power. r = k[A] [B] Each conc raised to first power. �k is the rate constant for the reaction �k- independent of conc but depends on temp. �Experimentally determined equation of this kindrate law of the reaction ERT 108 – Physical Chemistry Semester II- 2010/2011 10

�Rate law- an equation that expresses the rate of reaction as a function of conc of all species present in overall chemical equation r = f([A], [B], …. ) �Rate law is determined experimentally- cannot be inferred from the stoichiometry of balanced chemical equation. �Application of rate law: - To predict the rate of reaction from the composition of mixture - Guide to the mechanism of the reaction- for any proposed mechanism- must be consistent with the observed rate law. ERT 108 – Physical Chemistry Semester II- 2010/2011 11

Reaction order �Many reactions are found to have rate laws of the form r = k [A]a[B]b……. �The power to which the conc of a species is raised in a rate law- the order of the reaction with respect to that species. �A reaction with rate law r = k [A] [B] is first-order in A and first-order in B. �The overall order of a reaction, second-order overallthe sum of the individual orders. �Some reactions obey zero-order rate law- rate that is independent of conc of the reactant. Thus, r=k ERT 108 – Physical Chemistry Semester II- 2010/2011 12
![Integrated rate laws �First-order rate law is or [A] = [A] 0 e-kt Where Integrated rate laws �First-order rate law is or [A] = [A] 0 e-kt Where](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-13.jpg)
Integrated rate laws �First-order rate law is or [A] = [A] 0 e-kt Where [A]0 is the initial conc of A at t = 0 �If ln ([A]/[A]0) is plotted against t- first-order reaction will give a straight line of slope= -k. ERT 108 – Physical Chemistry Semester II- 2010/2011 13

Half lives and time constants �Useful indication of the rate of a first-order chemical reaction- half-life, t 1/2 of a substance. �Half-life: time taken for the conc of reactant to fall to half its initial value. �Time for [A] to decrease from [A]0 to ½[A]0 in first-order reaction: kt 1/2 = -ln ½ = ln 2 ∴ t 1/2 = �Another indication of the rate of a first-order reactiontime constant, τ �time required for the conc of reactant to fall to 1/e of its initial value. kτ = -ln 1/e = 1 ∴ time constant, τ = 1/k ERT 108 – Physical Chemistry Semester II- 2010/2011 14
![Second-order reactions �Second-order rate law: � is or �Where [A]0 is the initial conc Second-order reactions �Second-order rate law: � is or �Where [A]0 is the initial conc](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-15.jpg)
Second-order reactions �Second-order rate law: � is or �Where [A]0 is the initial conc of A (at t = 0) �To plot a straight line for second order reaction- plot 1/[A] against t. the slope= k. �The half life for second order reaction is ERT 108 – Physical Chemistry Semester II- 2010/2011 15

Half-life of nth-order reaction �In general for nth-order reaction (with n > 1) of the form A products, the half-life is related to the rate constant and the initial conc of A by ERT 108 – Physical Chemistry Semester II- 2010/2011 16
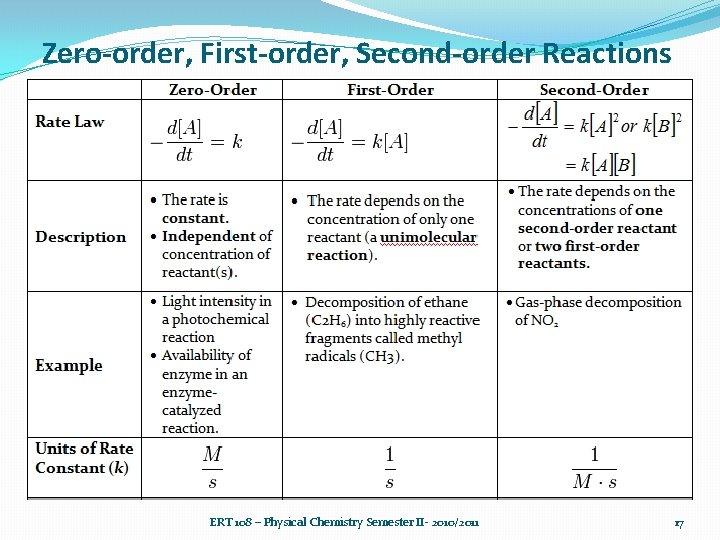
Zero-order, First-order, Second-order Reactions ERT 108 – Physical Chemistry Semester II- 2010/2011 17

Zero-order, First-order, Second-order Reactions ERT 108 – Physical Chemistry Semester II- 2010/2011 18

Zero-order, First-order, Secondorder Reactions ERT 108 – Physical Chemistry Semester II 2010/2011 Zero order First order Second order 19
![Example 2 (a) When [N 2 O 5] =0. 44 M, the rate of Example 2 (a) When [N 2 O 5] =0. 44 M, the rate of](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-20.jpg)
Example 2 (a) When [N 2 O 5] =0. 44 M, the rate of decomposition of N 2 O 5 is 2. 6 x 10 -4 mol L-1 s-1. Ø what is the value of k for this first-order reaction? (b) N 2 O 5 initially at a concentration of 1. 0 mol/L in CCl 4, is allowed to decompose at 450 C. At what time will [N 2 O 5] be reduced to 0. 50 M? ERT 108 – Physical Chemistry Semester II 2010/2011 20
![Example 3 Time, min [A], M log [A] 1/[A] 0 1. 00 0. 00 Example 3 Time, min [A], M log [A] 1/[A] 0 1. 00 0. 00](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-21.jpg)
Example 3 Time, min [A], M log [A] 1/[A] 0 1. 00 0. 00 1. 00 5 0. 63 -0. 20 1. 59 10 0. 46 -0. 34 2. 17 15 0. 36 -0. 44 2. 78 25 0. 25 -0. 60 4. 00 �The data of the above table were obtained for the decomposition reaction: A → 2 B + C. (a) Establish the order of the reaction. (b) What is the rate constant, k? ERT 108 – Physical Chemistry Semester II 2010/2011 21
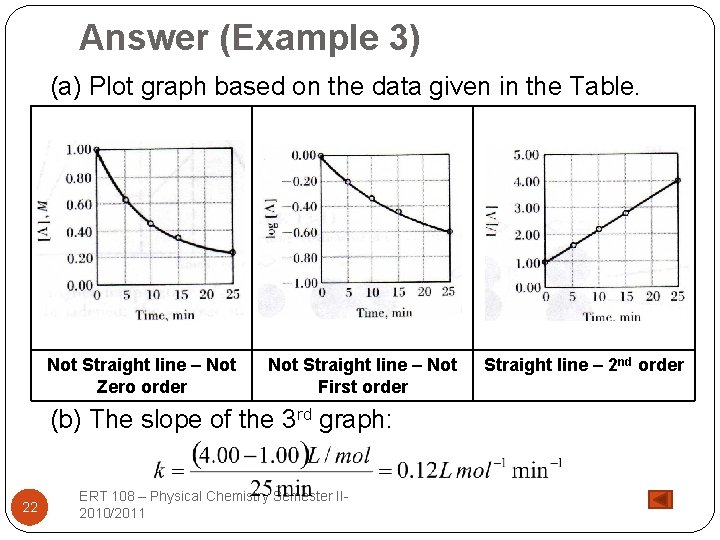
Answer (Example 3) (a) Plot graph based on the data given in the Table. Not Straight line – Not Zero order (b) The slope of 22 Not Straight line – Not First order the 3 rd graph: ERT 108 – Physical Chemistry Semester II 2010/2011 Straight line – 2 nd order

Determination of the rate law �Experimental data gives species conc at various times during the reaction. � a few methods to determine the rate law from experimental conc vs. time data. �Consider the following: r = k [A]a[B]b……. it is usually the best to find the order of a, b, … first and then the rate constant, k. ERT 108 – Physical Chemistry Semester II- 2010/2011 23

Determination of rate law- half-life method �Applies when the rate law has the form r = k[A]n. then this equation and apply. �If n = 1, then t 1/2 is independent of [A]0. If n ≠ 1, then gives: �A plot of log 10 t 1/2 vs. log 10 [A] – gives straight line of slope = n-1. ERT 108 – Physical Chemistry Semester II- 2010/2011 24
![�To use the half-life method for determination of rate law: - Plot [A] vs. �To use the half-life method for determination of rate law: - Plot [A] vs.](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-25.jpg)
�To use the half-life method for determination of rate law: - Plot [A] vs. t - Pick any [A] value, eg. [A]’ and finds the point where [A] has fallen to ½ [A]’. The time interval between this 2 points is t 1/2 for the initial conc [A]’. - Pick another point [A]” and determines the t 1/2 for this A conc. - Repeat this process several times - Plot log 10 t 1/2 vs. the log of the corresponding initial A conc and measures the slope. ERT 108 – Physical Chemistry Semester II- 2010/2011 25

Example 4 �Data for the dimerization 2 A A 2 of certain nitrile oxide (compound A) is ethanol solution at 40 o. C follow: [A]/(mmol /dm-3) t/min 68. 0 50. 2 40. 3 33. 1 28. 4 22. 3 18. 7 14. 5 0 40 80 120 160 240 300 420 Find the reaction order using the half-life method ERT 108 – Physical Chemistry Semester II- 2010/2011 26

Method of Initial Rates �This simple method of establishing the exponents in a rate equation involves measuring the initial rate of reaction, ro for different sets of initial concentration. �Suppose we measure r 0 for the 2 different initial A conc [A]0, 1 and [A]0, 2 while keeping [B]0, [C]o, … fixed. �With only [A]0 changed and with the rate law assumed to have form r = k[A]a[B]b…[L]l, the ratio of initial rates for run 1 and 2 is ro, 2/ro, 1 = ([A]o, 2/[A]0, 1)n 27

Example 5 �The data of three reactions involving S 2 O 82 - and I- were given in the below table. (i) Use the data to establish the order of reaction: with respect to S 2 O 82 -, the order with respect to I- & the overall order. ERT 108 – Physical Chemistry Semester II 2010/2011 28

Example 5 (ii) Determine the value of k for the above reaction. (iii) What is the initial rate of disappearance of in a S 2 O 82 - reaction in which the initial concentrations are [S 2 O 82 - ] =0. 050 M & [I-]=0. 025 M? (iv) What is the rate of formation of SO 42 - in Experiment 1? ERT 108 – Physical Chemistry Semester II- 2010/2011 29

The temperature dependence of reaction rates �Chemical reaction begins with a collision between molecules of A and molecules of B. �Chemical reactions tend to go faster at higher temperature. Ø slow down some reactions by lowering the temperature. �Increasing the temperature increases the fraction of the molecules that have energies in excess of the activation energy. Ø this factor is so important that for many chemical reactions it can lead to a doubling or tripling of the reaction rate for a temperature increase of only 100 C. ERT 108 – Physical Chemistry Semester II- 2010/2011 30

Reaction rates: Effect of temperature �In 1889, Arrhenius noted that the k data for many reactions fit the equation: where A & Ea are constants characteristics of the reaction & R = the gas constant. Ø Ea – the Arrhenius activation energy (k. J/mol or kcal/mol) Ø A – the pre-exponential factor (Arrhenius factor). Ø the unit of A is the same as those of k. �Taking log of the above equation: ERT 108 – Physical Chemistry Semester II 2010/2011 31

Reaction rates: Effect of temperature �If the Arrhenius equation is obeyed: Ø a plot of ln k versus 1/T is a straight line with slope = (-Ea/R) and A is the intercept of the line at 1/T = 0. Ø This enables Ea and A to be found. • Another useful equation: (eliminate the constant A). Ø T 2 and T 1 - two kelvin temperatures. Ø k 2 and k 1 - the rate constants at these temperatures. Ø Ea – the activation energy (J/mol) Ø R – the gas constant (8. 314 Jmol-1 K-1). ERT 108 – Physical Chemistry Semester II- 2010/2011 32

�The activation energy, Ea: the minimum kinetic energy that reactants must have in order to form products. �The pre-exponential factor, A: a measure of the rate at which collisions occur irrespective of their energy. ERT 108 – Physical Chemistry Semester II 2010/2011 33

Reaction Mechanisms �The mechanism of reaction is the sequence of elementary steps involved in a reaction �Most reactions occur in a sequence of steps called elementary reactions. �A mechanism is a hypothesis about the elementary steps through which chemical change occurs. �A typical elementary reaction is H + Br 2 HBr + Br �Chemical equation for elementary reaction: equation only represents the specific process occurring to individual molecules �Molecularity of an elementary reaction is the no of molecules coming together to react in an elementary reaction. ERT 108 – Physical Chemistry Semester II- 2010/2011 34

Reaction Mechanisms �Elementary processes in which a single molecule dissociates (unimolecular) or two molecules collide (bimolecular) much more probable than a process requiring the simultaneous collision of three bodies (termolecular). �All elementary processes are reversible and may reach a steady-state condition. In the steady state the rates of the forward & reverse processes become equal. The concentration of some intermediate becomes constant with time. �One elementary process may occur much more slower than all the others. In this case, it determines the rate at which the overall reaction proceeds & is called the ratedetermining step. ERT 108 – Physical Chemistry Semester II- 2010/2011 35

Rate laws and equilibrium constants for elementary reactions �An overall reaction occurs as a series of elementary steps �These steps constituting the mechanism of reaction �This section consider the rate law for elementary reaction. �Consider a bimolecular elementary reaction A + B products, the rate of reaction, will be proportional to ZAB, the rate of A-B collisions per unit of time. �∴ r for an elementary bimolecular ideal-gas reaction will be r = k[A][B] ERT 108 – Physical Chemistry Semester II- 2010/2011 36

�For unimolecular ideal-gas reaction B products, fixed probability that any particular B molecule will decompose/isomerize to products per unit time. �The rate of reaction, r = k[B] �Similar considerations apply to reactions in ideally/ideally dilute solution. �In summary, in an ideal system, the rate law for the elementary reaction a. A + b. B products is r = k[A]a[B]b, where a + b is 1, 2 or 3. �For an elementary reaction, the orders in the rate law equal the coefficients of the reactants. ERT 108 – Physical Chemistry Semester II- 2010/2011 37

�Relation between the equilibrium constant for a reversible elementary reaction and rate constants forward and reverse reactions. �Consider the reversible elementary reaction kf a. A + b. B ⇌ c. C + d. D kb �rate laws forward (f) and back (b) elementary reactions are rf = kf [A]a[B]b and rb = kb [C]c[D]d. �At equilibrium, these opposing rates are equal: rf, eq = rb, eq or kf([A]eq)a([B]eq)b = kb([C]eq)c([D]eq)d and � = Kc = kf/kb ERT 108 – Physical Chemistry Semester II- 2010/2011 38

(b) The rate-determining-step approximation �In rate-determining step approximation- reaction mechanism assumed to consist 1 or more reversible reactions that stay close to equilibrium during most of the reaction. � followed by relatively slow rate-determining step then in turn followed by 1 or more rapid reactions. �As an example: k 1 k 3 k 2 A⇌B⇌C⇌D k-1 k-2 k-3 where step 2 (B⇌C)- assumed to be rate-determining step. �For this assumption to be valid- k-1 >> k 2 ERT 108 – Physical Chemistry Semester II- 2010/2011 39

�The slow rate of B C compared with B A – ensures that most B molecules go back to A rather than to C- ensuring that step 1 (A ⇌ B) remain close to equilibrium. �k 3 >> k 2 and k 3 >> k-2, to ensure that step 2 acts as ‘bottleneck’ and product D is rapidly formed from C. �The overall rate is controlled by the rate-limiting step B C. �Since we are examining rate of the forward reaction A D, we further assume that k 2[B] >> k-2[C]. �During early stage- the conc of C will be lower than Bthis condition will hold. Thus, we neglect reverse reaction for step 2. ERT 108 – Physical Chemistry Semester II- 2010/2011 40

�The relative magnitude of k 1 compared with k 2 is irrelevant to the validity of the rate-determining-step approximation. �∴ the rate constant k 2 of the rate-determining step might be larger than k 1. �However, the rate r 2 = k 2[B] of the rate-determing step must be smaller than r 1=k 1[A] of the first step. �This follows from k 2<<k-1 and k 1/k-1 [B]/[A] (the conditions for step 1 –near equilibrium). �For reverse overall reaction, the rate-determining step is the reverse of that forward reaction. �For example: the rate-determining step is C B. So, k-2 << k 3 (ensures that step D⇌C is in equilibrium) and k-1>> k 2 (ensures that B A is rapid). ERT 108 – Physical Chemistry Semester II- 2010/2011 41

The rate determining-step approximation �The rate law for the Br- - catalyzed aqueous reaction H+ +HNO 2 + C 6 H 5 NH 2 C 6 H 5 N 2+ + 2 H 2 O is observed to be r = k[H+][HNO 2][Br-] …. . (1) A proposed mechanism is k 1 + HNO 2⇌ H 2 NO 2+ rapid equilib k-1 2 Br- k H 2 NO 2+ + ONBr + H 2 O slow ……(2) k 3 ONBr + C 6 H 5 NH 2 C 6 H 5 N 2+ +H 2 O + Brfast Deduced the rate law for this mechanism and relate the observed rate constant, k in (1)to the rate constants in assumed mechanism (2). ERT 108 – Physical Chemistry Semester II- 2010/2011 42

�The second step in (2) is rate limiting. Since step 3 is much faster than 2, we can take d[C 6 H 5 N 2+]/dt as = rate of formation of ONBr in step 2. therefore, the reaction rate is r = k 2[H 2 NO 2+][Br-] …. . (3) (since step 2 is an elementary reaction, its rate law is determined by its stoichiometry. The species H 2 NO 2+ in (3) is a reaction intermediate and we want to express r in terms of reactants and products. Since step 1 is in near equilib, gives: Kc, 1 = k 1/k-1 =[H 2 NO 2+]/[H+][HNO 2] And [H 2 NO 2+] = (k 1/k-1) [H+][HNO 2] Substitute in (3) gives r = (k 1 k 2/k-1)[H+][HNO 2][Br-] k = k 1 k 2/k-1 = Kc, 1 k 2 ERT 108 – Physical Chemistry Semester II- 2010/2011 43

The steady-state approximation �Multistep reaction mechanisms usually involve 1 or more species that do not appear in overall equation. a b Ak Ik P �After an initial induction period, [I] will start at 0, rise to max, [I]max, and then fall back to 0. �During the major part of the reaction, the rates of change of conc of all reaction intermediates are negligibly small, therefore d[I]/dt = 0 for each reaction intermediate. �The steady-state approximation assumes that rate of formation of reaction intermediate = rate of destruction. ERT 108 – Physical Chemistry Semester II- 2010/2011 44

Steady-state approximation �Apply steady-state approximation to the mechanism for Br H+ + HNO 2 + C 6 H 5 NH 2 C 6 H 5 N 2+ + 2 H 2 O Given in the preceding exercise to find the predicted law. - ERT 108 – Physical Chemistry Semester II- 2010/2011 45

�To apply for rate-determining step approximation: (a) Take the reaction rate, r = rate of determining step (divided by the stoichiometric no srds of rate-determining step, if srds ≠ 1. (b) Eliminates the conc of any reaction intermediates that occur in the rate expression obtained in (a) by using equilibrium-constant expressions. �To apply steady-state approximation: (a) Take the reaction rate, r = rate of formation of product (b) Eliminate the conc of any reaction intermediates that occur in (a) by using d[I]/dt = 0 to find the conc of each I (c) If step (b) introduces conc of other I, apply d[I]/dt = 0 to eliminate their conc. ERT 108 – Physical Chemistry Semester II- 2010/2011 46

The Hydrogen-Iodine Reaction H 2 (g) + I 2 (g) → 2 HI (g) �Rate of formation of HI = k [H 2][I 2] �The hydrogen-iodine reaction is proposed to be a twostep mechanism [Sullivan J. (1967). J. Chem. Phys. 46: 73]. Ø 1 st step: iodine molecules are believed to dissociate into iodine atoms. Ø 2 nd step: simultaneous collision of two iodine atoms and a hydrogen molecule. (this termolecular step is expected to occur much more slowly – the rate-determining step). ERT 108 – Physical Chemistry Semester II- 2010/2011 47
![The Hydrogen-Iodine Reaction 1 st step: [Fast] 2 nd step: Net: [Slow] �If the The Hydrogen-Iodine Reaction 1 st step: [Fast] 2 nd step: Net: [Slow] �If the](http://slidetodoc.com/presentation_image_h/16c9e35f6880a673129e42f77ff7ae89/image-48.jpg)
The Hydrogen-Iodine Reaction 1 st step: [Fast] 2 nd step: Net: [Slow] �If the reversible step reaches a steady state condition: Ø rate of disappearance of I 2 = rate of formation of I 2 ERT 108 – Physical Chemistry Semester II- 2010/2011 48

The Hydrogen-Iodine Reaction �For the rate-determining step: Rate of formation of HI = k 3 [I]2[H 2] = K[H 2][I 2] (K=k 1 k 3/k 2) ERT 108 – Physical Chemistry Semester II- 2010/2011 49

Example 6 �The thermal decomposition of ozone to oxygen: 2 O 3 (g) → 3 O 2 (g) �The observed rate law: Rate of disappearance of O 3 = �Show that the following mechanism is consistent with this experiment rate law. 1 st: 2 nd: ERT 108 – Physical Chemistry Semester II- 2010/2011 50

Experimental methods for fast reactions �Many reactions are too fast to follow by the classical methods. �Several ways to study fast reactions : 1. Rapid flow methods: (i) Continuous flow (ii) Stopped flow 2. Relaxation methods: (i) Temperature jump (T-jump) method (ii) Pressure jump method (iii)Electric field jump method 3. Flash photolysis 4. Shock tube 5. Nuclear-magnetic-resonance (NMR) spectroscopy ERT 108 – Physical Chemistry Semester II- 2010/2011 51

Experimental methods for fast reactions �Continuous flow system �Liquid phase: Ø Reactant A & B are rapidly drive into the mixing chamber M by pushing in the plungers of the syringes. Ø Mixing occurs in 0. 5 – 1 ms. Ø The reaction mixture then flows through the narrow observation tube, where one measures the light absorption at a wavelength (at which one species absorbs to determine the concentration of that species). ERT 108 – Physical Chemistry Semester II- 2010/2011 52
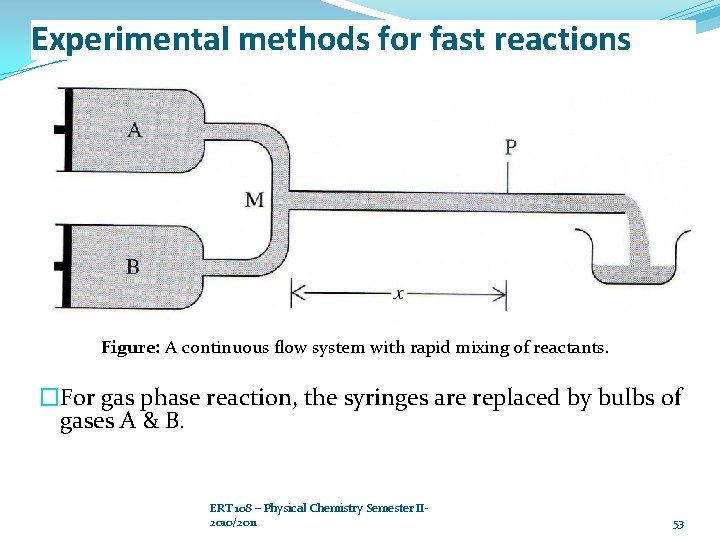
Experimental methods for fast reactions Figure: A continuous flow system with rapid mixing of reactants. �For gas phase reaction, the syringes are replaced by bulbs of gases A & B. ERT 108 – Physical Chemistry Semester II 2010/2011 53

Experimental methods for fast reactions �Stopped flow method: Ø the reactants mixed at M & rapidly flow through the observation tube into the receiving syringe, driving its plunger against a barrier & thereby stopping the flow. Ø this plunger hits a switch which stops the motor driven plungers & triggers the oscilloscope sweep. Ø One observes the light absorption at P as a function of time. • The continuous flow & stopped flow methods are applicable to reactions with half-lives in the range of 0. 001 to 10 s. ERT 108 – Physical Chemistry Semester II- 2010/2011 54
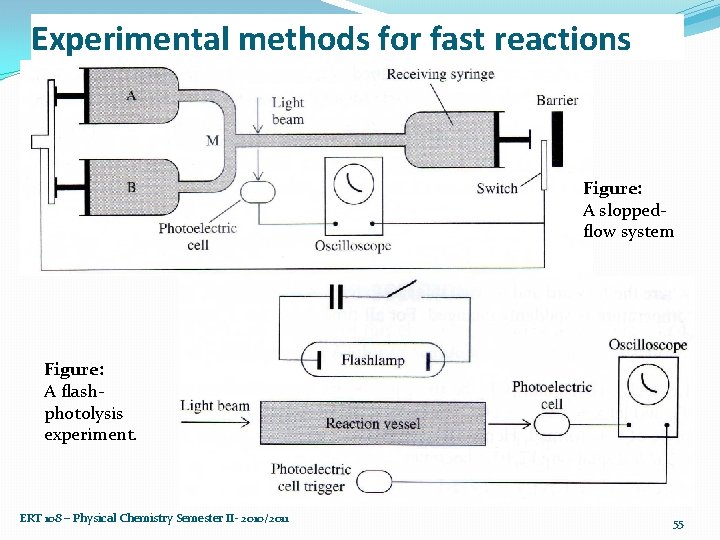
Experimental methods for fast reactions Figure: A sloppedflow system Figure: A flashphotolysis experiment. ERT 108 – Physical Chemistry Semester II- 2010/2011 55

Experimental methods for fast reactions �Relaxation methods: Ø Take a system in reaction equilibrium & suddenly change one of the variables that determine the equilibrium position. Relaxation methods 1. Temperature jump (T-jump) method 2. Pressure jump method 3. Electric field jump method Descriptions A sudden change in T shifts the equilibrium. A sudden change in P shifts the equilibrium. A sudden applied electric field shifts the equilibrium (a change in total dipole moment). �A limitation on relaxation methods – the reaction must be reversible, with detectable amounts of all species present in equilibrium. ERT 108 – Physical Chemistry Semester II- 2010/2011 56

Experimental methods for fast reactions �Rapid flow & relaxation method have been used to measure the rates of proton transfer (acid-base) reactions, complex-ion-formation reactions, ion-pairformation reactions & enzyme-substrate-complex formation system. �Relaxation methods apply rather small perturbations to a system & do not generate new chemical species. �The flash-photolysis and shock tube methods apply a large perturbation to a system, thereby generating one or more reactive species whose reactions are then followed. �NMR spectroscopy is used to measure the rates of certain rapid isomerization & exchange reactions. ERT 108 – Physical Chemistry Semester II- 2010/2011 57
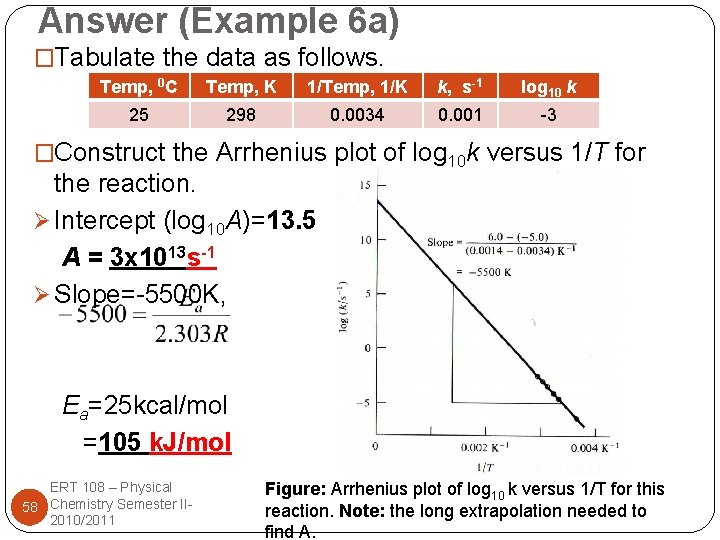
Answer (Example 6 a) �Tabulate the data as follows. Temp, 0 C Temp, K 1/Temp, 1/K k, s-1 log 10 k 25 298 0. 0034 0. 001 -3 �Construct the Arrhenius plot of log 10 k versus 1/T for the reaction. Ø Intercept (log 10 A)=13. 5 A = 3 x 1013 s-1 Ø Slope=-5500 K, Ea=25 kcal/mol =105 k. J/mol ERT 108 – Physical 58 Chemistry Semester II 2010/2011 Figure: Arrhenius plot of log 10 k versus 1/T for this reaction. Note: the long extrapolation needed to find A.

Answer (Example 6 b) �Based on the given info: Ø k 2 = 2 k 1 , ØT 1 = room temperature (298 K), T 2=298+10 = 308 K, �The Arrhenius equation: �Substitute: Ea = 53 k. J/mol 59 ERT 108 – Physical Chemistry Semester II 2010/2011

Answer (Example 7) �Assume the 1 st step reaches the steady state condition: Rate of formation of O = Rate of disappearance of O k 1 [O 3] = k 2 [O 2] [O] �Assume the 2 nd step is the rate-determining step: Rate of disappearance of O 3 = k 3 [O][O 3] 60 ERT 108 – Physical Chemistry Semester II 2010/2011 k = k 1 k 3/k 2) (where
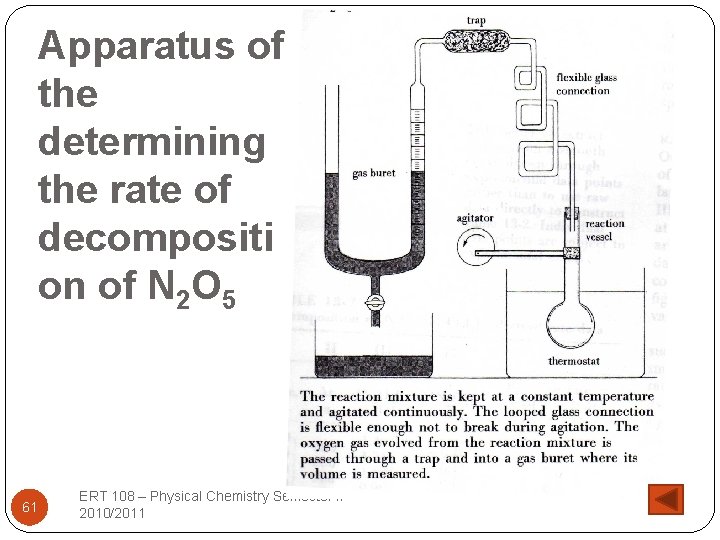
Apparatus of the determining the rate of decompositi on of N 2 O 5 61 ERT 108 – Physical Chemistry Semester II 2010/2011
- Slides: 61