Chemical Formulas The Basics Subscripts in Chemical Formulas

Chemical Formulas: The Basics

Subscripts in Chemical Formulas n n numbers inside formulas lowered below line of type show # of atoms of each element No subscript – understood to be 1 S 8 8 is the subscript, so there are 8 Sulfur toms

Chemical Formulas n n n H 2 O has: 2 hydrogens and 1 oxygen NH 3 has: 1 nitrogen and 3 hydrogens CH 4 has ? 1 carbon and 4 hydrogen CO 2 has ? 1 carbon and 2 oxygen Ag. NO 3 has ? 1 silver, 1 nitrogen, 3 oxygen

# atoms in formulas n n n Subscript outside ( ) multiplied by subscript inside ( ) = # of atoms Al(NO 3)3 Al = 1 N= 1 x 3=3 O= 3 x 3=9 Total = 13 atoms

Coefficients Numbers in front of formulas n Refers to everything following in formula n No coefficient? It’s understood to be 1 n n 4 CO 2 4 is the coefficient

Coefficients 2 H 2 O means 2(H 2 O) n To count atoms multiply: coefficient x subscript n n 2 x 2 = 4 H ’s n 2 x 1 = 2 O ’s

Chemical Formulas n n n 3 H 2 O has: 6 hydrogens and 3 oxygens 2 NH 3 has: 2 nitrogen and 6 hydrogens 4 CH 4 has ? 4 carbon and 16 hydrogen 3 CO 2 has ? 3 carbon and 6 oxygen 2 Ag. NO 3 has ? 2 Ag, 2 N, 6 O
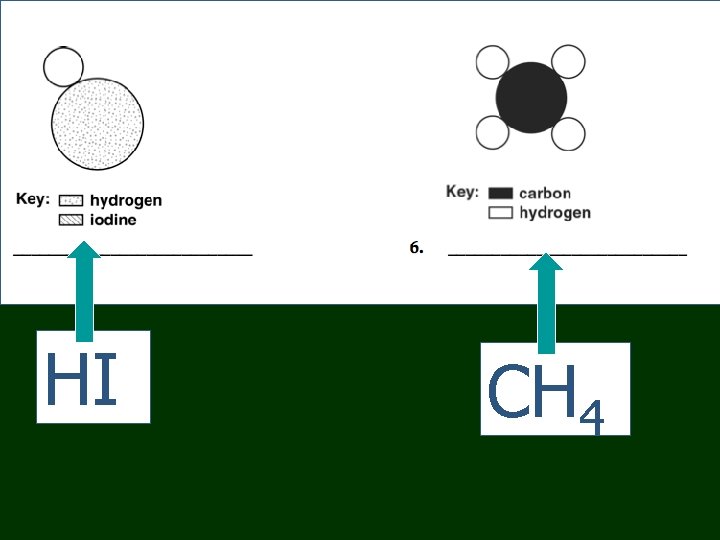
HI CH 4

Lets Try one more n 4 Al(NO 3)3

Hydrates n n group of salts that have water molecules stuffed in their empty spaces Ex: Cu. SO 4 5 H 2 O n means “is associated with” or “included” n n DOES NOT refer to multiplication Not a true chemical bond: n structure is definite so n n # empty spaces is definite & # water molecules is definite

Cu. SO 4 5 H 2 O n Count up the atoms! Cu x 1 Sx 1 O x 4 H x 10 Ox 5 total = 21 atoms
![5[Cu. SO 4 5 H 2 O] n Count up the atoms! Cu x 5[Cu. SO 4 5 H 2 O] n Count up the atoms! Cu x](http://slidetodoc.com/presentation_image_h2/b1416b706439a7d44618a88b68a72d36/image-13.jpg)
5[Cu. SO 4 5 H 2 O] n Count up the atoms! Cu x 5 Coefficients x Subscripts Sx 5 O x 20 (5 x 4) H x 50 (5 x 5 x 2) O X 25 (5 x 5) total = 105 atoms
- Slides: 13