Chemical Formulas Coefficient Subscript of molecules 5 CO
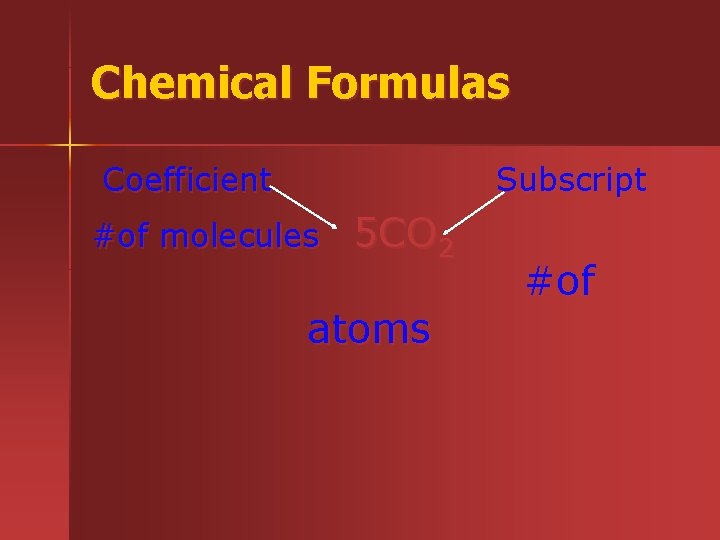
Chemical Formulas Coefficient Subscript #of molecules 5 CO 2 atoms #of

Chemical Reactionstakes place when chemical bonds between atoms are broken and new bonds form. There are the same number of total atoms of each element at the end.

Chemical Equations. Reactants - the chemicals to be combined (left of the arrow) Products - Results of the combinations (right of the arrow) Ex) Na + Cl reactants Na. Cl products

3 Rules to Balancing Equations 1. Check to make sure all formulas are written correctly. 2. Do NOT change subscripts in formulas. Only change coefficients.

Coefficients. Numbers placed in front of a formula to make the number of atoms on each side of the equation equal. Ex) Na + Cl 2 2 Na. Cl Na is NOT equal! A coefficient of 2 must be placed in front of Na to be balanced! 2 Na + Cl 2 Na. Cl

3. Count atoms, then begin with an element that only occurs once on each side of an equation. Ex. A) H 2 + 02 H 2 O Which element is not equal? 1. H 2 +O 2 2 H 2 + O 2 2 H 20 O= H≠ 2 H 2 O balanced!

Ex. B) Na + 2 H 20 Na. OH + H 2 Which element is not equal? Coefficient of 2 added to Na. OH Na + 2 H 20 2 Na. OH + H 2 O= H= Na ≠

4 Types of Chemical Reactions 1. Synthesis Reaction in which two or more substances combine to forms a compound. Sodium + Chloride 2 Na + Cl 2 2 Na. Cl Salt

2. Decomposition Reaction Compound breaks into two or more substances. 2 Hg. O 2 Hg + O 2

3. Single Replacement Reaction One element replaces another element in a compound to form a new compound Fe + Cu. SO 4 Fe. SO 4 + Cu

4. Double Replacement Reaction Occurs when two compounds in a reaction exchange atoms Ag. NO 3 + Na. Cl Ag. Cl + Na. NO 3
- Slides: 11