Chemical Formulas Chemical reactions occur when old bonds

Chemical Formulas
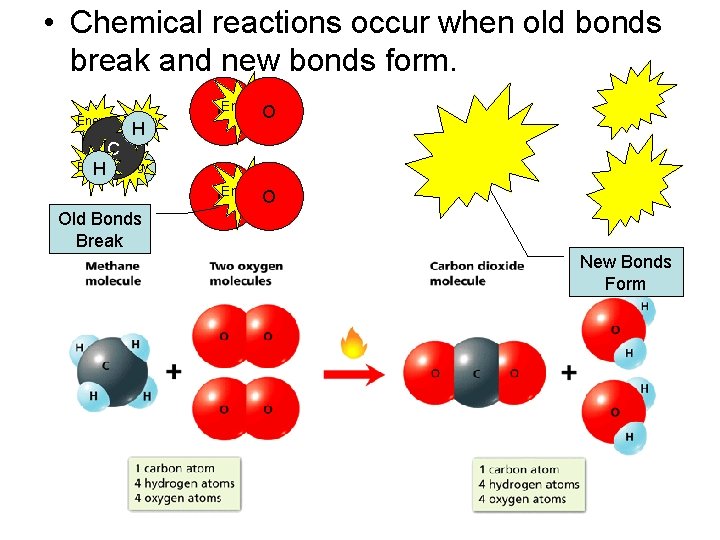
• Chemical reactions occur when old bonds break and new bonds form. Energy H Energy C H Energy. O O Energy H Energy. O O Old Bonds Break New Bonds Form

Chemical Formulas • A chemical formula helps us to see what actually happened in a reaction. Take the easy Sodium Chloride reaction: Na + Cl Na. Cl *Reactants (what goes in) are ALWAYS listed on the left of the arrow *Products (what comes out) are ALWAYS listed on the right of the arrow
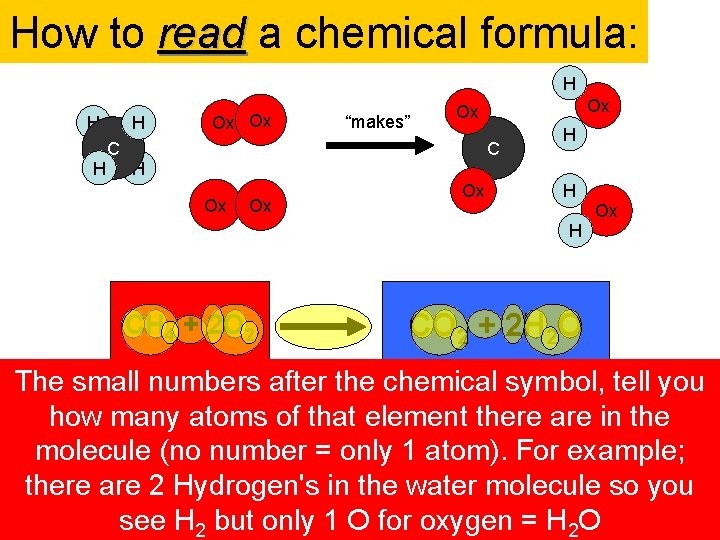
How to read a chemical formula: H H C Ox Ox “makes” Ox Ox C H Ox Ox Ox H H H CH 4 + 2 O 2 Ox CO 2 + 2 H 2 O The small numbersuses after the chemical symbol, tell you The formula the symbols from the The big numbers inare of the together. molecule, you The molecules “+” sign means written “and”. The arrow CH is how many atoms offront that element there are tells in sign the 4 periodic table for each element: C for how many of that molecule there are in the molecule. Reactant (no = only 1 atom). For example; Methane; means “makes”. O 2 number is an. You Oxygen can also molecule; think of CO the is Product 2 Carbon, HHydrogen's for andyou. O Oxygen (What endfor with) reaction (no number 1 molecule). For example; (What you start out Hydrogen with)= only there are 2 in the water molecule so you Carbon arrow sign Dioxide like an and= H sign O is in Water. math. 2 in molecules this theresee are. H 22 but water in this reaction. only 1 Ocase. for oxygen =H 2 O
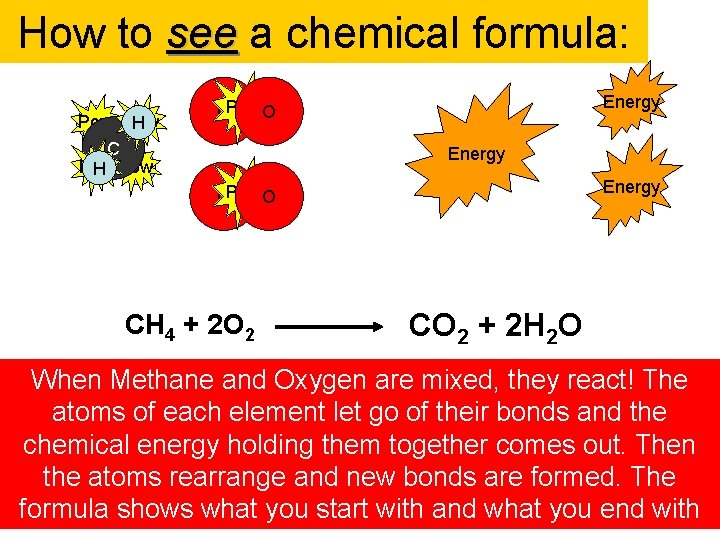
How to see a chemical formula: Pow H C Pow H Energy OPow O CH 4 + 2 O 2 CO 2 + 2 H 2 O When Methane and Oxygen are mixed, they react! The atoms of each element let go of their bonds and the chemical energy holding them together comes out. Then the atoms rearrange and new bonds are formed. The formula shows what you start with and what you end with
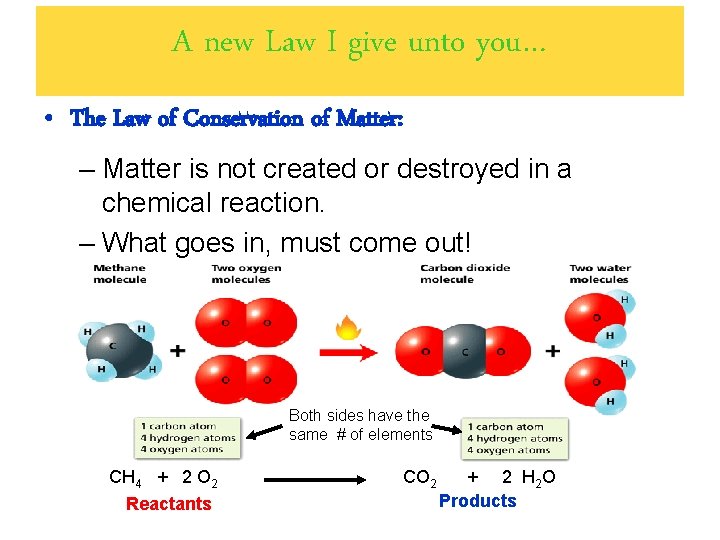
A new Law I give unto you… • The Law of Conservation of Matter: – Matter is not created or destroyed in a chemical reaction. – What goes in, must come out! Both sides have the same # of elements CH 4 + 2 O 2 Reactants CO 2 + 2 H 2 O Products

Conservation of Mass Explained: • During a chemical reaction, matter is neither created or destroyed. The atoms just trade partners around. All the atoms present at the start of the reaction are still present at the end. They are just someplace else. EX: I can change my seating chart around, but that does not mean I have a new class! You are just in different places

- Describing Chemical Reactions If you add up the amounts of Iron and Sulfur What goes in MUST equal what you will see that it equals the amount of Iron out! Sulfide comes made in the reaction.

Notice that the number of Oxygen atoms are not equal on a both sides of the arrow! Step 1: Write the Equation How to write chemical formula The Law of Conservation of Matter says they must be so we have to change up the formula a little… In other words, how in the world do Count the Atoms we Step know 2: that a formula uses 2 or more molecules of one type but only one of another? How in the world do we know that? Well, you gotta do a little math to find out!
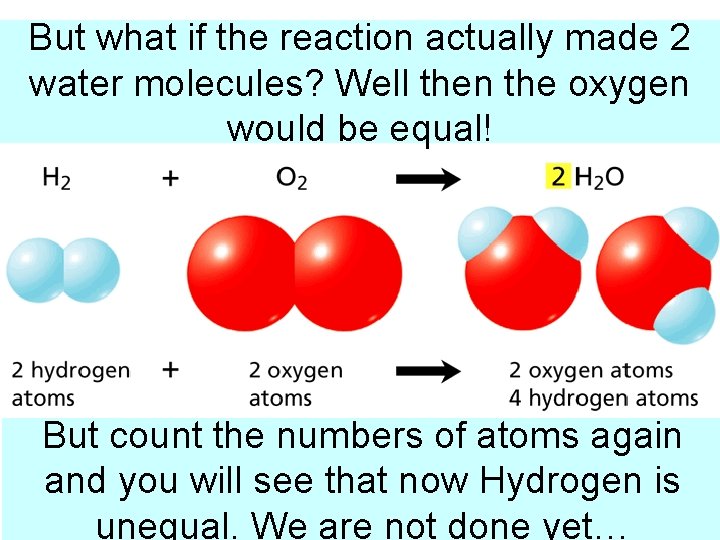
But what if the reaction actually made 2 Step molecules? 3: Use Coefficients Balance water Well thentothe oxygen would be equal! But count the numbers of atoms again and you will see that now Hydrogen is unequal. We are not done yet…
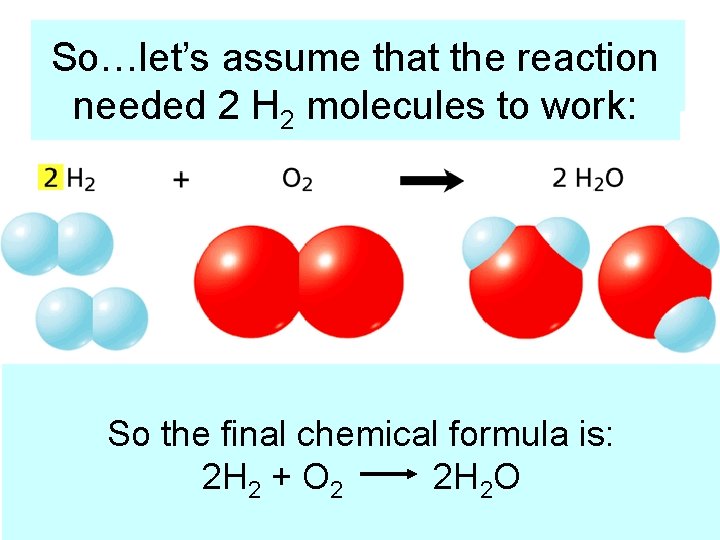
So…let’s assume that thetoreaction Step 3: Use Coefficients Balance needed 2 H 2 molecules to work: Count the atoms one last time and you can see that it! chemical The number of Hydrogen So we thedid final formula is: and Oxygen 2 H on 2 + both are O 2 sides 2 H 2 Oequal! What goes in = what came out
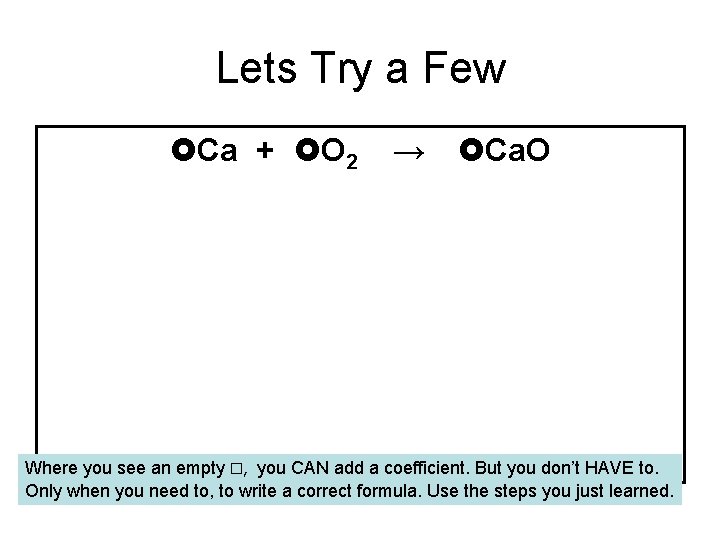
Lets Try a Few Ca + O 2 → Ca. O Where you see an empty □, you CAN add a coefficient. But you don’t HAVE to. Only when you need to, to write a correct formula. Use the steps you just learned.
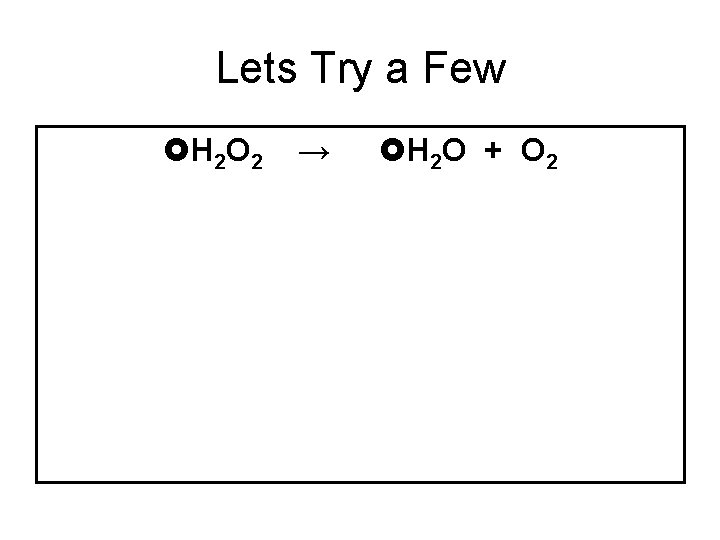
Lets Try a Few H 2 O 2 → H 2 O + O 2
- Slides: 13