CHEMICAL FORMULAS AND CHEMICAL COMPOUNDS MISS MOORE CHEMISTRY











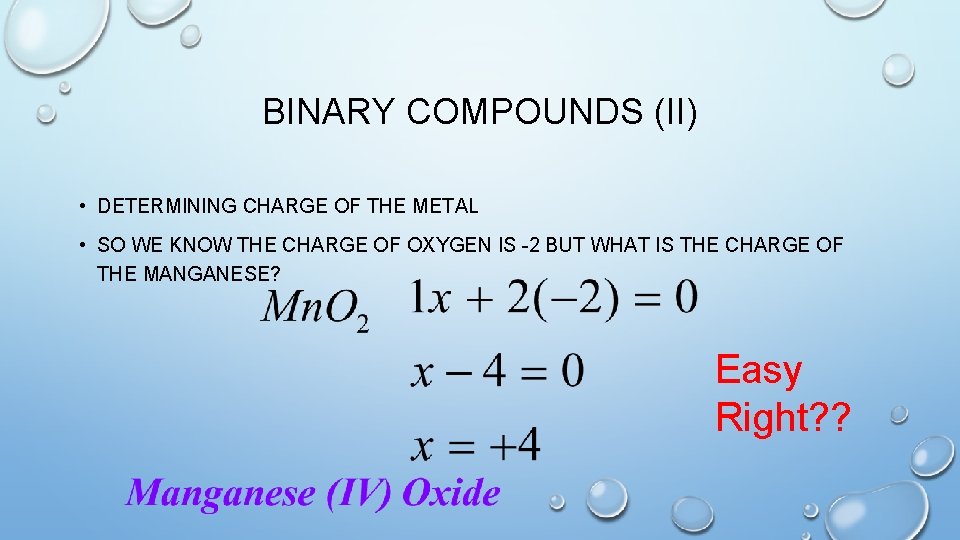













- Slides: 27

CHEMICAL FORMULAS AND CHEMICAL COMPOUNDS MISS MOORE CHEMISTRY

IONS • ION IS AN ATOM OR GROUP OF BONDED ATOMS THAT HAS A POSITIVE OR NEGATIVE CHARGE. • IONIZATION- ANY PROCESS THAT RESULTS IN THE FORMATION OF AN ION. • CATIONS- POSITIVELY CHARGED ION ALL IONS END IN IDE EX: BROMIDE • ANIONS- NEGATIVELY CHARGE IONS Cations Anions Na+ Cr 6+ Br- N -3 11 Protons 24 Protons 35 Protons 7 Protons 10 Electrons 18 Electrons 36 Electrons 10 Electrons

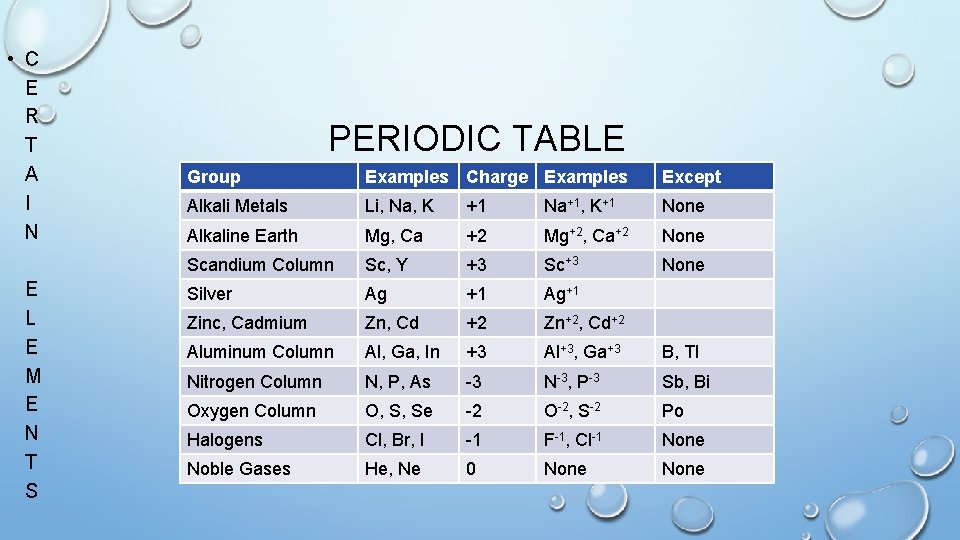
• C E R T A I N E L E M E N T S PERIODIC TABLE Group Examples Charge Examples Except Alkali Metals Li, Na, K +1 Na+1, K+1 None Alkaline Earth Mg, Ca +2 Mg+2, Ca+2 None Scandium Column Sc, Y +3 Sc+3 None Silver Ag +1 Ag+1 Zinc, Cadmium Zn, Cd +2 Zn+2, Cd+2 Aluminum Column Al, Ga, In +3 Al+3, Ga+3 B, Tl Nitrogen Column N, P, As -3 N-3, P-3 Sb, Bi Oxygen Column O, S, Se -2 O-2, S-2 Po Halogens Cl, Br, I -1 F-1, Cl-1 None Noble Gases He, Ne 0 None


IONIC COMPOUNDS • CONSIST OF ONE METAL AND ONE NON-METAL( ALWAYS) Metal KF Non- Metal • COMPOUNDS THAT ARE MADE UP OF IONS • THE COMPOUND MUST BE NEUTRAL: MEANING THAT THE CHARGES MUST BALANCE OUT TO ZERO.

IONIC COMPOUND • WHAT COMPOUND WOULD BE PRODUCED BY THE COMBINATION OF CALCIUM AND IODINE? • WHAT COMPOUND WOULD BE PRODUCED BY THE COMBINATION OF ALUMINUM AND SULFUR?

BINARY COMPOUNDS(TYPE 1) • FORMULAS- TELL THE NUMBER AND KINDS OF ATOMS IN A COMPOUND • METALS WITH A FIXED CHARGED ARE CONSIDERED TYPE 1 • EX: ALKALI METALS • ALKALINE EARTH METALS • SILVER, ZINC AND CADMIUM • ALUMINUM, GALLIUM, INDIUM

BINARY COMPOUNDS (TYPE II) • THESE METALS DO NOT HAVE A FIXED CHARGE • EX: TRANSITION METALS. - OR ALL OTHER METALS - * ALWAYS WRITE THE METAL FIRST AND THEN THE NON-METAL.

NAMING BINARY COMPOUNDS(TYPE 1) • 1) ALWAYS NAME THE CATION FIRST( POSITIVELY CHARGED) • NAME OF THE ELEMENT • 2) ANION REPLACES THE END OF THE ELEMENT WITH IDE • EX: CHLORINE= CHLORIDE • EX: SULFUR= SULFIDE • 3) COMBINE CATION AND ANION Na. Cl- Sodium Chloride K 2 O- Potassium Oxide

BINARY COMPOUNDS (TYPE I) Al 2 O 3 Na 3 N

BINARY COMPOUNDS(TYPE I) • LITHIUM NITRIDE • CALCIUM OXIDE • ALUMINUM SULFIDE

NAMING BINARY COMPOUNDS TYPE (II) • FOLLOW THE SAME RULES FOR TYPE 1 • EXCEPT THE METAL CHARGE MUST BE IDENTIFIED AND THEREFORE NAMED USING ROMAN NUMERALS. • COPPER(I)SULFIDE • IRON(II)CHLORIDE • PROBLEM: WHAT IF WE DON’T KNOW THE CHARGE OF THE METAL • ALGEBRA!!!!!!!
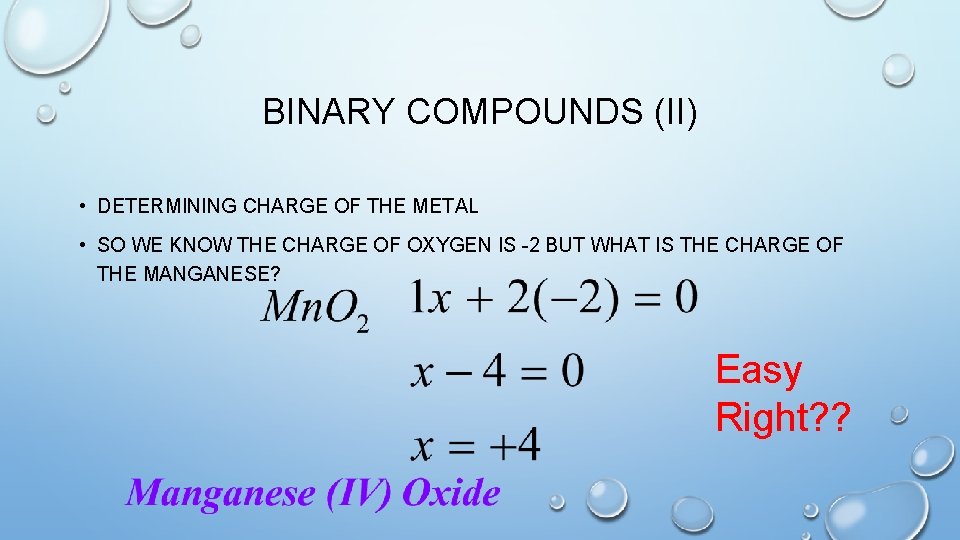
BINARY COMPOUNDS (II) • DETERMINING CHARGE OF THE METAL • SO WE KNOW THE CHARGE OF OXYGEN IS -2 BUT WHAT IS THE CHARGE OF THE MANGANESE? Easy Right? ?

PRACTICE MAKES PERFECT! Fe 2 O 3 Pb. O Ti. S 2

COVALENT COMPOUNDS • OCCURS WITH THE BONDING OF TWO NONMETALS • COMPOUNDS ARE FOUND BY SHARING ELECTRONS • NO CHARGES ARE INVOLVED (YAY NO MATH) • MEMORIZE- WE USE PREFIXES NOW Prefix # of Atoms Mono- 1 Penta- 5 Di- 2 Hexa- 6 Tri- 3 Hepta- 7 Tetra- 4 Octa- 8

RULES FOR NAMING COVALENT COMPOUNDS • 1) IDENTIFY THAT THE COMPOUND CONSISTS OF TWO NON-METALS • 2) WRITE THE NAMES OF THE ELEMENTS CHANGING THE ENDING TO IDE • 3) IF THERE IS MORE THAN ONE ATOM YOU MUST USE A PREFIX • WE DO NOT USE MONO FOR THE FIRST ATOM • EX: CO 2 • CARBONDIOXIDE

PRACTICE • CO- Carbon Monoxide • NO 2 Nitrogen Dioxide • PCL 3 Phosphorus Trichloride • SF 6 Sulfur Hexafluoride

COVALENT CONT…. • WHEN CHANGING THE ENDING TO IDE AVOID DOUBLE VOWELS SUCH AS : AO, OO, ETC • EX: SULFUR TRIOXIDE SO 3 N 2 O 4 • DINITROGEN TETROXIDE • SOME COMPOUNDS HAVE COMMON NAMES SUCH AS : • H 2 O – WATER • NH 3 – AMMONIA • CH 4 – METHANE (NATURAL GAS)

REVIEW IONIC AND COVLALENT Diboron Trioxide • B 2 O 3 Ni. Cl 2 Nickel(II)Chloride Gallium Nitride Ga. N

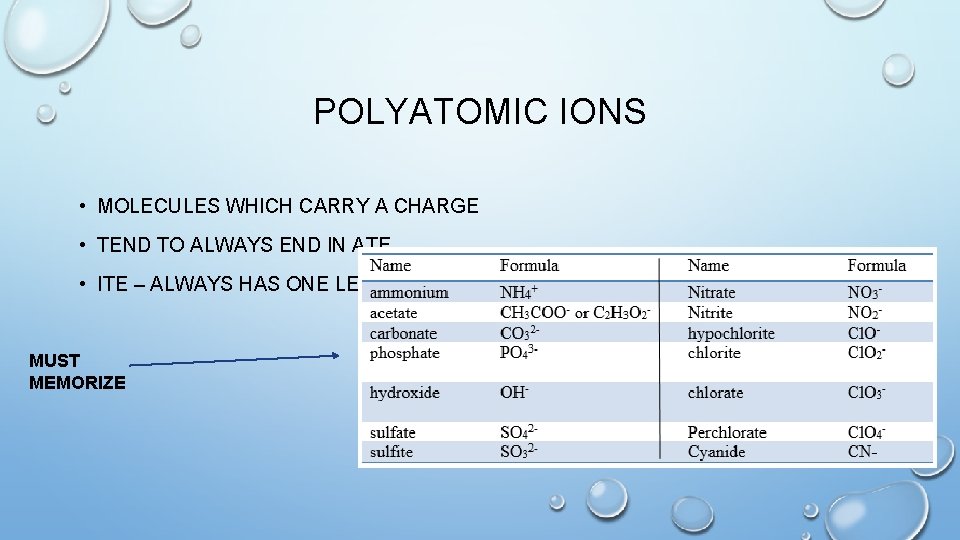
POLYATOMIC IONS • MOLECULES WHICH CARRY A CHARGE • TEND TO ALWAYS END IN ATE • ITE – ALWAYS HAS ONE LESS MUST MEMORIZE


(WE HAVE TO BALANCE THE CHARGES ON POLYATOMIC IONS JUST LIKE IONIC COMPOUNDS) P O L Y A T O Na. NO 3 Sodium Nitrate Al 2(SO 4)3 Aluminum Sulfate Fe 2(CO 3)3 Iron (III) Carbonate

ACIDS • ACID- ANY SUBSTANCE THAT PRODUCES H+ IN WATER • TENDS TO BE SOUR • WRITTEN WITH H AT THE BEGINNING OF THE FORMULA • TEND TO REACT WITH METALS

ACIDS NOT CONTAINING OXYGEN If the formula does not contain oxygen Begin the name with Hydro. Change the end of the anion to –ic End with the word “Acid” HCl(aq) HCN(aq) H 2 S(aq) Hydro. Chloric Acid Hydro. Cyanic Acid Hydro. Sulfuric Acid

ACID CONTAINS Formula will always contain a polyatomic Ion Identify the Polyatomic ion by removing H H 2 SO 4 has SO 4 (Sulfate) Drop the end of the ion -ate becomes –ic -ite becomes –ous Contains Oxygen: NO HYDRO! HCl. O 3 Chloric Acid HNO 2 Nitrous Acid

PRACTICE •

Properties of Ionic Compounds • A N I O N I C B O N D