Chemical Equilibrium What is equilibrium Expressions for equilibrium

Chemical Equilibrium • • What is equilibrium? Expressions for equilibrium constants, Keq; Calculating Keq using equilibrium concentrations; Calculating equilibrium concentrations using initial concentration and Keq value;

What is Equilibrium?

This is not Equilibrium?

Chemical Equilibrium in Nature: (The formation of stalagmites and Stalactites)

Chemical Equilibrium • Consider the following reactions: Ca. CO 3(s) + CO 2(aq) + H 2 O(l) Ca 2+(aq) + 2 HCO 3 -(aq). . (1) and Ca 2+(aq) + 2 HCO 3 -(aq) Ca. CO 3(s) + CO 2(aq) + H 2 O(l). . (2) Reaction (2) is the reverse of reaction (1). At equilibrium the two opposing reactions occur at the same rate. Concentrations of chemical species do not change once equilibrium is established.

Expression for Equilibrium Constant Consider the following equilibrium system: a. A + b. B ⇄ c. C + d. D Keq = • The numerical value of Keq is calculated using the concentrations of reactants and products that exist at equilibrium.

Expressions for Equilibrium Constants • Examples: N 2(g) + 3 H 2(g) ⇄ 2 NH 3(g); Keq = PCl 5(g) ⇄ PCl 3(g) + Cl 2(g); Keq = CH 4(g) + H 2 O (g) ⇄ CO(g) + 3 H 2(g); Keq =

Calculating Equilibrium Constant • Example-1: 0. 200 mole of H 2 gas and 0. 200 mole of I 2 vapor are introduced into a 1. 00 -liter sealed flask. The mixture is heated to a certain temperature and the following reaction occurs until equilibrium is established. H 2(g) + I 2(g) ⇄ 2 HI(g) At equilibrium, the mixture is found to contain 0. 316 mole of HI. (a) What are the concentrations of H 2, I 2 and HI at equilibrium? (b) Calculate the equilibrium constant Keq.
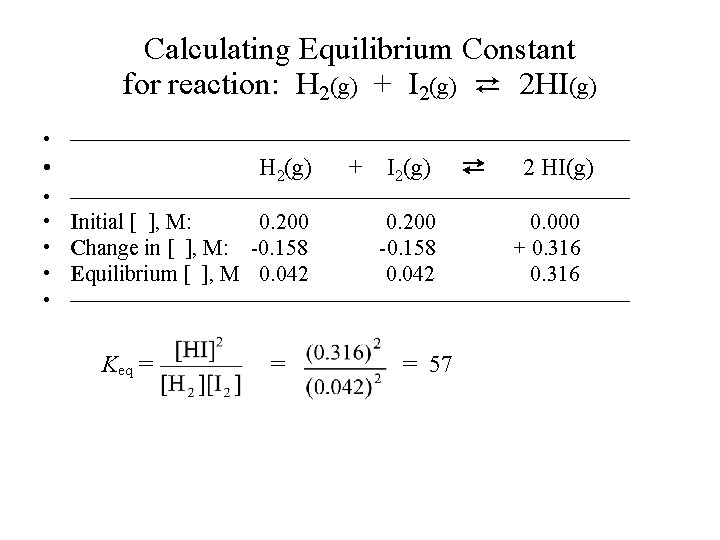
Calculating Equilibrium Constant for reaction: H 2(g) + I 2(g) ⇄ 2 HI(g) • —————————————— • H 2(g) + I 2(g) ⇄ 2 HI(g) • —————————————— • Initial [ ], M: 0. 200 • Change in [ ], M: -0. 158 • Equilibrium [ ], M 0. 042 0. 200 -0. 158 0. 042 0. 000 + 0. 316 • —————————————— Keq = = = 57

Calculating Equilibrium Constant • Example-2: 0. 500 mole of HI is introduced into a 1. 00 liter sealed flask and heated to a certain temperature. Under this condition HI decomposes to produce H 2 and I 2 until an equilibrium is established. An analysis of the equilibrium mixture shows that 0. 105 mole of HI has decomposed. Calculate the equilibrium concentrations of H 2, I 2 and HI, and the equilibrium constant Keq for the following reaction: H 2(g) + I 2(g) ⇄ 2 HI(g),
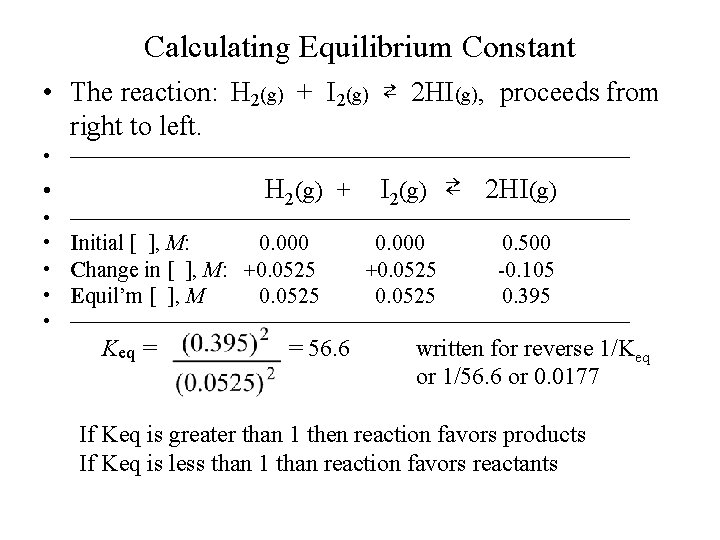
Calculating Equilibrium Constant • The reaction: H 2(g) + I 2(g) ⇄ 2 HI(g), proceeds from right to left. • —————————————— H 2(g) + • I 2(g) ⇄ 2 HI(g) • —————————————— • Initial [ ], M: 0. 000 • Change in [ ], M: +0. 0525 • Equil’m [ ], M 0. 0525 0. 000 +0. 0525 0. 500 -0. 105 0. 395 • —————————————— Keq = = 56. 6 written for reverse 1/Keq or 1/56. 6 or 0. 0177 If Keq is greater than 1 then reaction favors products If Keq is less than 1 than reaction favors reactants

Expression and Value of Equilibrium Constant for a Reaction • The expression for Keq depends on the equation; • The value of Keq applies to that equation; it does not depend on how the reaction occurs; • Concentrations used to calculate the value of Keq are those measured at equilibrium.

Relationships between chemical equations and the expressions of equilibrium constants • The expression of equilibrium constant depends on how the equilibrium equation is written. For example, for the following equilibrium: • H 2(g) + I 2(g) ⇄ 2 HI(g); • For the reverse reaction: • 2 HI(g) ⇄ H 2(g) + I 2(g);

Solubility Eqilibrium Pb. Cl 2(s) ⇄ Pb 2+(aq) + 2 Cl-(aq); 2+ - 2 Ksp = [Pb ][Cl ] ______ 1 Pb. Cl 2 is a solid so denominator is 1 (Ksp is called solubility product)

Applications of Equilibrium Constant For any system or reaction: 1. Knowing the equilibrium constant, we can predict whether or not a reaction mixture is at equilibrium, and we can predict the direction of net reaction. • Qc = Keq equilibrium (no net reaction) • Qc < Keq a net forward reaction; • Qc > Keq a net reverse reaction 2. The value of K tells us whether a reaction favors the products or the reactants.

Equilibrium constant is used to predict the direction of net reaction • For a reaction of known Keq value, the direction of net reaction can be predicted by calculating the reaction quotient, Qc. • Qc is called the reaction quotient, where for a reaction such as: • a. A + b. B ⇄ c. C + d. D; • Qc has the same expression as Keq , but • Qc is calculated using concentrations that are not necessarily at equilibrium.

What does the reaction quotient tell us? If Qc = Keq, the reaction is at equilibrium; If Qc < Keq, the reaction is not at equilibrium and there’s a net forward reaction; If Qc > Keq, the reaction is not at equilibrium and there’s a net reaction in the opposite direction.

Why is Equilibrium Constant Important? • Knowing Keq and the initial concentrations, we can determine the concentrations of components at equilibrium. • We can also determine what factors can be changed to force a reaction in either direction to optimize amounts at equilibrium.

Equilibrium Exercise • For the reaction: CO(g) + 2 H 2(g) ⇄ CH 3 OH(g), Keq = 14. 5 at 500 K. • Predict whether a mixture that contains 1. 50 mol of H 2, 1. 00 mol of CO, and 0. 50 mol of CH 3 OH in a 10. 0 -L vessel at 500 Keq is at equilibrium. • If not, indicate the direction in which the net reaction will occur to reach equilibrium.

Answer Qc= (CH 3 OH) (CO) (H 2)2 = (0. 5 mol/10 L) (1. 0 mol/10 L)(1. 5 moles/10 L)2 = (0. 05 M) = (0. 10 M)(0. 15 M)2 22. 2 > 14. 5 so reaction is driven to the left

Calculating equilibrium concentrations using initial concentrations and value of Keq • Consider the reaction: • H 2(g) + I 2(g) ⇄ 2 HI(g), • where Keq = 55. 6 at 425 o. C. • If [H 2]0 = [I 2]0 = 0. 1000 M, and [HI]0 = 0. 0 M, what are their concentrations at equilibrium?
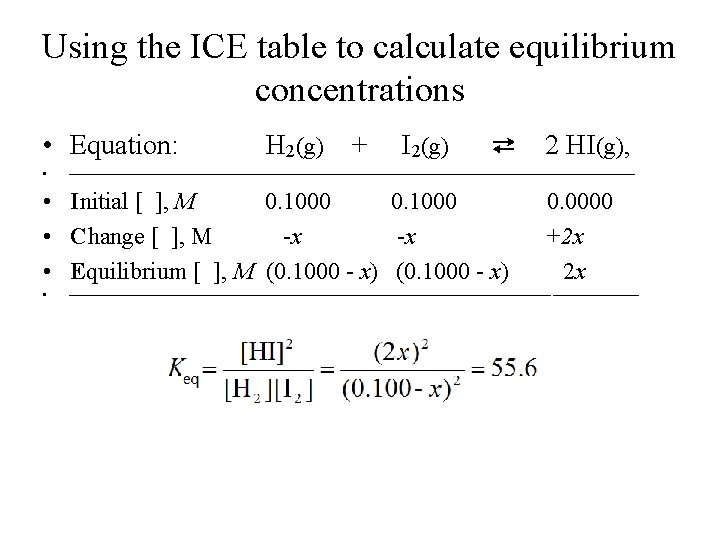
Using the ICE table to calculate equilibrium concentrations • Equation: • H 2(g) + I 2(g) ⇄ • Initial [ ], M 0. 1000 • Change [ ], M -x -x • Equilibrium [ ], M (0. 1000 - x) • 2 HI(g), 0. 0000 +2 x 2 x

Calculation of equilibrium concentrations

Le Châtelier’s Principle • The Le Châtelier's principle states that: when factors that influence an equilibrium are altered, the equilibrium will shift to a new position that tends to minimize those changes. • Factors that influence equilibrium: Concentration, temperature, and partial pressure (for gaseous)

The Effect of Changes in Concentration • Consider the reaction: N 2(g) + 3 H 2(g) ⇄ 2 NH 3(g); • If [N 2] and/or [H 2] is increased, Qc < Kc • a net forward reaction will occur to reach new equilibrium position. • If [NH 3] is increased, Qc > Kc, and a net reverse reaction will occur to come to new equilibrium position.

Effects of Pressure Change on Equilibrium • If the volume of a gas mixture is compressed, the overall gas pressure will increase. In which direction the equilibrium will shift in either direction depends on the reaction stoichiometry. • However, there will be no effect to equilibrium if the total gas pressure is increased by adding an inert gas that is not part of the equilibrium system.

Reactions that shift right when pressure increases and shift left when pressure decreases Consider the reaction: 2 SO 2(g) + O 2(g) ⇄ 2 SO 3(g), 1. The total moles of gas decreases as reaction proceeds in the forward direction. 2. If pressure is increased by decreasing the volume (compression), a forward reaction occurs to reduce the stress. 3. Reactions that result in fewer moles of gas favor high pressure conditions.

Reaction that shifts left when pressure increases, but shifts right when pressure decreases Consider the reaction: PCl 5(g) ⇄ PCl 3(g) + Cl 2(g); 1. Forward reaction results in more gas molecules. 2. Pressure increases as reaction proceeds towards equilibrium. 3. If mixture is compressed, pressure increases, and reverse reaction occurs to reduce pressure; 4. If volume expands and pressure drops, forward reaction occurs to compensate. 5. This type of reactions favors low pressure condition

Reactions not affected by pressure changes Consider the following reactions: 1. CO(g) + H 2 O(g) ⇄ CO 2(g) + H 2(g); 2. H 2(g) + Cl 2(g) ⇄ 2 HCl(g); 1. Reactions have same number of gas molecules in reactants and products. 2. Reducing or increasing the volume will cause equal effect on both sides – no net reaction will occur. 3. Equilibrium is not affected by change in pressure.

The Effect Temperature on Equilibrium • Consider the following exothermic reaction: N 2(g) + 3 H 2(g) ⇄ 2 NH 3(g); DHo = -92 k. J, • The forward reaction produces heat => heat is a product. • When heat is added to increase temperature, reverse reaction will take place to absorb the heat; • If heat is removed to reduce temperature, a net forward reaction will occur to produce heat. • Exothermic reactions favor low temperature conditions.

The Effect Temperature on Equilibrium Consider the following endothermic reaction: CH 4(g) + H 2 O(g) ⇄ CO(g) + 3 H 2(g), DHo = 205 k. J 1. 2. 3. 4. Endothermic reaction absorbs heat is a reactant; If heat is added to increasing the temperature, it will cause a net forward reaction. If heat is removed to reduce the temperature, it will cause a net reverse reaction. Endothermic reactions favor high temperature condition.

Chemical Equilibria in Industrial Processes The production of ammonia by the Haber-Bosch process: N 2(g) + 3 H 2(g) ⇄ 2 NH 3(g); DH = -92 k. J • This reaction is exothermic and very slow at low temperature. • Increasing the temperature will increase reaction rate, but will lower the yield. • An optimum condition is achieved at moderate temperature of 250 to 300 o. C with catalyst added to increase the reaction rate. • Increasing the pressure will favor product formation. • Reaction favors low temperature and high pressure conditions.

Chemical Equilibria in Industrial Processes The production of hydrogen gas: • Reaction: CH 4(g) + H 2 O(g) ⇄ CO(g) + 3 H 2(g); • This reaction is endothermic with DH = 206 k. J • Increasing the reaction temperature will increase both the rate and the yield. • This reaction favors high temperature and low pressure conditions.

- Slides: 34