Chemical Equilibrium What is equilibrium Chemical reaction stops

Chemical Equilibrium

What is equilibrium? • Chemical reaction stops BEFORE the limiting reactant is consumed. • All of the reactants are not COMPLETELY converted to products. o Amount of reactant converting to product = converting back to reactant amount of product • Represented in a chemical reaction by arrows pointing in BOTH directions. ( ) • Reactant and product concentrations are STABLE

• Ex. H 2 + O 2 H 2 O
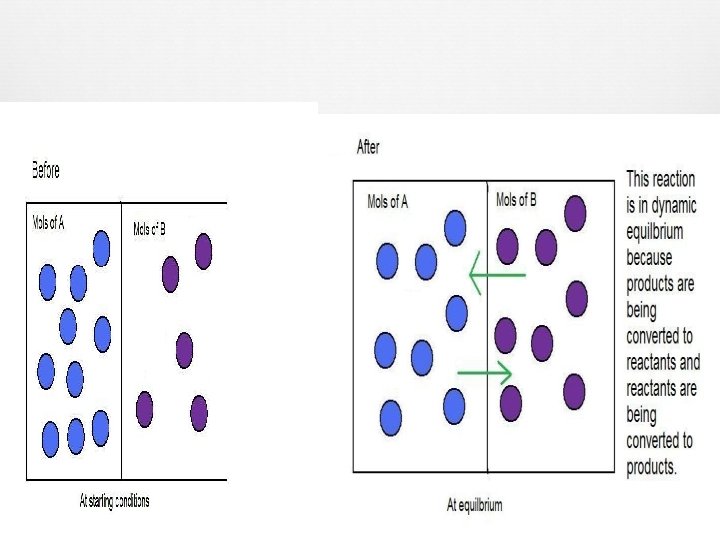

Chemical Reactions at Equilibrium Big Picture/Forest Reaction looks like it is STOPPED. Ex. Helicopter view of traffic jam. Small Scale/Trees Reaction is moving, “dynamic” balance between forward/reverse reactions. Ex. Road view of traffic jam.

Chemical Reactions at Equilibrium (cont. ) • Rate of forward = Rate of backward • Equilibrium constant expression: o o Ratio of the reactant concentration and product concentration. Concentration MUST be in molarity (M) o A + B o KC = [products] [reactants] C + D [C][D] [A][B]

Equilibrium Constant (KC) constant value for each chemical equation at equilibrium numerical value of ratio The equilibrium constant allows us to …. 1) Predict the direction in which a reaction mixture will proceed to achieve equilibrium. 2) Calculate the concentrations of reactants and products once equilibrium has been reached.

a. A + b. B ↔ c. C + d. D at equilibrium at a given temperature K varies with temperature and is UNITLESS.

Example (a): Write the equilibrium expression for… PCl 5(g) PCl 3(g) + Cl 2(g) NOTE: [ ] denotes concentration.

Example (b): Write the equilibrium expression for… 4 NH 3(g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(g)

Now you try…… 1) 2 NO 2 N 2 O 4 2) 2 SO 3 2 SO 2 3) N 2 + + O 2 3 H 2 2 NH 3

Ex 1: A mixture was found to contain the following concentrations: 0. 172 M PCl 3, 0. 086 M Cl 2 and 0. 028 M PCl 5. Write the equilibrium expression and calculate K. PCl 5 ↔ PCl 3 + Cl 2

How do we use a value like k=0. 53 to determine a reaction’s direction? ? ? • When k >> 1, PRODUCTS are favored. • When k << 1, REACTANTS are favored. • Think about this statement using ………… o KC = [products] [reactants]

Equilibrium Worksheet
- Slides: 15