Chemical Equilibrium Standard 9 Standard 9 Standard 9

Chemical Equilibrium Standard 9

Standard 9 • Standard 9 a – I will know how to use Le. Chatelier’s principle to predict the effect of changes in concentration, temperature, and pressure (volume). • Standard 9 b – I will know equilibrium is established when forward and reverse reaction rates are equal.

Standard 9 a Symbols for Reversible Reactions Symbols for one way reactions
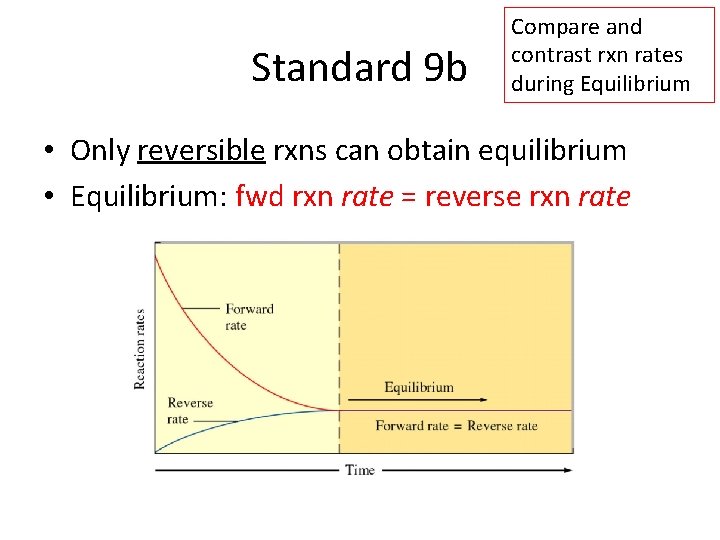
Standard 9 b Compare and contrast rxn rates during Equilibrium • Only reversible rxns can obtain equilibrium • Equilibrium: fwd rxn rate = reverse rxn rate
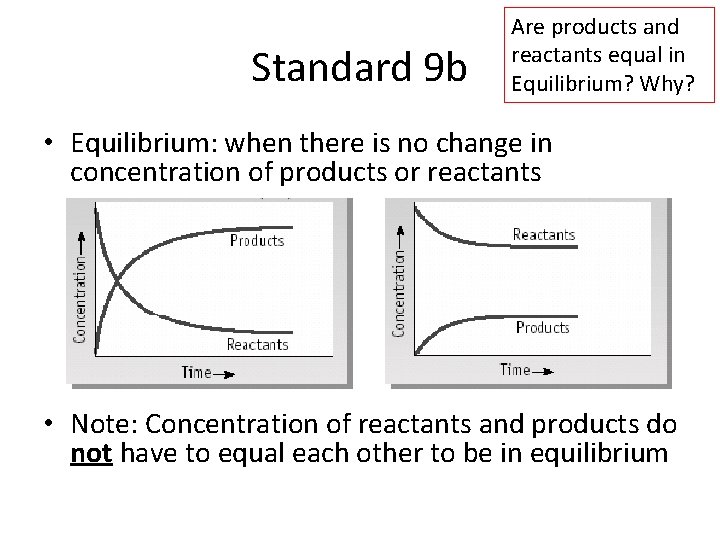
Standard 9 b Are products and reactants equal in Equilibrium? Why? • Equilibrium: when there is no change in concentration of products or reactants • Note: Concentration of reactants and products do not have to equal each other to be in equilibrium

Standard 9 a Define Le. Chatelier’s Principle • Le. Chatelier's Principle: if a system at equilibrium is subjected to a stress, then the equilibrium will shift in a direction to relieve that stress. • Stress: – Concentration – Temperature – Pressure (Volume)
![Standard 9 a Concentration • If more [H 2] is added…. How does Concentration Standard 9 a Concentration • If more [H 2] is added…. How does Concentration](http://slidetodoc.com/presentation_image_h2/7d9fda09e7e35f08f12200258d9b4d0b/image-7.jpg)
Standard 9 a Concentration • If more [H 2] is added…. How does Concentration affect equilibrium?
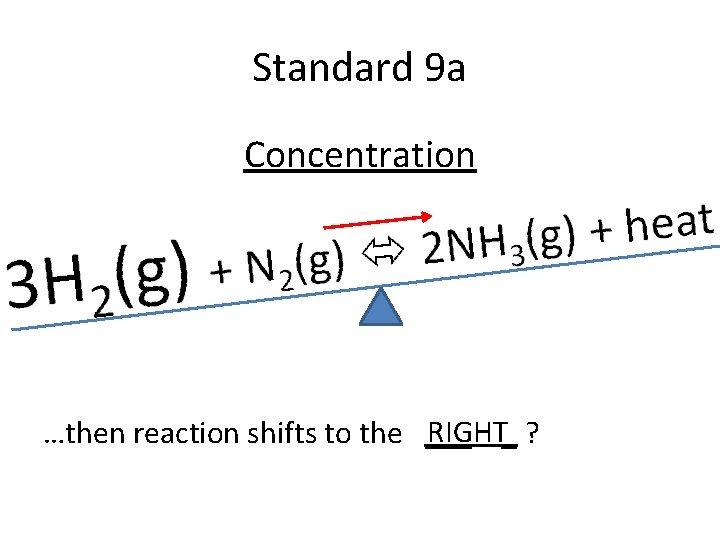
Standard 9 a Concentration RIGHT_ ? …then reaction shifts to the ___
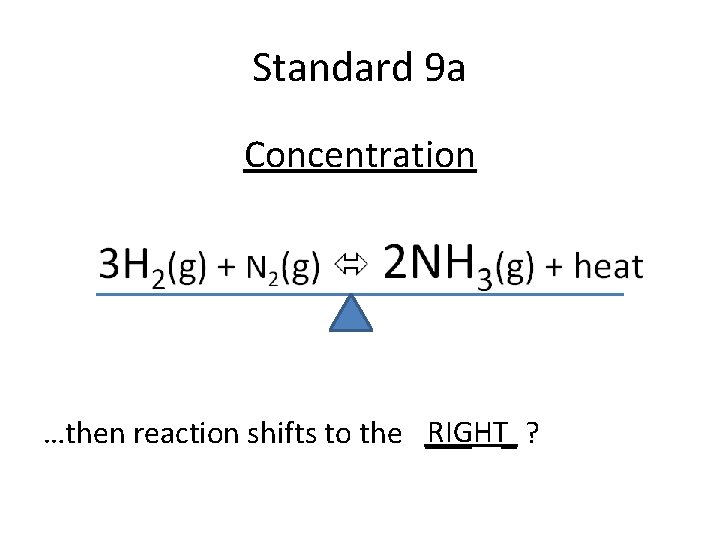
Standard 9 a Concentration RIGHT_ ? …then reaction shifts to the ___
![Standard 9 a Concentration If [H 2] is removed…. Standard 9 a Concentration If [H 2] is removed….](http://slidetodoc.com/presentation_image_h2/7d9fda09e7e35f08f12200258d9b4d0b/image-10.jpg)
Standard 9 a Concentration If [H 2] is removed….
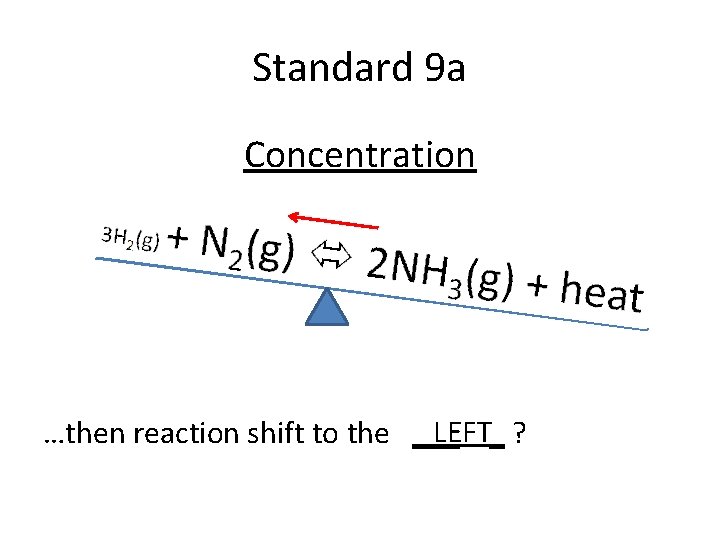
Standard 9 a Concentration LEFT_ ? …then reaction shift to the ___
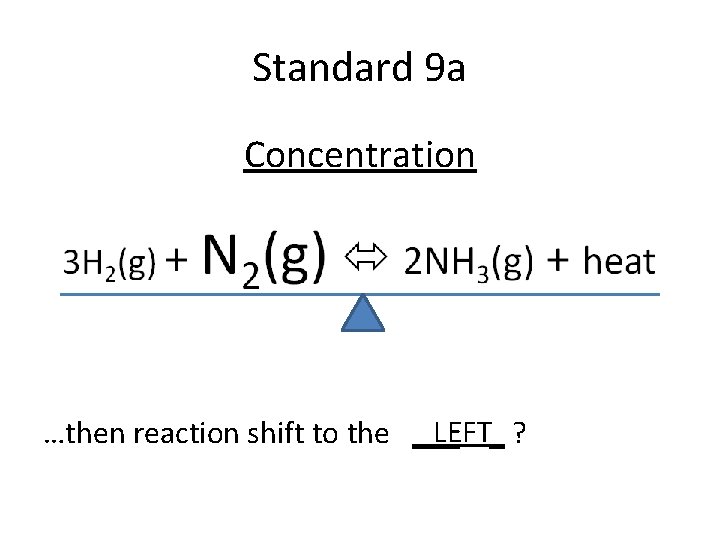
Standard 9 a Concentration LEFT_ ? …then reaction shift to the ___

Standard 9 a Temperature Endothermic or Exothermic? How does Temperature affect equilibrium?

Standard 9 a Temperature If Temperature is increased…. .
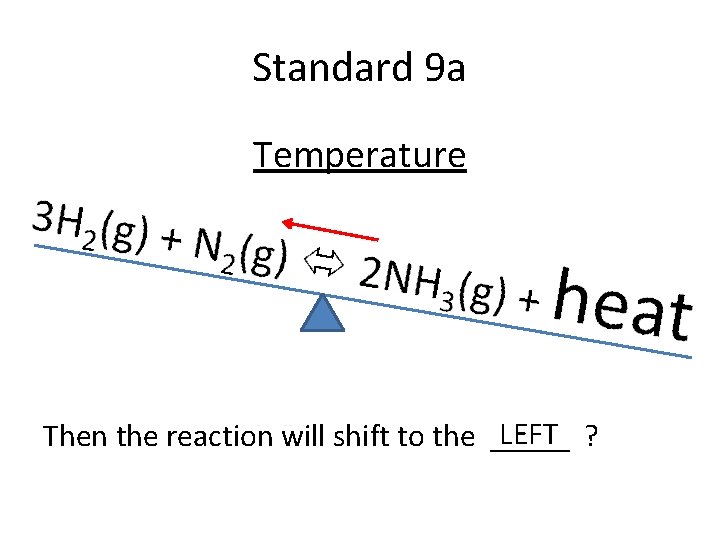
Standard 9 a Temperature LEFT ? Then the reaction will shift to the _____
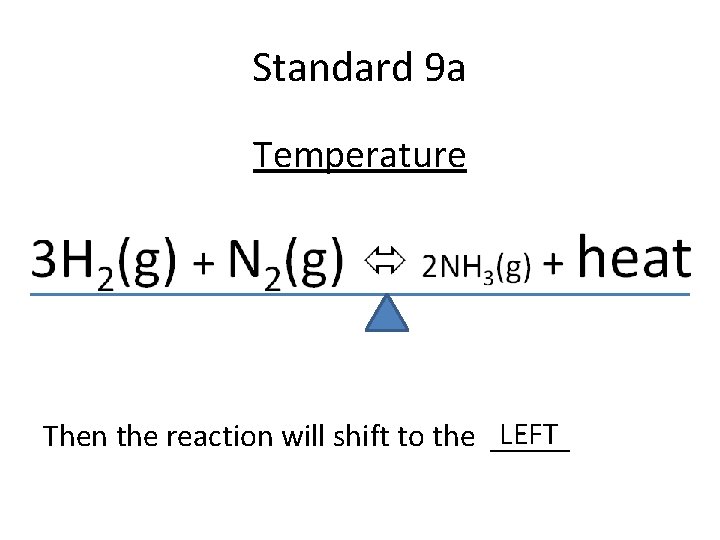
Standard 9 a Temperature LEFT Then the reaction will shift to the _____

Standard 9 a Temperature If Temperature is decreased…. . …………then the reaction will shift to the RIGHT _____

Standard 9 a Temperature Endothermic or Exothermic? Heat + H 2 O(s) H 2 O(l) If Temperature is increased…. . …………then the reaction will shift to the RIGHT _____

Standard 9 a How does Pressure/Volume affect equilibrium? Pressure/Volume 3 H 2(g) + N 2(g) 2 NH 3(g) + heat Incr. Pressure/Decr. Vol. shifts the equilibrium. RIGHT ______ ?

Standard 9 a Pressure/Volume 3 H 2(g) + N 2(g) 2 NH 3(g) + heat Decr. Pressure/Incr. Vol. shifts the equilibrium ______ LEFT ?

Standard 9 a • Decrease in Pressure/Increase of Volume shifts equilibrium which way? ? ?

Summary • What is equilibrium? • Are products and reactants equal at equilibrium? • How do the following change equilibrium: – Concentration? – Temperature? – Pressure?
- Slides: 22