CHEMICAL EQUILIBRIUM Some chemical reactions are reversible A

CHEMICAL EQUILIBRIUM: Some chemical reactions are reversible. A reversible reaction is one in which the conversion of reactants to products & the conversion of products to reactants occur at the same time. ? Ex. Forward 2 SO 2(g) + O 2(g) → 2 SO 3(g) ∆H < 0 Reverse Exothermic ∆H > 0 Endothermic The two equations can be combined in one using a double arrow:

CHEMICAL EQUILIBRIUM: Reversible Reaction: 2 SO 2(g) + O 2(g) 2 SO 3(g) (2 mol/L) (3 mol/L) ΔH < 0 (4 mol/L) Forward V 1 (Rate of reaction) Reverse V 2 (Rate of reaction) Equilibrium: V 1=V 2 The concentration of all substances remains constant.
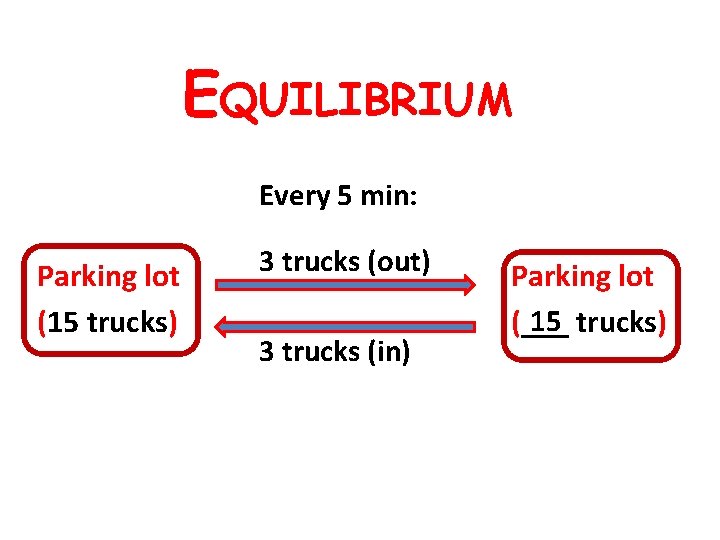
EQUILIBRIUM Every 5 min: Parking lot (15 trucks) 3 trucks (out) 3 trucks (in) Parking lot 15 trucks) (___

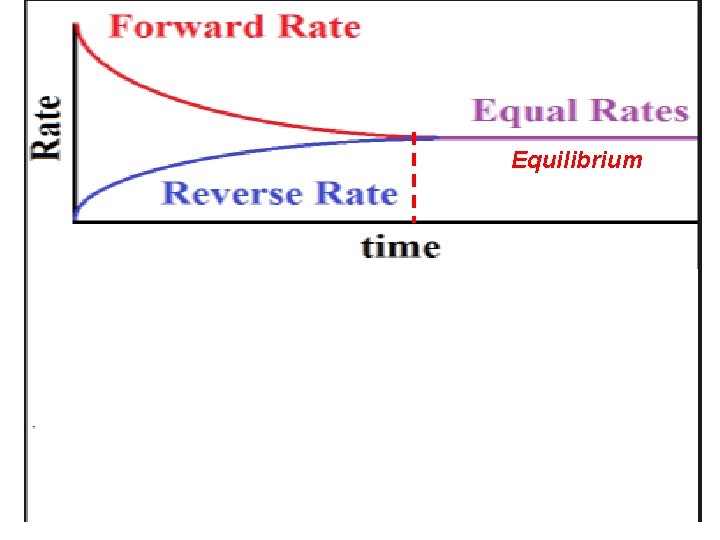
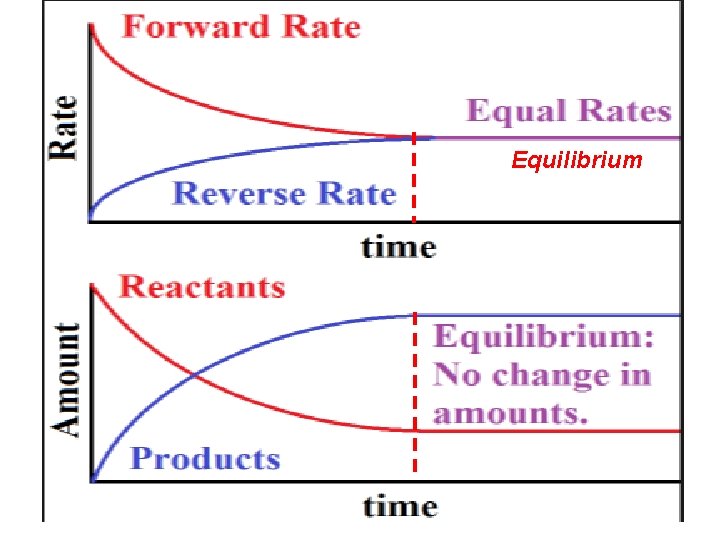
When the rates of the forward & reverse reactions are equal, the reaction has reached a state of balance called Chemical Equilibrium. V 1=V 2 In a Chemical Equilibrium, the forward & the reverse reactions continue, but their rates are equal, no net change occur in the concentration of the reaction components. Ex. Analyze the following graphs:
Equilibrium
Equilibrium
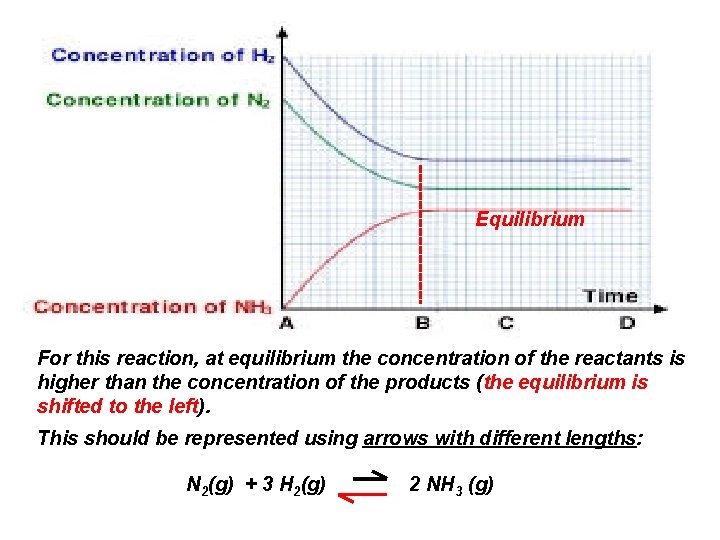
Equilibrium For this reaction, at equilibrium the concentration of the reactants is higher than the concentration of the products (the equilibrium is shifted to the left). This should be represented using arrows with different lengths: N 2(g) + 3 H 2(g) 2 NH 3 (g)

Factors Affecting equilibrium: Le Chatelier’s Principle: If a stress is applied to a system in dynamic equilibrium, the system changes in a way that relieves the stress. Stresses: • Changes in concentration (reactants or products). 2) Changes in temperature. 3) Changes in pressure (gases). Examples:

1) Concentration: Changing the concentration (amount) of any reactant or product disturbs the equilibrium and the system will adjust to minimize the effects of the change. Ex: N 2(g) + 3 H 2(g) 2 NH 3 (g) Adding N 2(g), increases its concentration, so the equilibrium shift to the right to minimize the effect of the change. As consequence of the increase of the [N 2] [H 2] (concentration of H 2) decreases [NH 3] (concentration of NH 3) increases
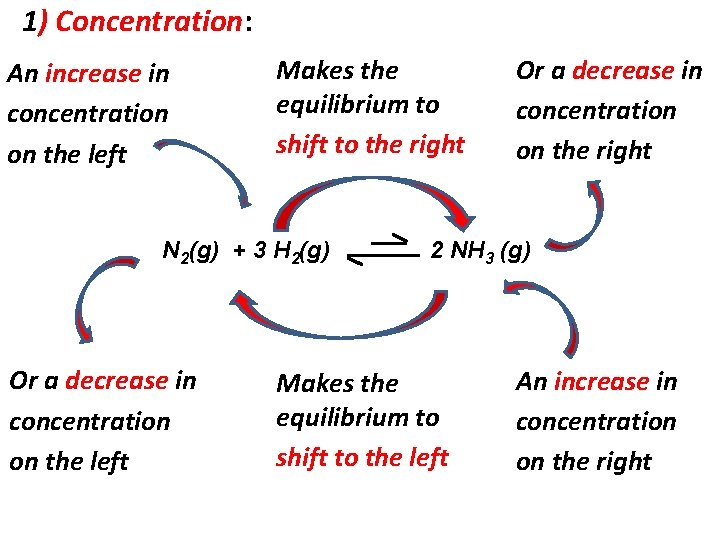
1) Concentration: An increase in concentration on the left Makes the equilibrium to shift to the right N 2(g) + 3 H 2(g) Or a decrease in concentration on the left Or a decrease in concentration on the right 2 NH 3 (g) Makes the equilibrium to shift to the left An increase in concentration on the right

2) Temperature: Increasing the temperature causes the equilibrium position to shift in the direction that absorbs heat to minimize the effects of the change. Ex: (∆H < 0) N 2(g) + 3 H 2(g) 2 NH 3 (g) + heat The equilibrium shift to the left (endothermic reaction) to minimize the effect of the change. As consequence of the increase in temperature [H 2] (concentration of H 2) increases [N 2] (concentration of N 2) increases [NH 3] (concentration of NH 3) decreases

2) Temperature: Increasing the temperature causes the equilibrium position to shift toward the endothermic reaction. (∆H < 0) N 2(g) + 3 H 2(g) 2 NH 3 (g) + heat Decreasing the temperature causes the equilibrium position to shift toward the exothermic reaction.

3) Pressure: Increasing the pressure (reducing the volume of the container) causes the equilibrium position to shift in the direction that reduce the number of moles to minimize the effects of the change. Ex: N 2(g) + 3 H 2(g) 2 NH 3 (g) + heat The equilibrium shift to the right (less number of molecules, less pressure) to minimize the effect of the change. As consequence of the increase in pressure [H 2] (concentration of H 2) decreases [N 2] (concentration of N 2) decreases [NH 3] (concentration of NH 3) increases
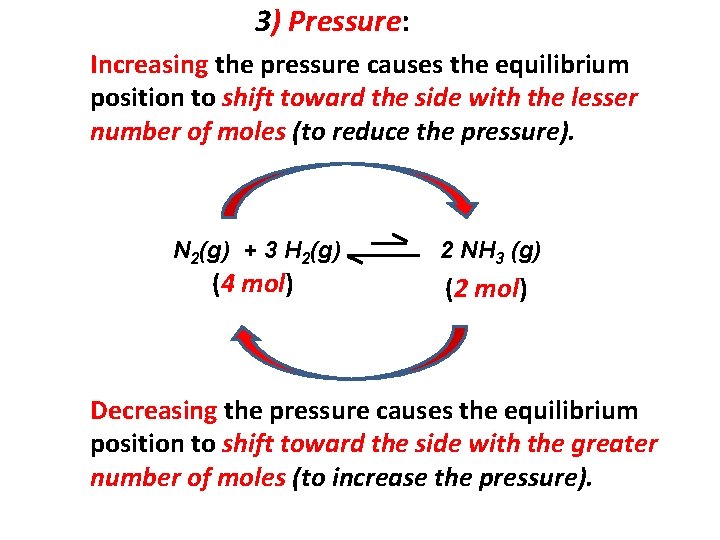
3) Pressure: Increasing the pressure causes the equilibrium position to shift toward the side with the lesser number of moles (to reduce the pressure). N 2(g) + 3 H 2(g) (4 mol) 2 NH 3 (g) (2 mol) Decreasing the pressure causes the equilibrium position to shift toward the side with the greater number of moles (to increase the pressure).

Catalysts? Do the catalysts affect the chemical equilibrium? Catalysts lower the activation energy for both, the forward reaction and the reverse reaction in the same way. Catalysts decrease the time it takes to establish the equilibrium, so catalysts don’t affect the chemical equilibrium.

Exercise (Practice):

Class work : W/S: Factors Affecting the Chemical Equilibrium. Home work: WS: Le Chatelier Principle (3) (Edmodo) Equations: 1 – 12: 5 CO(g) + I 2 O 5 (s) ⇄ I 2 (g) + 5 CO 2 (g) 13 -17: 4 NO(g) + 6 H 2 O (g) ⇄ 4 NH 3 (g) + 5 O 2 (g) ∆H = - 1 175 k. J ∆H = + 1 532 k. J
- Slides: 17