Chemical Equilibrium Reversible Reactions Both reactants and products

Chemical Equilibrium
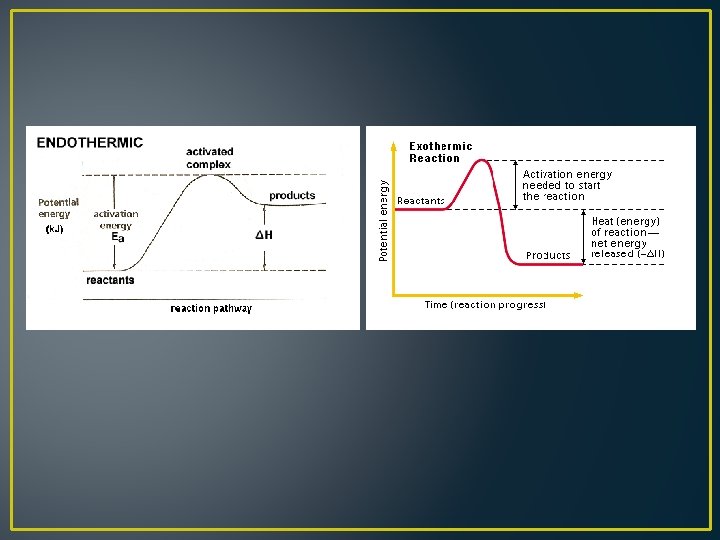
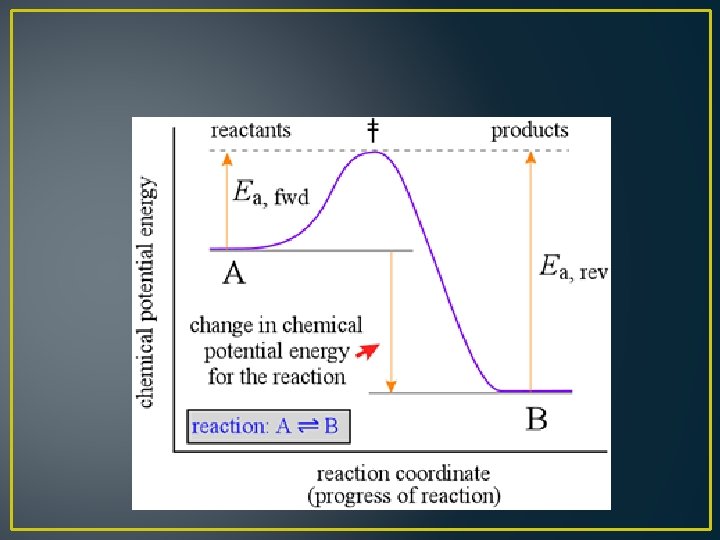

Reversible Reactions • Both reactants and products exist together as a mixture • The ratio in which reactants and products are present in the mixture depends on the relative speeds of the forward and reverse reactions • Changing the rate of either the forward or the reverse reaction changes the ratio of products and reactants in the reaction mixture

Equilibrium Tank • What is Equilibrium? • A chemical equilibrium exists when two opposing reactions occur simultaneously at the same rate, balancing each other
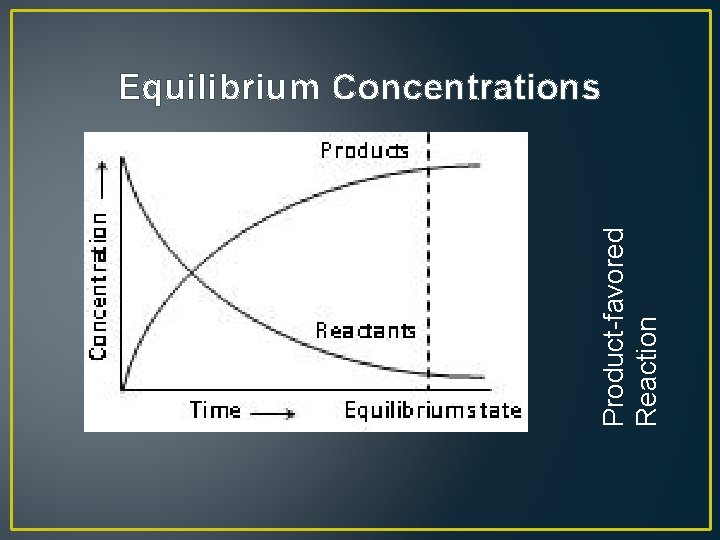
Product-favored Reaction Equilibrium Concentrations
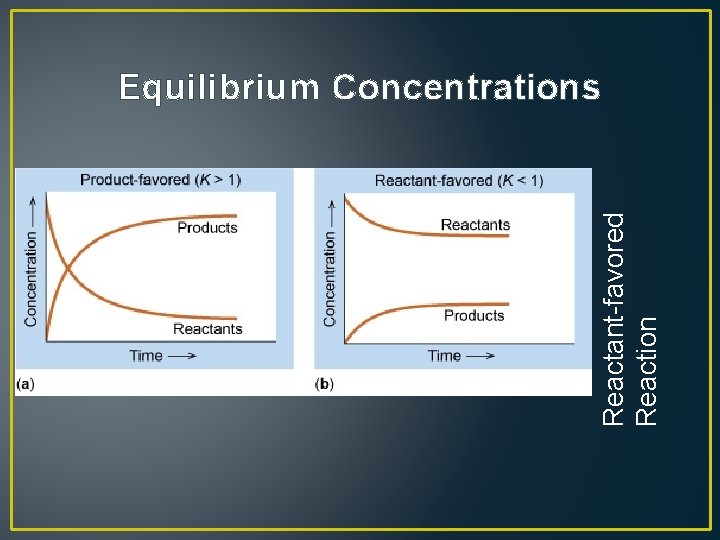
Reactant-favored Reaction Equilibrium Concentrations

• Reactants Products • If a reaction is product-favored, the concentration of the products is higher than that of the reactants – the equilibrium is said to lie to the right • If a reaction is reactant-favored, the concentration of the reactants is higher than that of the products – the equilibrium is said to lie to the left • Arrows can be used to indicate the favorable direction

Equilibrium Constants •

Equilibrium Constant Rules • The concentrations of pure solids and liquids are not included in equilibrium constants • ALL gases and aqueous substances are included

ICE CHARTS • Initial Concentration (M) • Change in Concentration • Concentration at Equilibrium

ICE CHARTS • Example: A mixture consisting initially of 3. 00 moles NH 3, 2. 00 moles of N 2, and 5. 00 moles of H 2, in a 5. 00 L container was heated to 900 K, and allowed to reach equilibrium. Determine the equilibrium concentration for each species present in the equilibrium mixture. 2 NH 3(g) N 2(g) + 3 H 2(g) Kc = 0. 0076 @ 900 K
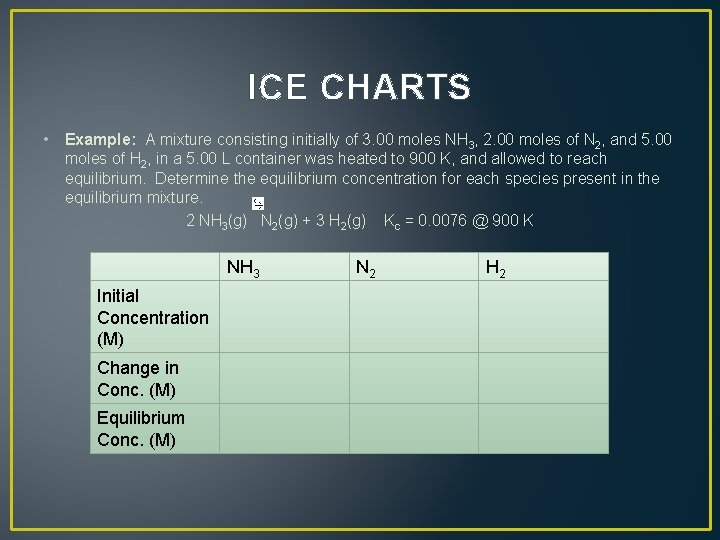
ICE CHARTS • Example: A mixture consisting initially of 3. 00 moles NH 3, 2. 00 moles of N 2, and 5. 00 moles of H 2, in a 5. 00 L container was heated to 900 K, and allowed to reach equilibrium. Determine the equilibrium concentration for each species present in the equilibrium mixture. 2 NH 3(g) N 2(g) + 3 H 2(g) Kc = 0. 0076 @ 900 K Initial Concentration (M) Change in Conc. (M) Equilibrium Conc. (M) NH 3 N 2 H 2
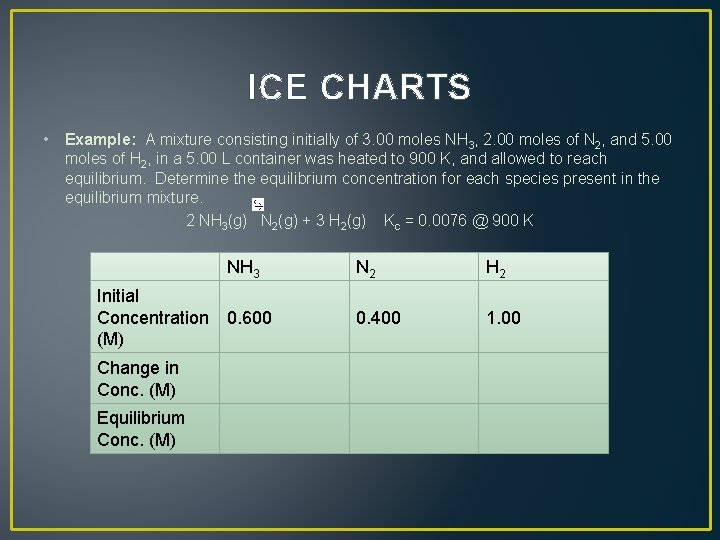
ICE CHARTS • Example: A mixture consisting initially of 3. 00 moles NH 3, 2. 00 moles of N 2, and 5. 00 moles of H 2, in a 5. 00 L container was heated to 900 K, and allowed to reach equilibrium. Determine the equilibrium concentration for each species present in the equilibrium mixture. 2 NH 3(g) N 2(g) + 3 H 2(g) Kc = 0. 0076 @ 900 K NH 3 Initial Concentration 0. 600 (M) Change in Conc. (M) Equilibrium Conc. (M) N 2 H 2 0. 400 1. 00
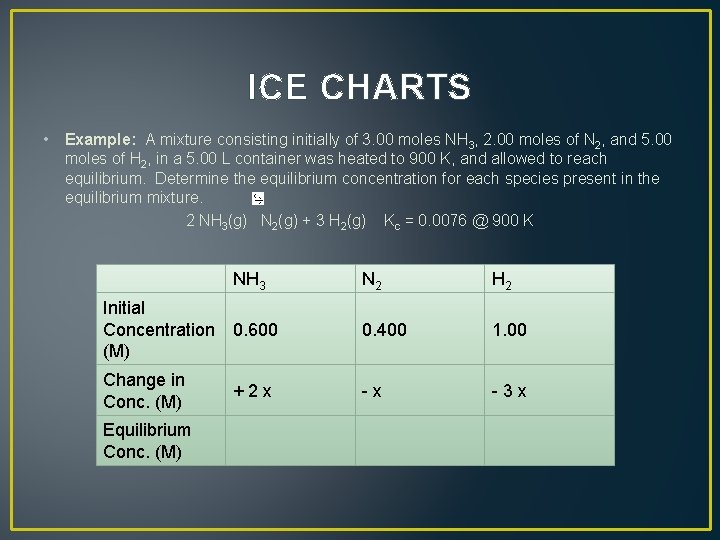
ICE CHARTS • Example: A mixture consisting initially of 3. 00 moles NH 3, 2. 00 moles of N 2, and 5. 00 moles of H 2, in a 5. 00 L container was heated to 900 K, and allowed to reach equilibrium. Determine the equilibrium concentration for each species present in the equilibrium mixture. 2 NH 3(g) N 2(g) + 3 H 2(g) Kc = 0. 0076 @ 900 K N 2 H 2 Initial Concentration 0. 600 (M) 0. 400 1. 00 Change in Conc. (M) - x - 3 x Equilibrium Conc. (M) NH 3 + 2 x
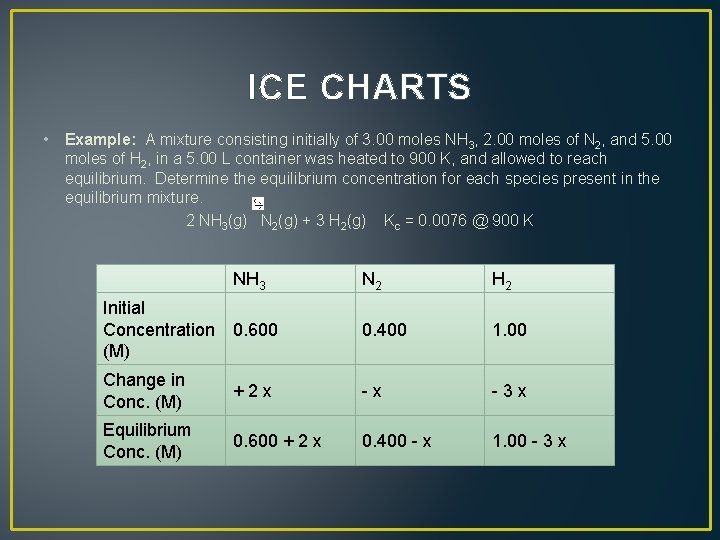
ICE CHARTS • Example: A mixture consisting initially of 3. 00 moles NH 3, 2. 00 moles of N 2, and 5. 00 moles of H 2, in a 5. 00 L container was heated to 900 K, and allowed to reach equilibrium. Determine the equilibrium concentration for each species present in the equilibrium mixture. 2 NH 3(g) N 2(g) + 3 H 2(g) Kc = 0. 0076 @ 900 K NH 3 N 2 H 2 Initial Concentration 0. 600 (M) 0. 400 1. 00 Change in Conc. (M) + 2 x - 3 x Equilibrium Conc. (M) 0. 600 + 2 x 0. 400 - x 1. 00 - 3 x

ICE CHARTS Example: 4. 00 moles of HI are placed in an evacuated 5. 00 L flask and then heated to 800 K. The system is allowed to reach equilibrium. What will be the equilibrium concentration of each species 2 HI(g) H 2(g) + I 2(g)
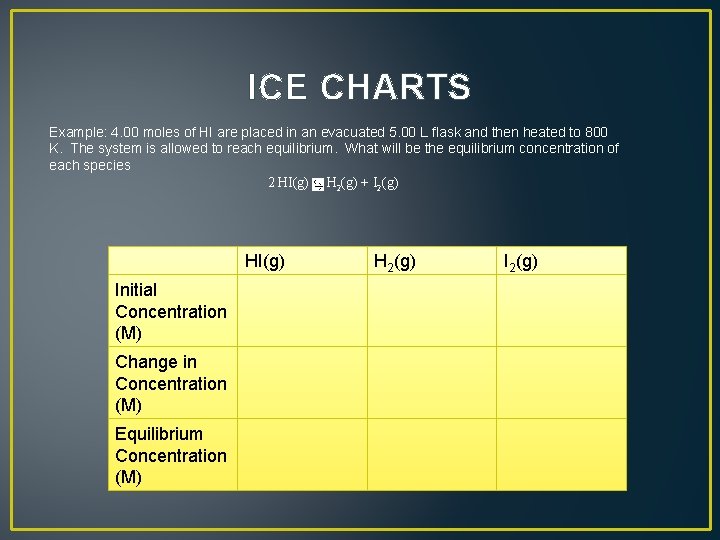
ICE CHARTS Example: 4. 00 moles of HI are placed in an evacuated 5. 00 L flask and then heated to 800 K. The system is allowed to reach equilibrium. What will be the equilibrium concentration of each species 2 HI(g) H 2(g) + I 2(g) Initial Concentration (M) Change in Concentration (M) Equilibrium Concentration (M) HI(g) H 2(g) I 2(g)
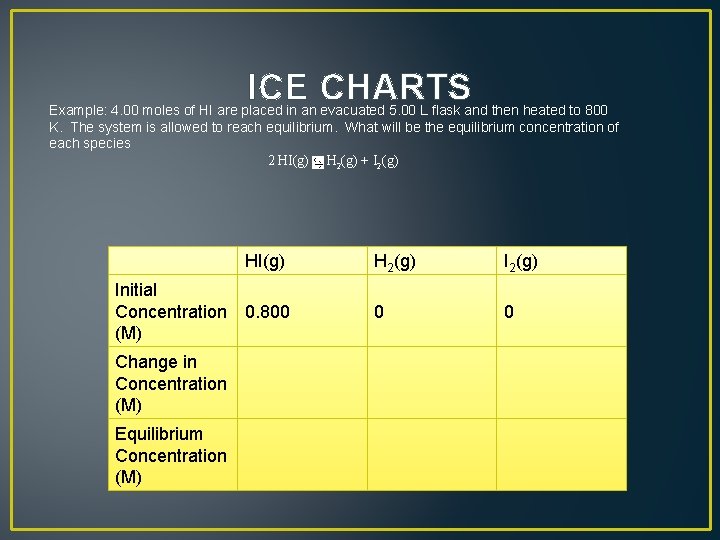
ICE CHARTS Example: 4. 00 moles of HI are placed in an evacuated 5. 00 L flask and then heated to 800 K. The system is allowed to reach equilibrium. What will be the equilibrium concentration of each species 2 HI(g) H 2(g) + I 2(g) HI(g) Initial Concentration 0. 800 (M) Change in Concentration (M) Equilibrium Concentration (M) H 2(g) I 2(g) 0 0
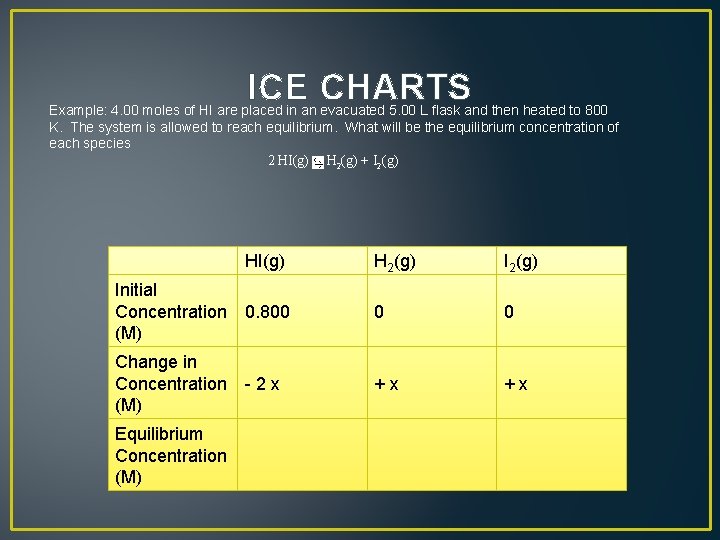
ICE CHARTS Example: 4. 00 moles of HI are placed in an evacuated 5. 00 L flask and then heated to 800 K. The system is allowed to reach equilibrium. What will be the equilibrium concentration of each species 2 HI(g) H 2(g) + I 2(g) H 2(g) Initial Concentration 0. 800 (M) 0 0 Change in Concentration - 2 x (M) + x Equilibrium Concentration (M) HI(g)
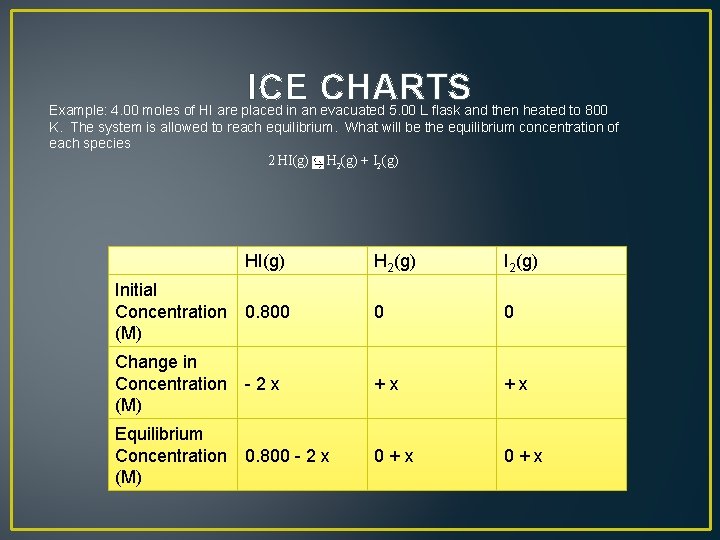
ICE CHARTS Example: 4. 00 moles of HI are placed in an evacuated 5. 00 L flask and then heated to 800 K. The system is allowed to reach equilibrium. What will be the equilibrium concentration of each species 2 HI(g) H 2(g) + I 2(g) HI(g) H 2(g) Initial Concentration 0. 800 (M) 0 0 Change in Concentration - 2 x (M) + x Equilibrium Concentration 0. 800 - 2 x (M) 0 + x

ICE CHARTS Example: 0. 600 moles of NO and 0. 750 moles of O 2 are placed in an empty 2. 00 L flask. The system is allowed to establish equilibrium. What will be the equilibrium concentration of each species in the flask? 2 NO 2(g) 2 NO(g) + O 2(g) Kc = 0. 50
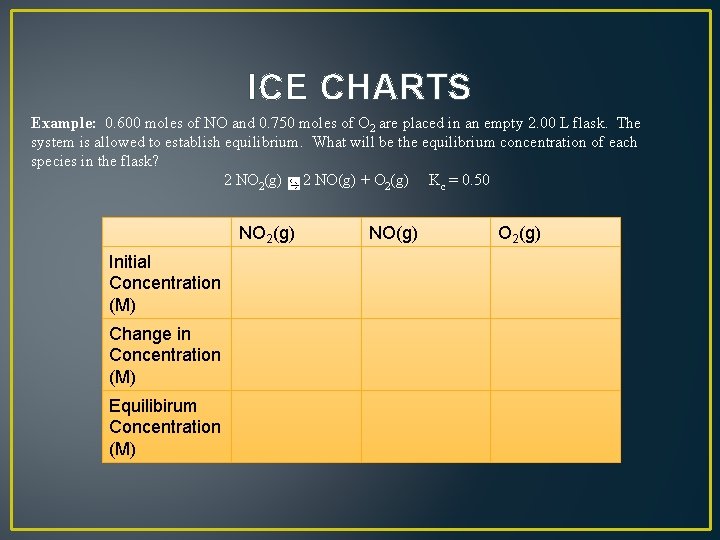
ICE CHARTS Example: 0. 600 moles of NO and 0. 750 moles of O 2 are placed in an empty 2. 00 L flask. The system is allowed to establish equilibrium. What will be the equilibrium concentration of each species in the flask? 2 NO 2(g) 2 NO(g) + O 2(g) Kc = 0. 50 Initial Concentration (M) Change in Concentration (M) Equilibirum Concentration (M) NO 2(g) NO(g) O 2(g)
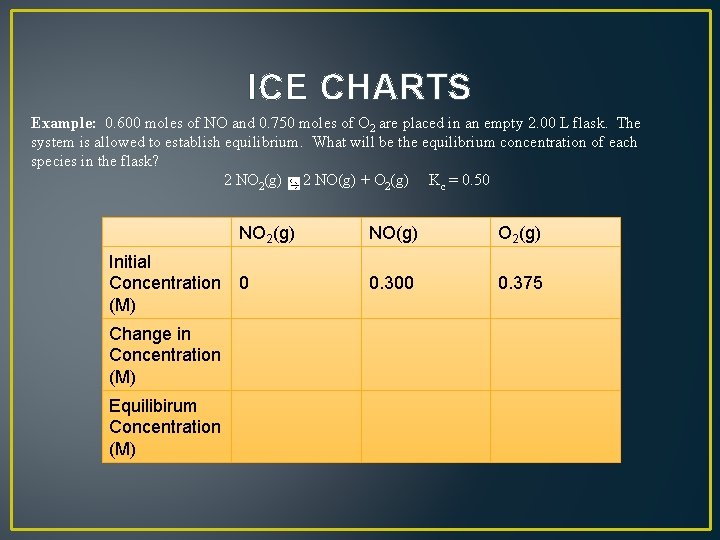
ICE CHARTS Example: 0. 600 moles of NO and 0. 750 moles of O 2 are placed in an empty 2. 00 L flask. The system is allowed to establish equilibrium. What will be the equilibrium concentration of each species in the flask? 2 NO 2(g) 2 NO(g) + O 2(g) Kc = 0. 50 NO 2(g) Initial Concentration 0 (M) Change in Concentration (M) Equilibirum Concentration (M) NO(g) O 2(g) 0. 300 0. 375
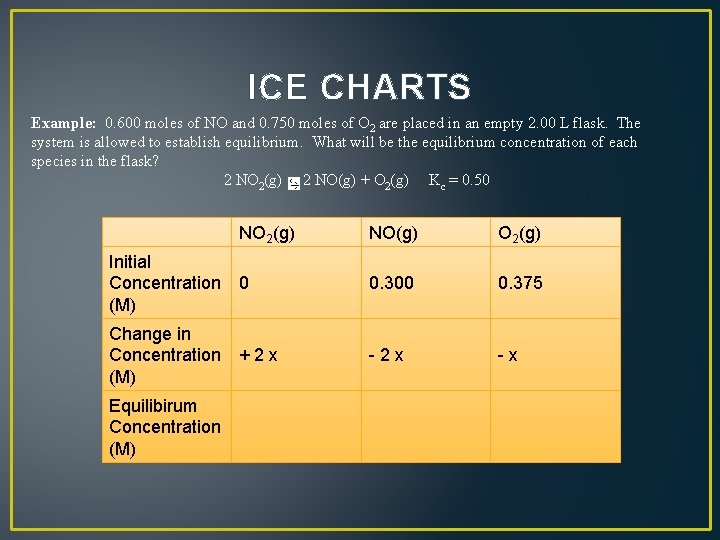
ICE CHARTS Example: 0. 600 moles of NO and 0. 750 moles of O 2 are placed in an empty 2. 00 L flask. The system is allowed to establish equilibrium. What will be the equilibrium concentration of each species in the flask? 2 NO 2(g) 2 NO(g) + O 2(g) Kc = 0. 50 NO(g) O 2(g) Initial Concentration 0 (M) 0. 300 0. 375 Change in Concentration + 2 x (M) - 2 x - x Equilibirum Concentration (M) NO 2(g)
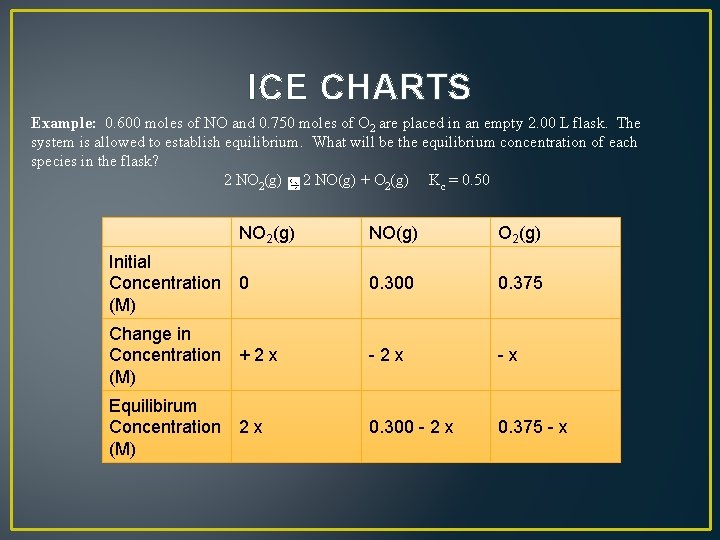
ICE CHARTS Example: 0. 600 moles of NO and 0. 750 moles of O 2 are placed in an empty 2. 00 L flask. The system is allowed to establish equilibrium. What will be the equilibrium concentration of each species in the flask? 2 NO 2(g) 2 NO(g) + O 2(g) Kc = 0. 50 NO 2(g) NO(g) O 2(g) Initial Concentration 0 (M) 0. 300 0. 375 Change in Concentration + 2 x (M) - 2 x - x Equilibirum Concentration 2 x (M) 0. 300 - 2 x 0. 375 - x
- Slides: 26