Chemical Equilibrium Relationship of concentrations at equilibrium a

Chemical Equilibrium

�Relationship of concentrations at equilibrium � � � a. A + b. B d. D + e. E Kc = [D]d [E]e [A]a [B]b

What the Constant tells you If K is very large (K>> 1) then products are more than reactants. Equilibrium lies to the right. (Numerator is large) If K is very small (K<< 1) then reactants are more than products. Equilibrium lies to the left. (Denominator is large) Rates are equal, not concentrations.

For Equilibrium to occur Reaction must be reversible Ex. H 2 CO 3 (aq) H 2 O(l) + CO 2(g) N 2 O 4 (g) 2 NO 2(g)

At Equilibrium rates are equal N 2 O 4 (g) 2 NO 2(g) ratef = kf [N 2 O 4] 2 NO 2(g) N 2 O 4(g) rater = kr [NO 2]2

At Equilibrium rates are equal If rates are equal then: Or kf [N 2 O 4] = kr [NO 2]2 kf kr = [NO 2]2 [N 2 O 4] = a constant “Kc”

K for reverse reactions are inverse of first reaction If for N 2 O 4 (g) 2 NO 2(g) : Kc = [NO 2]2 = 0. 212 (at 100°C) [N 2 O 4] Then for 2 NO 2(g) N 2 O 4(g) : Kc = [N 2 O 4] =4. 72 (at 100°C) [NO 2]2

Must specify reaction for K Kc = 0. 212 (at 100°C) for N 2 O 4 (g) 2 NO 2(g) : Kc = 4. 72 (at 100°C) for NO 2(g) N 2 O 4(g) : 2

If reaction is written different: If for 2 N 2 O 4 (g) 4 NO 2(g) (at 100°C): Kc = [NO 2]4 = (0. 212)2=0. 0449 [N 2 O 4]2 Must specify the balanced equation

If you have the Kc for two reactions 2 NOBr (g) 2 NO(g) + Br 2 (g) Kc = 0. 014 (at 100°C) Br 2 (g) + Cl 2 (g) 2 Br. Cl (g) Kc = 7. 2 (at 100°C) for

The sum of the two reactions 2 NOBr (g) + Cl 2 2 NO(g) + 2 Br. Cl (g) Will have the Kc that is the product of the two reactions. Kc = [NO]2 [Br. Cl]2 [NOBr]2 [Cl 2] [NO]2 [Br 2] [NOBr]2 X [Br. Cl]2 [Br 2] [Cl 2]

Homogeneous Reactions The species for the reactants and products are all the same.

Heterogeneous Reactions The species for the reactants and products are not all the same.

Heterogeneous Equilibria Whenever the species is different, pure solids and pure liquids are not involved in the equilibrium constant expression. Pure liquids and pure solids would have a constant density (mass/volume). When mass doubles, volume doubles. Consider concentration to be 1. Does not affect Kc.

Heterogeneous Equilibria What would the equilibrium-constant expression be for: H 2 CO 3 (aq) H 2 O (l) + CO 2 (g) Kc = [CO 2] [H 2 CO 3 ]

Problems involving Kc All the concentrations at equilibrium can be given and the equilibrium-constant can be calculated (easy) The Kc can be given and you can find the concentration of a reactant or product (easy) Initial concentrations are given, and one final concentration is given. How do we solve?

Reaction Quotient Number obtained by substituting reactant and product concentrations or partial pressures at any point into a reaction into the equilibriumconstant expression. Used to determine the direction a reaction will proceed to reach equilibrium.

Reaction Quotient Formula Relationship of concentrations at any point in the reaction (Qp can be written for pressures) a. A + b. B d. D + e. E Qc = [D]d [E]e [A]a [B]b

Reaction Quotient vs. Equilibrium Constant If: Q = K This reaction is already at equilibrium. Yeah! Q < K This reaction needs to decrease reactants and increase products to reach K. Q > K This reaction needs to increase reactants and decrease products to reach K.

Reaction Quotient vs. Equilibrium Constant Q < K This reaction needs to decrease reactants and increase products to reach K. Q is too small and it needs to increase. In order for that to happen the numerator needs to get bigger and the denominator needs to get smaller.

Reaction Quotient vs. Equilibrium Constant Q > K This reaction needs to decrease reactants and increase products to reach K. Q is too big and it needs to decrease. In order for that to happen the numerator needs to get smaller and the denominator needs to get bigger.

Notice! There is only one K at a specific temperature for a reaction. There can be many Q for a reaction at a temperature, because the reaction is proceeding in the direction towards K.

Now you should be ready to take on Stoichiometry, again!

Oh, Beta Ray Bill, you are so not funny! You are finally getting it!

What do you do if… Initial concentrations are given, and one final concentration is given. How do we solve? Stoichiometry (easy) Stoichiometry to find how the other concentrations have changed also

Stoich Example: A 1. 000 L container is filled with 1. 000 x 10 -3 mol H 2 (g) and 2. 000 x 10 -3 mol I 2 (g) at 448 °C. What is the value for the equilibrium constant Kc for the reaction H 2 (g) + I 2 (g) 2 HI (g) at that temperature?
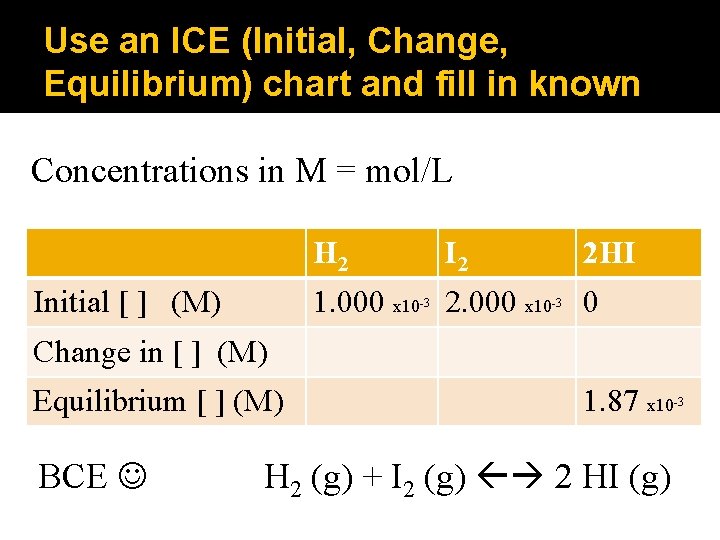
Use an ICE (Initial, Change, Equilibrium) chart and fill in known Concentrations in M = mol/L H 2 Initial [ ] (M) I 2 2 HI 1. 000 x 10 2. 000 x 10 0 -3 -3 Change in [ ] (M) Equilibrium [ ] (M) BCE 1. 87 x 10 H 2 (g) + I 2 (g) 2 HI (g) -3
![Express Change in [ ] by looking at BCE coefficients in ICE chart Concentrations Express Change in [ ] by looking at BCE coefficients in ICE chart Concentrations](http://slidetodoc.com/presentation_image_h2/1be2ccb3eb483a4a3fb34dab26e8adb5/image-28.jpg)
Express Change in [ ] by looking at BCE coefficients in ICE chart Concentrations in M = mol/L H 2 Initial [ ] (M) I 2 2 HI 1. 000 x 10 2. 000 x 10 0 -3 -3 Change in [ ] (M) +1. 87 x 10 Equilibrium [ ] (M) 1. 87 x 10 BCE -3 H 2 (g) + I 2 (g) 2 HI (g) -3
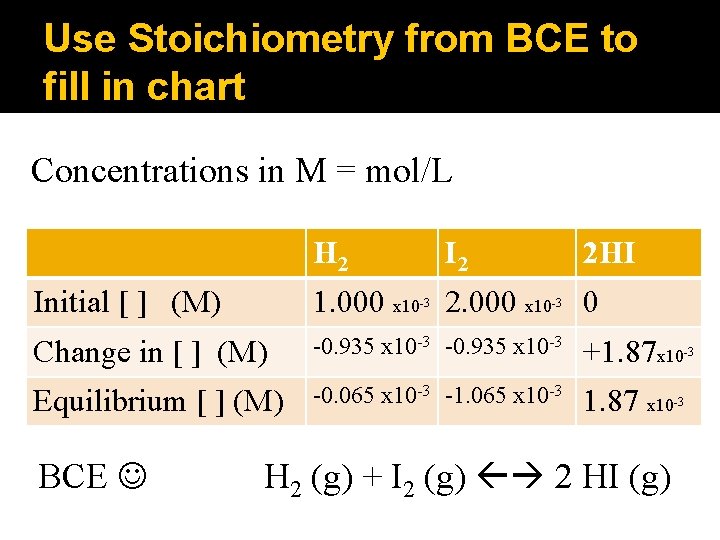
Use Stoichiometry from BCE to fill in chart Concentrations in M = mol/L H 2 I 2 2 HI Initial [ ] (M) 1. 000 x 10 2. 000 x 10 0 Change in [ ] (M) -0. 935 x 10 -3 +1. 87 x 10 Equilibrium [ ] (M) -0. 065 x 10 -3 -1. 065 x 10 -3 1. 87 x 10 BCE -3 -3 -3 H 2 (g) + I 2 (g) 2 HI (g) -3
![Use Kc equation to find K K = [HI]2 [H 2] [I 2] H Use Kc equation to find K K = [HI]2 [H 2] [I 2] H](http://slidetodoc.com/presentation_image_h2/1be2ccb3eb483a4a3fb34dab26e8adb5/image-30.jpg)
Use Kc equation to find K K = [HI]2 [H 2] [I 2] H 2 I 2 2 HI Initial [ ] (M) 1. 000 x 10 2. 000 x 10 0 Change in [ ] (M) -0. 935 x 10 -3 +1. 87 x 10 Equilibrium [ ] (M) -0. 065 x 10 -3 -1. 065 x 10 -3 1. 87 x 10 BCE -3 -3 -3 H 2 (g) + I 2 (g) 2 HI (g) -3
![K is equal to…. K = [HI]2 [H 2] [I 2] = 51 K is equal to…. K = [HI]2 [H 2] [I 2] = 51](http://slidetodoc.com/presentation_image_h2/1be2ccb3eb483a4a3fb34dab26e8adb5/image-31.jpg)
K is equal to…. K = [HI]2 [H 2] [I 2] = 51
- Slides: 31