Chemical equilibrium Reading Chapter 7 Atkins de Paula

Chemical equilibrium Reading Chapter 7, Atkins & de Paula. . chemistry comes aliveMOVIES 3NITRO 3 INI 3 IOD. MOV

Key ideas • • • It is all about Entropy of mixing! (again) Non-ideal solutions and Activities All thermodynamic functions are defined relative to a standard (reference) state. The equilibrium constant K quantifies extent to which a chemical reaction proceeds. How to measure Δr. GӨ Δr. HӨ, and Δr. SӨ van’t Hoff equation (II) for Δr. HӨ Le Chatelier’s principle
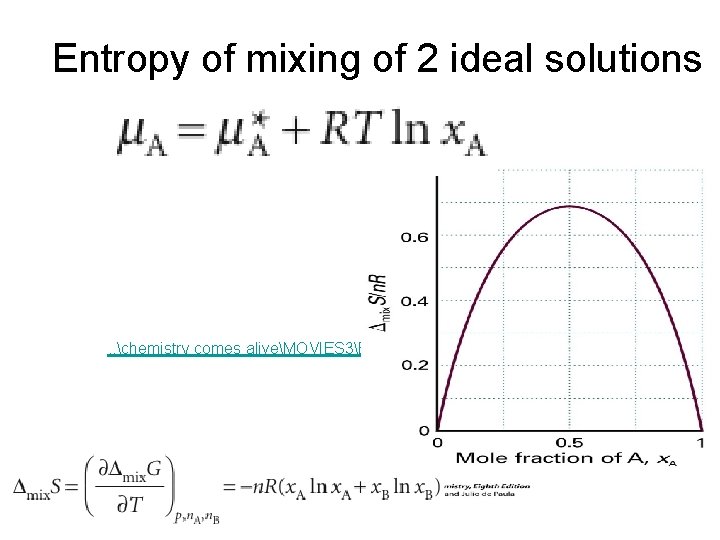
Entropy of mixing of 2 ideal solutions . . chemistry comes aliveMOVIES 3ENTROPYUNMIX. MOV

All thermodynamic functions are defined relative to a standard state bӨ =1 kg solute/(mole solvent) Biological standard state has p. H=7

Activity of non-ideal solutions
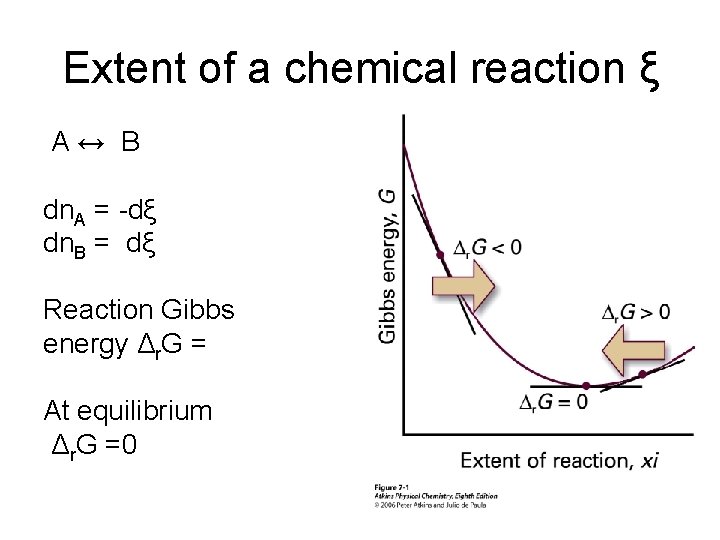
Extent of a chemical reaction ξ A↔ B dn. A = -dξ dn. B = dξ Reaction Gibbs energy Δr. G = At equilibrium Δr. G =0

Its all about entropy! • Getting mixed up! A chemical reaction never proceeds to completion

Perfect gas equilibrium A↔ B Standard reaction Gibbs energy Δr. GӨ = -RT ln K K is equilibrium constant K=p. B/p. A

Thermodynamic equilibrium constant “One of the most important equations in chemical thermodynamics” For reaction 0 = ΣJ νJ are stoichiometric numbers 0 = ΣJ νJμJ Δr. GӨ = −RT ln K

Example: ammonia synthesis reaction • Illustration 7. 1 & Example 7. 1 Atkins & de. Paula

Approximations for activities • Replace activities a. J by molalities, molar concentrations or partial pressures • Hence, if we measure equilibrium concentrations we can determine Δr. GӨ ! • Bad approximation for electrolytes
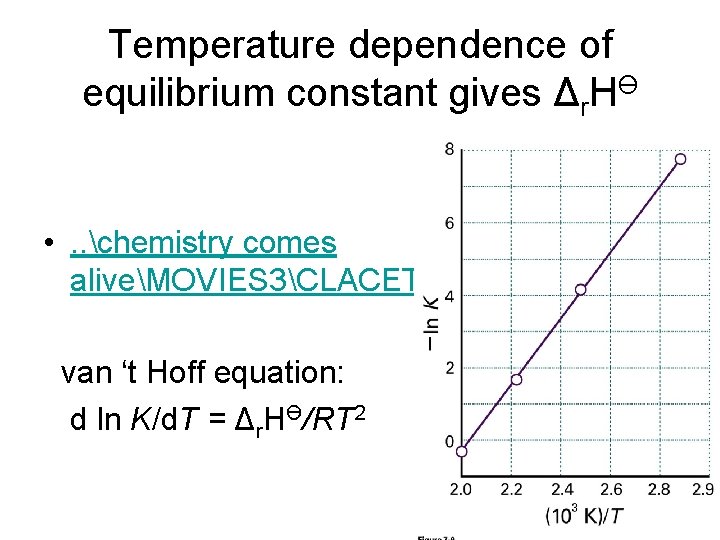
Temperature dependence of equilibrium constant gives Δr. HӨ • . . chemistry comes aliveMOVIES 3CLACET van ‘t Hoff equation: d ln K/d. T = Δr. HӨ/RT 2

How to measure Δr. GӨ Δr. HӨ, and Δr. SӨ • Δr. GӨ from K • Δ HӨ from K(T) r • Δr. SӨ =(Δr. HӨ - Δr. GӨ)/T • This is where tables of thermodynamic data come from.

Le Chatelier’s principle • A system at equilibrium, when subjected to a disturbance, responds in a way that tends to minimize the effect of the disturbance. • Example: Increased temperature favours the reactants in exothermic reactions and the products in endothermic reactions.

Biological standard state • Chemical standard state has the reference concentration bӨ =1 kg solute/(mole solvent) • Biological standard state has same reference concentrations except for hydrogen ions H+ which have the reference concentration p. H=7 i. e. , a. H+ = 10 -7

Summary • Due to the entropy of mixing chemical reactions never proceed to completion. • The equilibrium constant K defines the relative concentrations of the reactants and products in chemical equilibrium. • Δr. GӨ Δr. HӨ, and Δr. SӨ can be determined from K and its temperature dependence. • All thermodynamic functions are defined relative to a standard (reference) state.
- Slides: 16