Chemical Equilibrium Labs p 208 Thymol Blue Equilibrium
Chemical Equilibrium Labs p 208
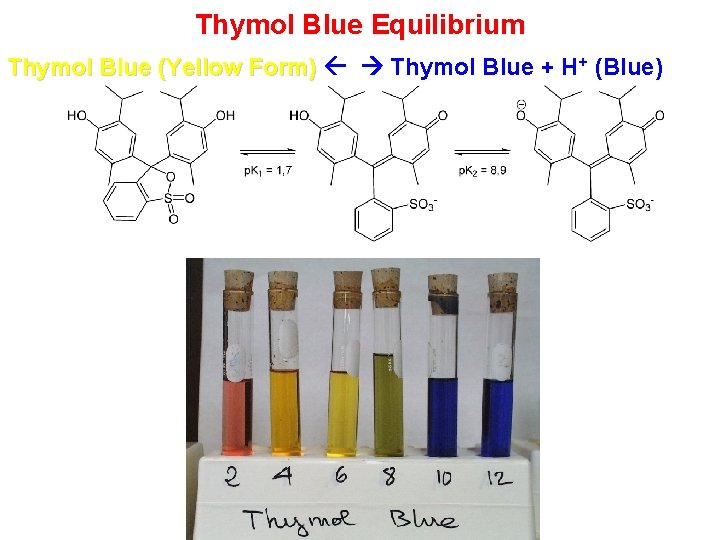
Thymol Blue Equilibrium Thymol Blue (Yellow Form) Thymol Blue + H+ (Blue)

Thymol Blue Equilibrium Stresses and Results Thymol Blue (Yellow Form) Thymol Blue + H+ (Blue) 1. 2. 3. Stress: Add HCl (H+). Result: Green Colour Stress: Add more HCl. Results: Yellow Colour Stress: Add Na. OH (OH-) reacts with H+ to form H 2 O) Result: Green Colour 4. Stress: Add more Na. OH. Results: Blue Colour
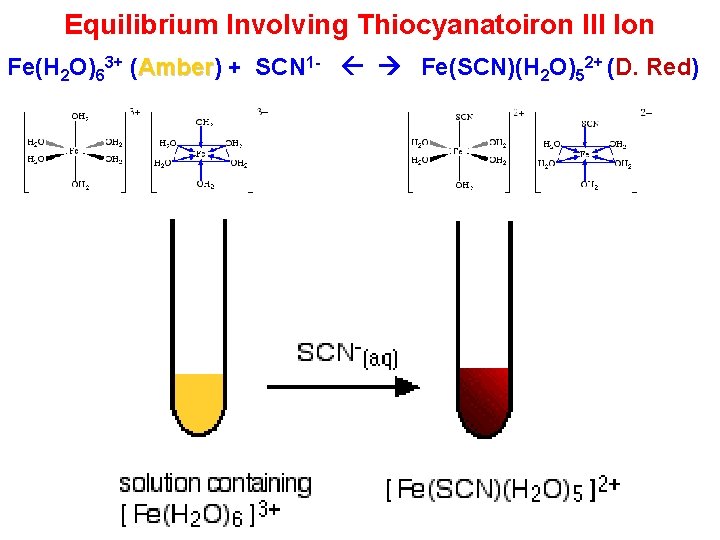
Equilibrium Involving Thiocyanatoiron III Ion Fe(H 2 O)63+ (Amber) Amber + SCN 1 - Fe(SCN)(H 2 O)52+ (D. Red)
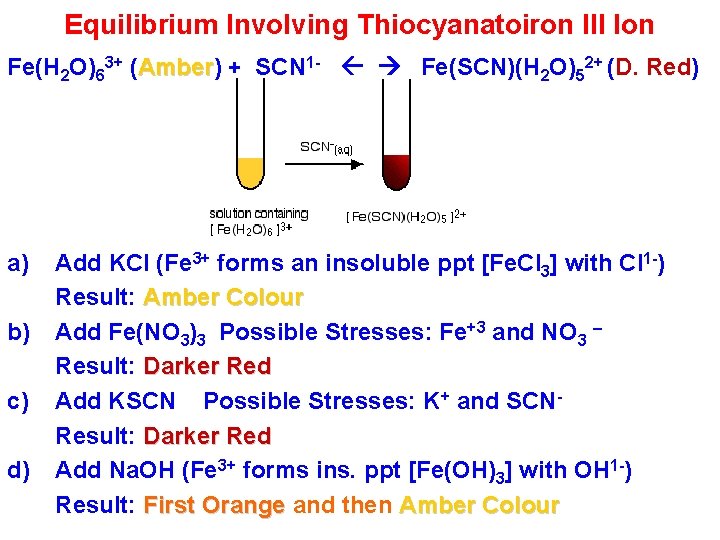
Equilibrium Involving Thiocyanatoiron III Ion Fe(H 2 O)63+ (Amber) Amber + SCN 1 - Fe(SCN)(H 2 O)52+ (D. Red) a) b) c) d) Add KCl (Fe 3+ forms an insoluble ppt [Fe. Cl 3] with Cl 1 -) Result: Amber Colour Add Fe(NO 3)3 Possible Stresses: Fe+3 and NO 3 – Result: Darker Red Add KSCN Possible Stresses: K+ and SCNResult: Darker Red Add Na. OH (Fe 3+ forms ins. ppt [Fe(OH)3] with OH 1 -) Result: First Orange and then Amber Colour
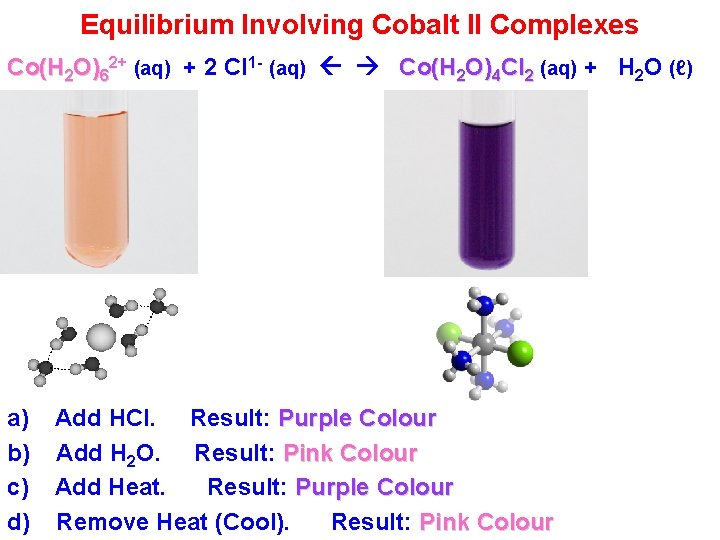
Equilibrium Involving Cobalt II Complexes Co(H 2 O)62+ (aq) + 2 Cl 1 - (aq) Co(H 2 O)4 Cl 2 (aq) + H 2 O (ℓ) a) b) c) d) Add HCl. Result: Purple Colour Add H 2 O. Result: Pink Colour Add Heat. Result: Purple Colour Remove Heat (Cool). Result: Pink Colour
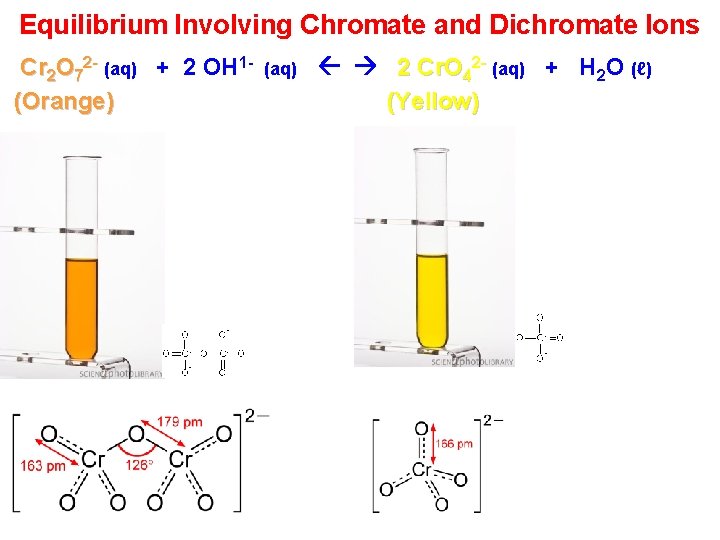
Equilibrium Involving Chromate and Dichromate Ions Cr 2 O 72 - (aq) + 2 OH 1(Orange) (aq) 2 Cr. O 42 - (aq) + H 2 O (ℓ) (Yellow)
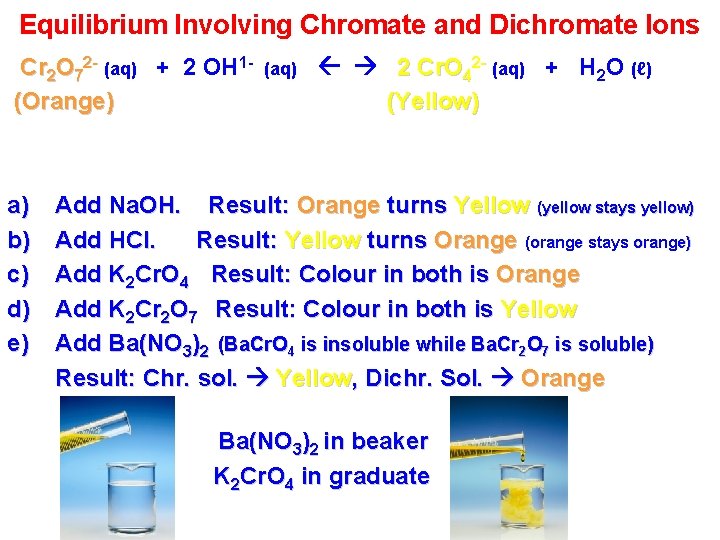
Equilibrium Involving Chromate and Dichromate Ions Cr 2 O 72 - (aq) + 2 OH 1(Orange) a) b) c) d) e) (aq) 2 Cr. O 42 - (aq) + H 2 O (ℓ) (Yellow) Add Na. OH. Result: Orange turns Yellow (yellow stays yellow) Add HCl. Result: Yellow turns Orange (orange stays orange) Add K 2 Cr. O 4 Result: Colour in both is Orange Add K 2 Cr 2 O 7 Result: Colour in both is Yellow Add Ba(NO 3)2 (Ba. Cr. O 4 is insoluble while Ba. Cr 2 O 7 is soluble) Result: Chr. sol. Yellow, Dichr. Sol. Orange Ba(NO 3)2 in beaker K 2 Cr. O 4 in graduate
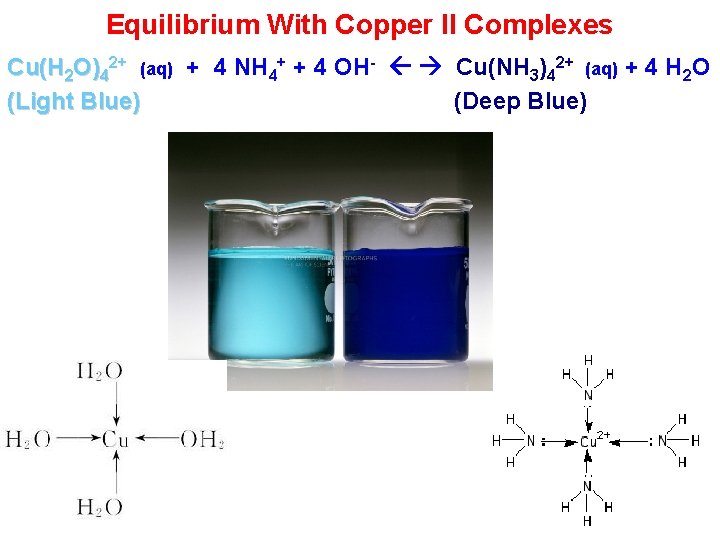
Equilibrium With Copper II Complexes Cu(H 2 O)42+ (aq) + 4 NH 4+ + 4 OH- Cu(NH 3)42+ (aq) + 4 H 2 O (Light Blue) (Deep Blue)

Equilibrium With Copper II Complexes Cu(H 2 O)42+ (aq) + 4 NH 4+ + 4 OH- Cu(NH 3)42+ (aq) + 4 H 2 O (Light Blue) (Deep Blue) a) Add NH 3 (aq) (NH 3 (aq) is NH 4+ + OH-) Result: The light blue colour turns a deeper blue b) c) Add more NH+. Result: The colour Yet Deeper Blue Add HCl (Yields H+ and Cl-). The deep Blue Light Blue

End of Lab Results

A A
- Slides: 12