Chemical Equilibrium Ksp of CaOH2 Equilibrium n Reaction

Chemical Equilibrium: Ksp of Ca(OH)2

Equilibrium n Reaction occurs in both forward and reverse directions q At equal rates! n Concentration of reactants and products in constant ratio n For example: a. A(g) + b. B(aq) + c. C(s) ⇌ x. X(g) + y. Y(aq) + z. Z(l)

Equilibrium q Represented by the equilibrium constant expression: n Written as products divided by reactants Each substance raised to power of balancing coefficient Only includes aqueous and gaseous substances n For example: n n a. A(g) + b. B(aq) + c. C(s) ⇌ x. X(g) + y. Y(aq) + z. Z(l) Kc is the equilibrium constant.

Solubility Product Constant (Ksp) n Equilibrium constant for a dissolution reaction q i. e. A solid breaking up into solution n Example:

The Salt for this Experiment n Calcium Hydroxide (Ca(OH)2) q More soluble than most hydroxides Dissolves a measureable amount in water: q Ksp calculated with molarities of the two ions q n Remember: There are 2 moles OH- for every 1 mol Ca 2+ (will have different molarities!)

Saturated Solutions n Contain maximum concentration of solute q q Created by adding excess solute to solvent Excess can then be filtered out n Max concentration changes with temperature n Two solutions in this experiment: q Room temperature n q Provided for you High temperature n Add solid to boiling water
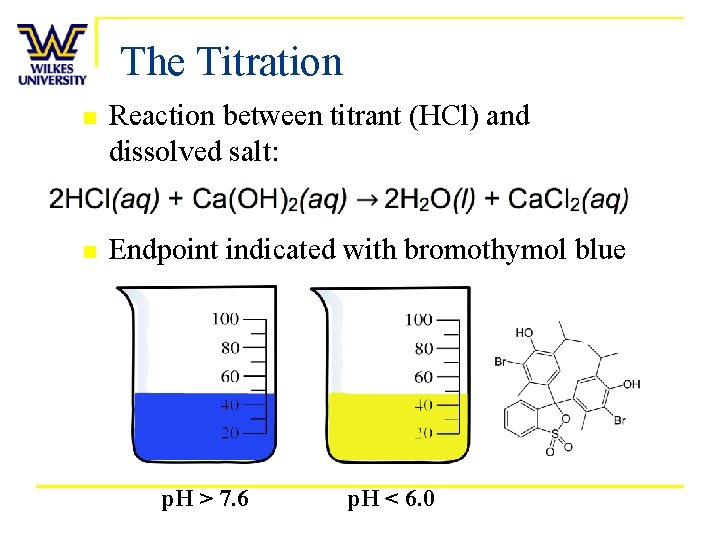
The Titration n Reaction between titrant (HCl) and dissolved salt: n Endpoint indicated with bromothymol blue p. H > 7. 6 p. H < 6. 0

Solubility Calculations n Calculate the moles of HCl added in the titration q n Determine moles of Ca(OH)2 in solution q n Divide moles by volume in L Determine [Ca 2+] and [OH-] q n Use stoichiometric ratio from titration reaction Calculate [Ca(OH)2] q n mol = Volume (L) x Molarity How many moles of each in compound? Calculate Ksp

Thermodynamics of a Reaction n

Thermodynamics of a Reaction Cont’d n

Variation of Ksp n Equilibrium constants vary with temperature q n n Effect depends on magnitude and sign of ΔH° Value of K also depends on ΔS° Combined relationship expressed with van’t Hoff equation: y = m(x) + b b. R=DS -m. R=DH q Note: R is 8. 314 J/(mol*K)
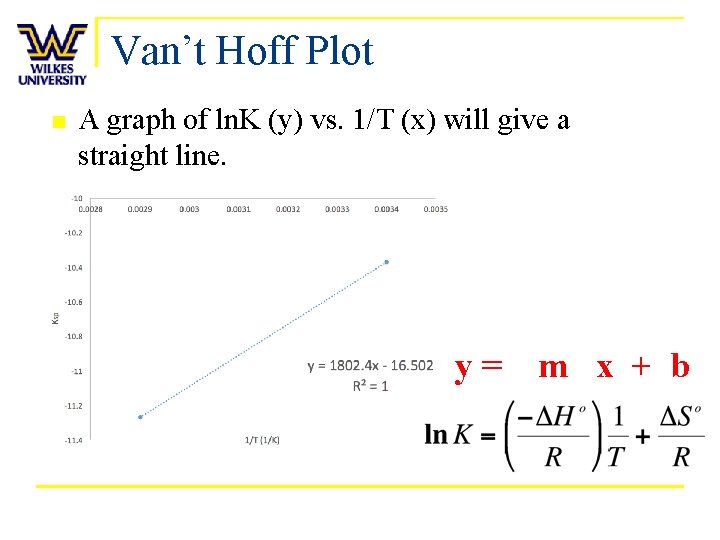
Van’t Hoff Plot n A graph of ln. K (y) vs. 1/T (x) will give a straight line. y= m x + b

Thermodynamic Calculations n Standard enthalpy change (ΔH°) in k. J/mol q n Standard entropy change (ΔS°) in J/(mol*K) q n Calculated with slope of van’t Hoff and R (turn to k. J!) Calculated with intercept of van’t Hoff and R Standard Gibbs Free Energy change (ΔG°) at 298 K q Calculated with equation: q Note: Make sure DH and DS are both in k. J!
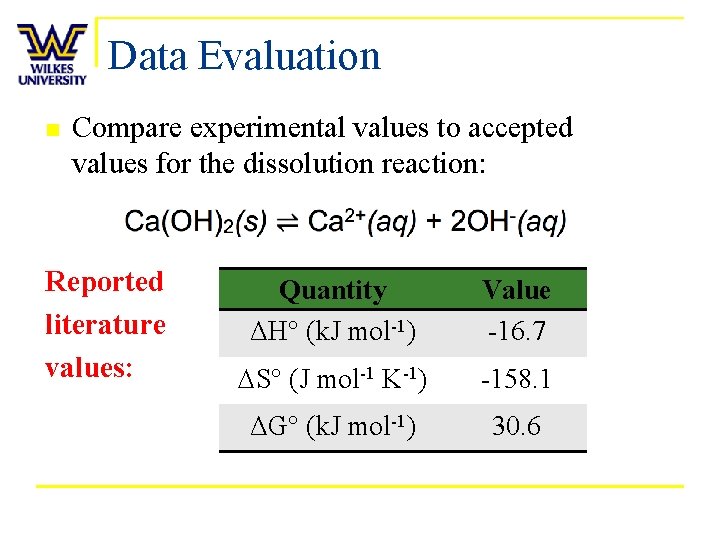
Data Evaluation n Compare experimental values to accepted values for the dissolution reaction: Reported literature values: Quantity ΔH° (k. J mol-1) Value -16. 7 DS° (J mol-1 K-1) -158. 1 ΔG° (k. J mol-1) 30. 6
- Slides: 14