Chemical Equilibrium Concept of Dynamic Equilibrium Heterogeneous Equilibrium
Chemical Equilibrium Concept of Dynamic Equilibrium Heterogeneous Equilibrium Constant Homogeneous Equilibrium Applications Le Chatelier’s Principle Temperature 12/4/2020 Reactants/Products Pressure/Volume

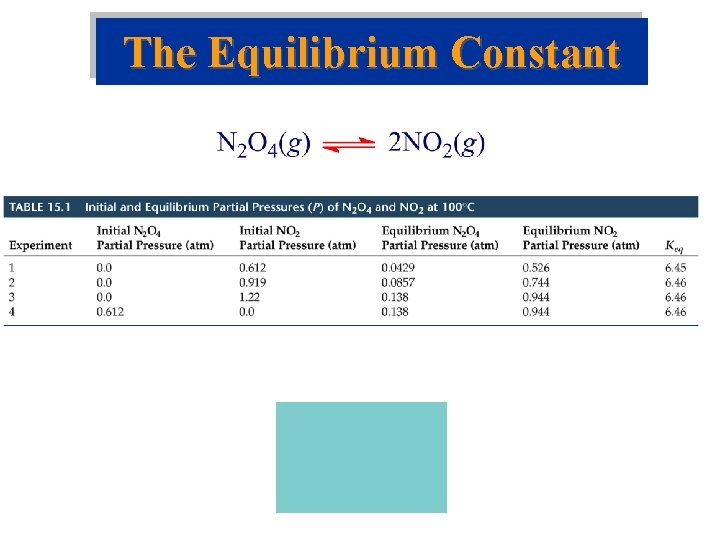
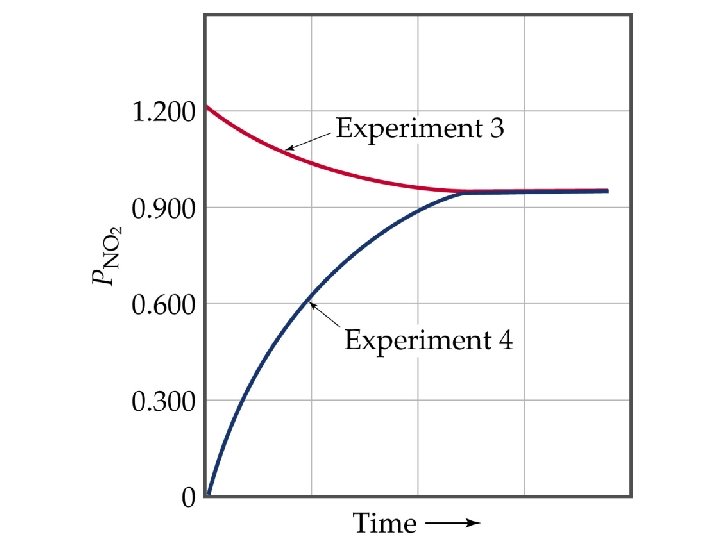
The Concept of Equilibrium • Consider colorless frozen N 2 O 4. At room temperature, it decomposes to brown NO 2: N 2 O 4(g) 2 NO 2(g). • At some time, the color stops changing and we have a mixture of N 2 O 4 and NO 2. • Chemical equilibrium is the point at which the concentrations of all species are constant.
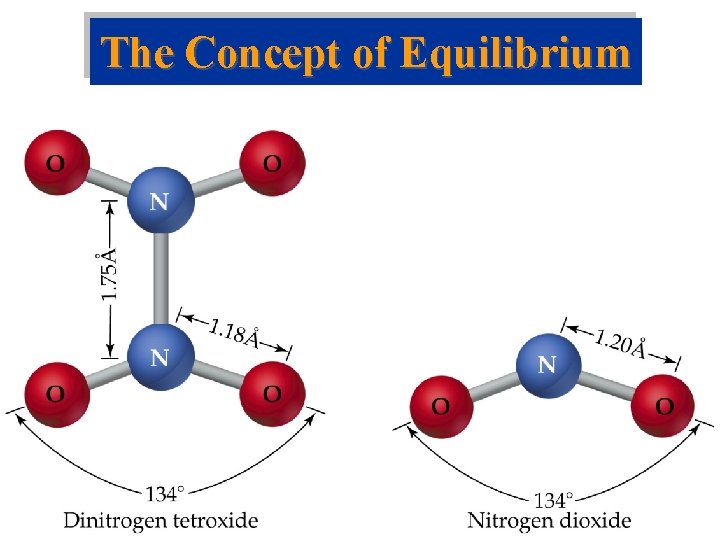
The Concept of Equilibrium
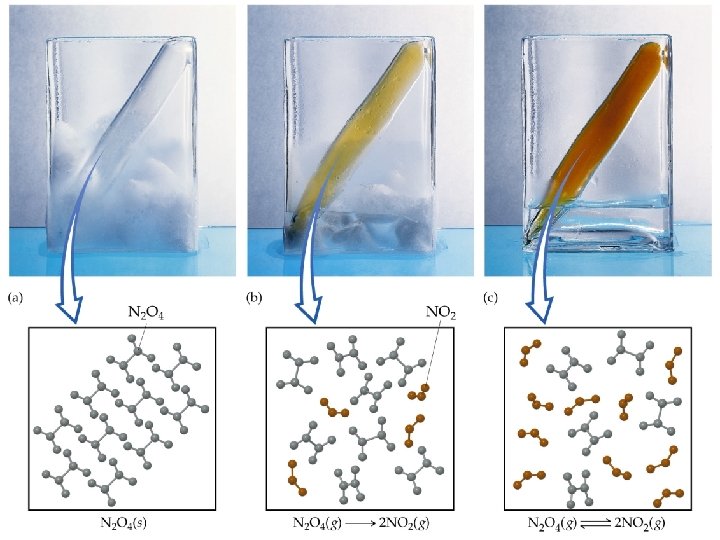

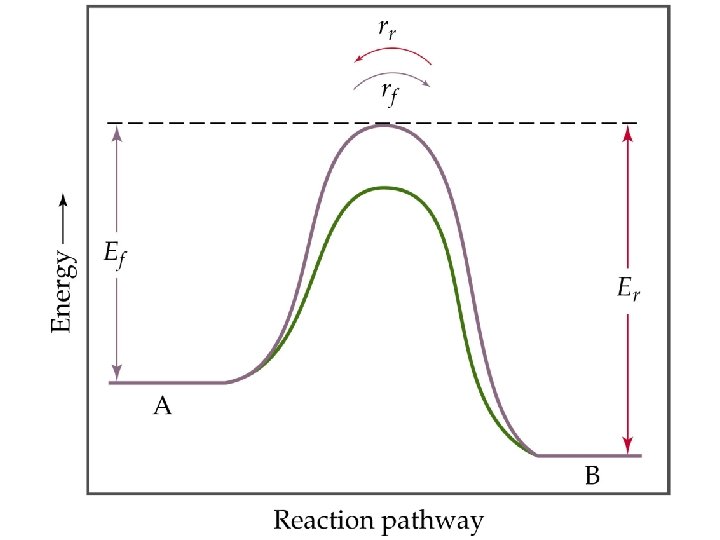
The Concept of Equilibrium • At equilibrium, as much N 2 O 4 reacts to form NO 2 as NO 2 reacts to re-form N 2 O 4: • The double arrow implies the process is dynamic. • Consider Forward reaction: A B Rate = kf[A] Reverse reaction: B A Rate = kr[B] • At equilibrium kf[A] = kr[B]. NO 2 – equilibrium
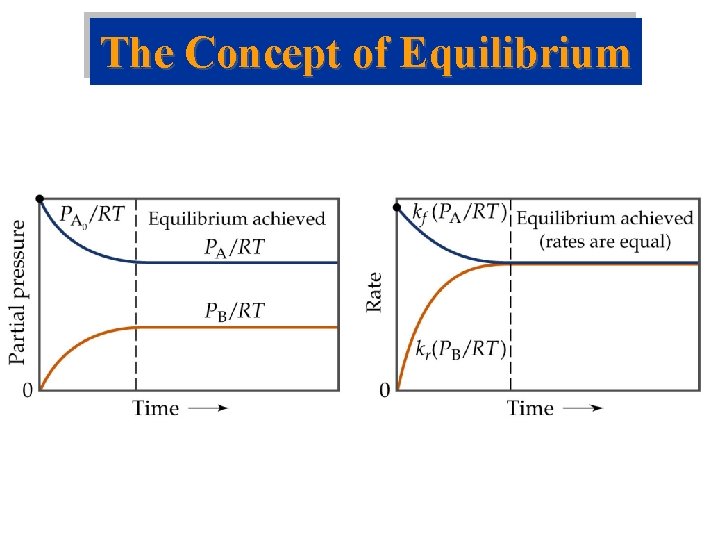
The Concept of Equilibrium

The Equilibrium Constant • For a general reaction in the gas phase the equilibrium constant expression is where Keq is the equilibrium constant.

The Equilibrium Constant • For a general reaction the equilibrium constant expression for everything in solution is where Keq is the equilibrium constant.
The Equilibrium Constant
The Equilibrium Constant The Magnitude of Equilibrium Constants

The Equilibrium Constant The Direction of the Chemical Equation and Keq • In the reverse direction:

The Equilibrium Constant Other Ways to Manipulate Chemical Equations and Keq Values • The reaction has which is the square of the equilibrium constant for

The Equilibrium Constant Other Ways to Manipulate Chemical Equations and Keq Values • Equilibrium constant for the reverse direction is the inverse of that for the forward direction. • When a reaction is multiplied by a number, the equilibrium constant is raised to that power. • The equilibrium constant for a reaction which is the sum of other reactions is the product of the equilibrium constants for the individual reactions.


The Equilibrium Constant Given at 700 K: (i) H 2(g) + I 2(g) 2 HI(g) Keq = 54. 0 (ii) N 2(g) + 3 H 2(g) 2 NH 3(g) Keq’ = 1. 04 x 10 -4 Determine Keq’’ at 700 K for: 2 NH 3(g) + 3 I 2(g) 6 HI(g) + N 2(g) Answer: 1. 51 x 109 Unit(s) of the Equilibrium Constant Dimensionless because: Ø Partial Pressures divided by Reference Pressure of 1 atm. Ø Molarities divided by Reference Concentration of 1 Molar.

Given at 700 K: (i) H 2(g) + I 2(g) 2 HI(g) Keq = 54. 0 (ii) N 2(g) + 3 H 2(g) 2 NH 3(g) Keq’ = 1. 04 x 10 -4 Determine Keq’’ at 700 K for: 2 NH 3(g) + 3 I 2(g) 6 HI(g) + N 2(g) Answer: 1. 51 x 109

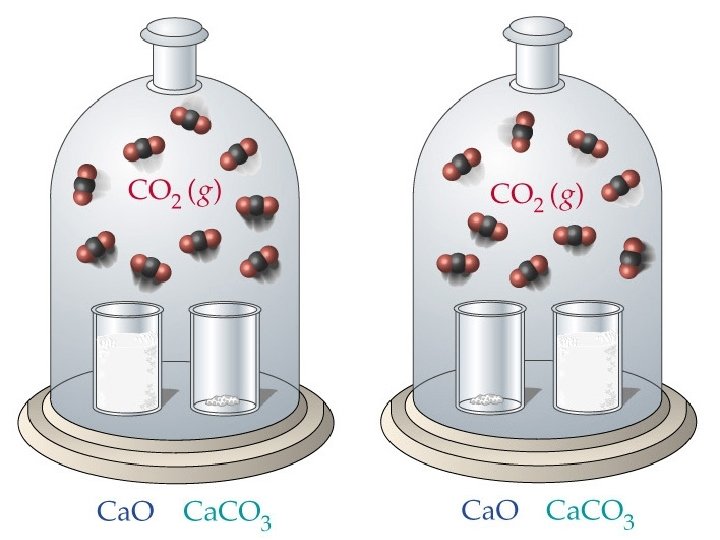
Heterogeneous Equilibria • When all reactants and products are in one phase, the equilibrium is homogeneous. • If one or more reactants or products are in a different phase, the equilibrium is heterogeneous. • Consider: – experimentally, the amount of CO 2 does not seem to depend on the amounts of Ca. O and Ca. CO 3. Why?

Heterogeneous Equilibria • The concentration of a solid or pure liquid is its density divided by molar mass. • Neither density nor molar mass is a variable, the concentrations of solids and pure liquids are constant. • For the decomposition of Ca. CO 3:

Heterogeneous Equilibria • We ignore the concentrations of pure liquids and pure solids in equilibrium constant expressions. • The amount of CO 2 formed will not depend greatly on the amounts of Ca. O and Ca. CO 3 present. • Examples:

Calculating Equilibrium Constants • Proceed as follows: – Tabulate initial and equilibrium concentrations (or partial pressures) given. – If an initial and equilibrium concentration is given for a species, calculate the change in concentration. – Use stoichiometry on the change in concentration line only to calculate the changes in concentration of all species. – Deduce the equilibrium concentrations of all species. • Usually, the initial concentration of products is zero. (This is not always the case. )

Calculating Equilibrium Constants 1. A mixture of H 2(g) and N 2(g) is allowed to attain equilibrium at 472 o. C. At equilibrium, the partial pressures of the gases were determined to be 7. 38 atm H 2, 2. 46 atm N 2 and 0. 166 atm NH 3. Calculate Keq. 2. Enough ammonia is dissolved in 5. 00 L of water at 25 o. C to produce a solution that is 0. 0124 M in ammonia. The solution is then allowed to come to equilibrium. Analysis of the equilibrium mixture shows that the concentration of OH- is 4. 64 x 10 -4 M. Calculate Keq at 25 o. C for the reaction. 3. Gaseous sulfur trioxide decomposes to gases of sulfur dioxide and oxygen at high temperature in a sealed container. Initially the vessel is charged at 1000 K with SO 3(g) at a partial pressure of 0. 500 atm. At equilibrium, the SO 3 partial pressure is 0. 200 atm. Calculate the value of Keq at 1000 K.

A mixture of H 2(g) and N 2(g) is allowed to attain equilibrium at 472 o. C. At equilibrium, the partial pressures of the gases were determined to be 7. 38 atm H 2, 2. 46 atm N 2 and 0. 166 atm NH 3. Calculate Keq. Answer: 2. 79 x 10 -5

Enough ammonia is dissolved in 5. 00 L of water at 25 o. C to produce a solution that is 0. 0124 M in ammonia. The solution is then allowed to come to equilibrium. Analysis of the equilibrium mixture shows that the concentration of OH- is 4. 64 x 10 -4 M. Calculate Keq at 25 o. C for the reaction. Answer: 1. 80 x 10 -5

Gaseous sulfur trioxide decomposes to gases of sulfur dioxide and oxygen at high temperature in a sealed container. Initially the vessel is charged at 1000 K with SO 3(g) at a partial pressure of 0. 500 atm. At equilibrium, the SO 3 partial pressure is 0. 200 atm. Calculate the value of Keq at 1000 K. Answer: 0. 338

Applications of Equilibrium Constants Predicting the Direction of Reaction • We define Q, the reaction quotient, for a general reaction as • Q = K only at equilibrium.

Applications of Equilibrium Constants Predicting the Direction of Reaction • If Q > K then the reverse reaction must occur to reach equilibrium (i. e. , products are consumed, reactants are formed, the numerator in the equilibrium constant expression decreases and Q decreases until it equals K). • If Q < K then the forward reaction must occur to reach equilibrium.
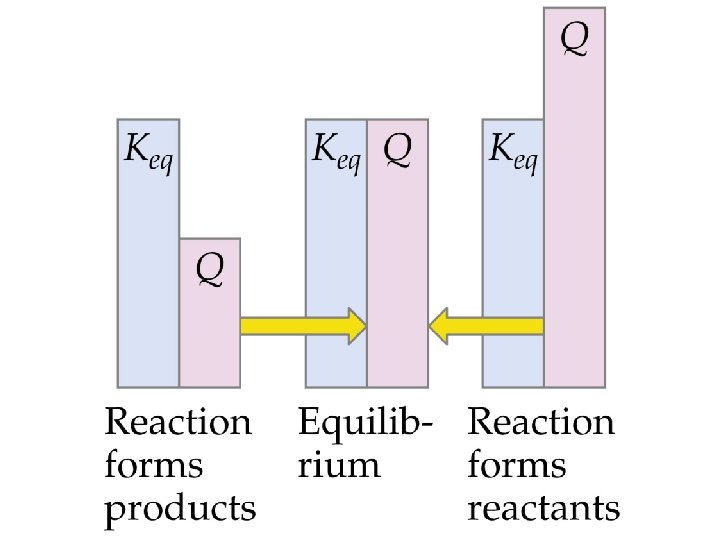

Applications of Equilibrium Constants • • Calculating Equilibrium Constants The same steps used to calculate equilibrium constants are used. Generally, we do not have a number for the change in concentration line. Therefore, we need to assume that x mol/L of a species is produced (or used). The equilibrium concentrations are given as algebraic expressions.

Applications of Equilibrium Constants Predicting the Direction of Reaction 1. At 1000 K the value of Keq for the reaction 2 SO 3(g) 2 SO 2(g) + O 2(g) is 0. 338. Calculate the value for Q, and predict the direction in which the reaction will proceed toward equilibrium if the initial partial pressures are 0. 16 atm for SO 3, 0. 41 atm for SO 2 , and 2. 5 atm for O 2. 2. At 448 o. C, Keq is 51 for: H 2(g) + I 2(g) 2 HI(g). Predict how the reaction will proceed to reach equilibrium at 448 o. C if we start with 2. 0 x 10 -2 mol HI , 1. 0 x 10 -2 mol H 2 , and 3. 0 x 10 -2 mol I 2 in a 2. 00 -L container. [Hint: Calculate partial pressures using Ideal Gas Law. Calculate Q. ] [Answer: Q=1. 33 Reaction proceeds from left to right (i. e. more products)]


Applications of Equilibrium Constants Calculating Equilibrium Concentrations 1. At 500 K the reaction: PCl 5(g) PCl 3(g) + Cl 2(g) , has Keq = 0. 497. In an equilibrium mixture at 500 K, the partial pressure of PCl 5 is 0. 860 atm and that of PCl 3 is 0. 350 atm. What is the partial pressure of Cl 2 in the equilibrium mixture? 2. A 1. 000 -L flask is filled with 1. 000 mol of H 2 and 2. 000 mol of I 2 at 448 o. C. The value of the Keq for the reaction: H 2(g) + I 2(g) 2 HI(g) , at 448 o. C is 50. 5. What are the partial pressures of H 2 , I 2 , and HI in the flask at equilibrium? [Hint: Solve for initial partial pressures using Ideal Gas law. Set up initial/change/equil’m table and define x as change in partial pressures of reactants. Solve for x in Keq expression using quadratic formulation. ] [Answer: x = 55. 3 atm; H 2 = 3. 90 atm, I 2 = 63. 1 atm, HI = 111 atm. ]

2. A 1. 000 -L flask is filled with 1. 000 mol of H 2 and 2. 000 mol of I 2 at 448 o. C. The value of the Keq for the reaction: + I 2(g) 2 HI(g) , at 448 o. C is 50. 5. What are the partial pressures of H 2 , I 2 , and HI in the flask at equilibrium? H 2(g) [Hint: Solve for initial partial pressures using Ideal Gas law. Set up initial/change/equil’m table and define x as change in partial pressures of reactants. Solve for x in Keq expression using quadratic formulation. ] [Answer: x = 55. 3 atm; H 2 = 3. 90 atm, I 2 = 63. 1 atm, HI = 111 atm. ]

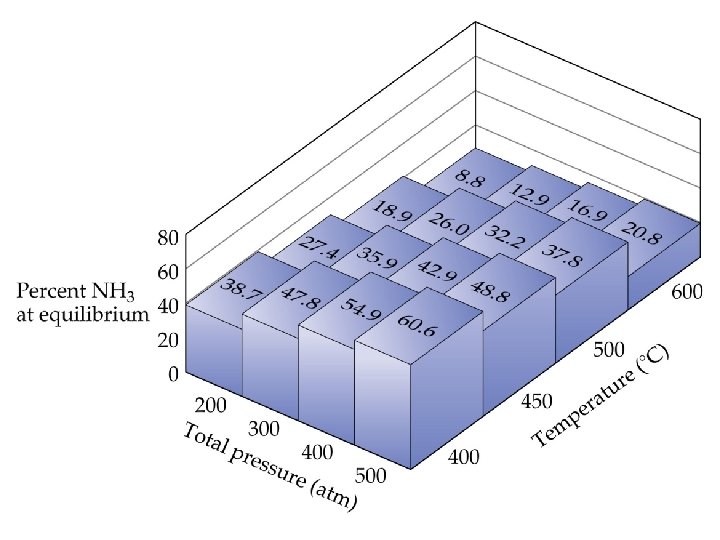
Le Châtelier’s Principle Consider the production of ammonia • As the pressure increases, the amount of ammonia present at equilibrium increases. • As the temperature decreases, the amount of ammonia at equilibrium increases. • Can this be predicted? Le Châtelier’s Principle: if a system at equilibrium is disturbed, the system will move in such a way as to counteract the disturbance.

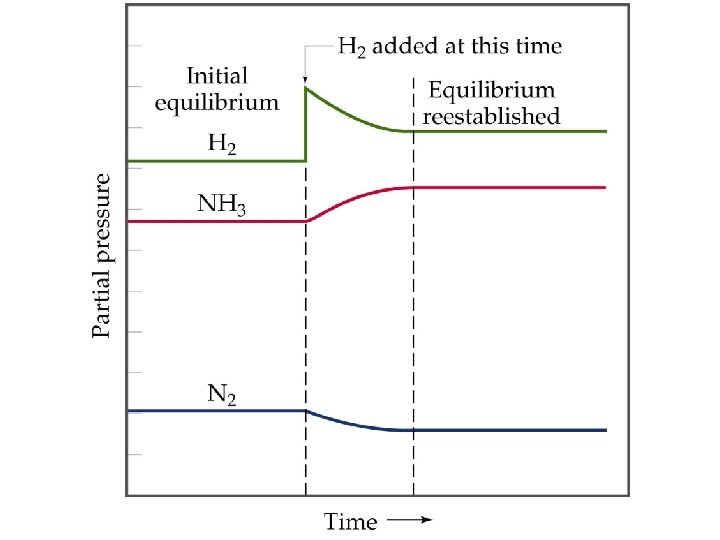
Le Châtelier’s Principle Change in Reactant or Product Concentrations • Consider the Haber process • If H 2 is added while the system is at equilibrium, the system must respond to counteract the added H 2 (by Le Châtelier). • The system must consume the H 2 and produce products until a new equilibrium is established. • So, [H 2] and [N 2] will decrease and [NH 3] increases.

Le Châtelier’s Principle • • Change in Reactant or Product Concentrations Adding a reactant or product shifts the equilibrium away from the increase. Removing a reactant or product shifts the equilibrium towards the decrease. To optimize the amount of product at equilibrium, we need to flood the reaction vessel with reactant and continuously remove product (Le Châtelier). We illustrate the concept with the industrial preparation of ammonia.
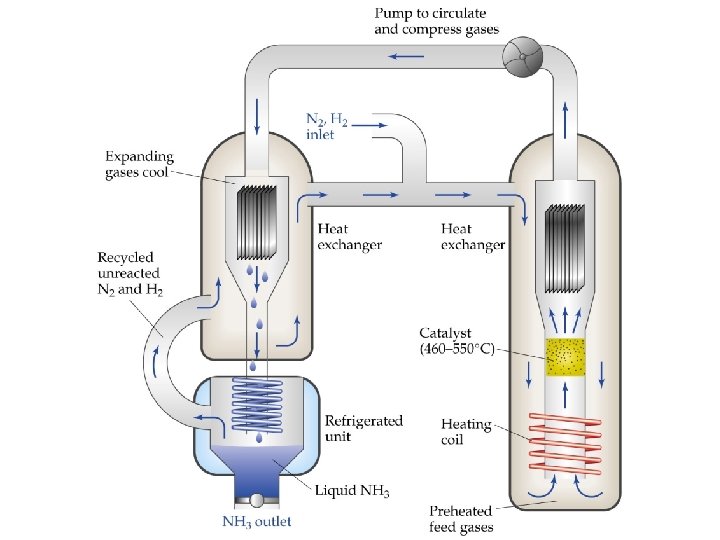

Le Châtelier’s Principle • • • Effects of Volume and Pressure Changes As volume is decreased pressure increases. Le Châtelier’s Principle: if pressure is increased the system will shift to counteract the increase. That is, the system shifts to remove gases and decrease pressure. An increase in pressure favors the direction that has fewer moles of gas. In a reaction with the same number of product and reactant moles of gas, pressure has no effect.

Le Châtelier’s Principle Effects of Volume and Pressure Changes • An increase in pressure (by decreasing the volume) favors the formation of colorless N 2 O 4. • The instant the pressure increases, the system is not at equilibrium and the concentration of both gases has increased. • The system moves to reduce the number moles of gas (i. e. backward reaction is favored).

Le Châtelier’s Principle Effect of Temperature Changes • The equilibrium constant is temperature dependent. • For an endothermic reaction, H > 0 and heat can be considered as a reactant. • For an exothermic reaction, H < 0 and heat can be considered as a product.

Le Châtelier’s Principle Effect of Temperature Changes • Adding heat (i. e. heating the vessel) favors away from the increase: – if H > 0, adding heat favors the forward reaction, – if H < 0, adding heat favors the reverse reaction. • Removing heat (i. e. cooling the vessel), favors towards the decrease: – if H > 0, cooling favors the reverse reaction, – if H < 0, cooling favors the forward reaction.

Le Châtelier’s Principle Effect of Temperature Changes • Consider for which H > 0. – Co(H 2 O)62+ is pale pink and Co. Cl 42 - is blue. – If a light purple room temperature equilibrium mixture is placed in a beaker of warm water, the mixture turns deep blue. – Since H > 0 (endothermic), adding heat favors the forward reaction, i. e. the formation of blue Co. Cl 42 -. – If the room temperature equilibrium mixture is placed in a beaker of ice water, the mixture turns bright pink. – Since H > 0, removing heat favors the reverse reaction which is the formation of pink Co(H 2 O)62+.


Le Châtelier’s Principle The Effect of Catalysis • A catalyst lowers the activation energy barrier for the reaction. • Therefore, a catalyst will decrease the time taken to reach equilibrium. • A catalyst does not affect the composition of the equilibrium mixture.

Le Châtelier’s Principle Summary If a system at equilibrium is disturbed, the system will move in such a way as to counteract the disturbance. Ø Adding a reactant or product shifts the equilibrium away from the increase. Ø Upon pressure increase, reaction will proceed towards the side with the fewest number of gaseous moles. Ø Adding heat (i. e. heating the vessel) favors away from the increase: if ∆H > 0, adding heat favors the forward reaction, if ∆H < 0, adding heat favors the reverse reaction. Ø A catalyst does not affect the composition of the equilibrium mixture.
Chemical Equilibrium Concept of Dynamic Equilibrium Heterogeneous Equilibrium Constant Homogeneous Equilibrium Applications Le Chatelier’s Principle Temperature Reactants/Products Pressure/Volume
- Slides: 50