Chemical Equilibrium Chemical Equilibrium When a reaction is

Chemical Equilibrium

Chemical Equilibrium When a reaction is not totally converted from reactants to products, a condition can be set up known as chemical equilibrium. In this state, the concentrations of all reactants and products remain constant with time, as both forward and reverse reactions are occurring at the same rate. On the molecular level, there is frantic activity. Equilibrium is not static, but is a highly dynamic situation.

• Chemical Equilibrium: occurs when opposing reactions are proceeding at equal rates and the concentrations of products and reactants no longer change with time. Ratef = Rater

Chemical Equilibrium For this to occur, the system must be closed, no products or reactants may escape the system.
![For Reaction: A B Forward Reaction: A B Rate = kf[A] kf= forward rate For Reaction: A B Forward Reaction: A B Rate = kf[A] kf= forward rate](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-5.jpg)
For Reaction: A B Forward Reaction: A B Rate = kf[A] kf= forward rate constant Reverse reaction: B A Rate = kr[B] kr= reverse rate constant
![• As forward reaction occurs, – [A] decreases and forward rate slows. – • As forward reaction occurs, – [A] decreases and forward rate slows. –](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-6.jpg)
• As forward reaction occurs, – [A] decreases and forward rate slows. – [B] increases and the rate of reverse reaction increases. – Eventually the forward and reverse reactions reach the same rate. (Chemical Equilibrium)
![kf[A]= kr[B] • Once equilibrium is established, the concentrations of A and B do kf[A]= kr[B] • Once equilibrium is established, the concentrations of A and B do](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-7.jpg)
kf[A]= kr[B] • Once equilibrium is established, the concentrations of A and B do not change.
![Writing equilibrium expressions Keq = [products] [reactants] NOTE: [at equilibrium] Note: eq is the Writing equilibrium expressions Keq = [products] [reactants] NOTE: [at equilibrium] Note: eq is the](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-8.jpg)
Writing equilibrium expressions Keq = [products] [reactants] NOTE: [at equilibrium] Note: eq is the general subscript for an equilibrium constant. Your textbook uses Keq throughout. Please look at the equilibrium constants given on the AP exam. These are the same as Keq, but are more specific to a type of reaction (examples: gaseous reaction or acid base reaction).

Two common ways of describing equilibrium: Kc = Concentration of substances in the reaction are known. Kp = Units of partial pressure are used instead of concentration. *K p is used when the reactants and products are in the gaseous state. *Solids and liquids are left out of equilibrium expressions because their concentrations do not change during chemical reactions.

Equilibrium Expressions Summary - - The K value for a reaction that is reversed is the reciprocal of that K for the forward reaction There are no units for K because of corrections to the nonideal behavior of substances in the reaction.

Law of Mass Action: Expresses the relationship between concentrations of reactants and products at equilibrium. Example: a. A + b. B c. C + d. D Kc = [C]c[D]d [A]a[B]b
![Kc = c d [C] [D] a b [A] [B] • The expression above Kc = c d [C] [D] a b [A] [B] • The expression above](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-12.jpg)
Kc = c d [C] [D] a b [A] [B] • The expression above is known as the equilibrium-constant expression. • Kc is the equilibrium constant. – Subscript c indicates concentration in molarity.

Write the equilibrium constant expressions for the following reaction in the forward direction. 2 SO 2 (g) + O 2 (g) 2 SO 3(g)

Homework #22 • K= = = 3. 2 × 1011 14

Example Calculations can be made if the equilibrium concentrations of reactants and products are known. If [NH 3] = 3. 1 x 10 -2 mol/L, [N 2]=8. 5 x 10 -1 mol/L, and [H 2] = 3. 1 x 10 -3 mol/L, N 2 (g) + 3 H 2 (g) 2 NH 3 (g) a. What is the value of K? b. What is the value of K’ (for the reverse reaction)? c. What would be the K for this reaction? ½ N 2 (g) + 3/2 H 2 (g) NH 3 (g) (3. 1 x 10 -2 )2 = (8. 5 x 10 -1 )(3. 1 x 10 -3 )3 = 3. 8 x 104 K’ = 1/K = 1/ 3. 8 x 104 =2. 6 x 10 -5 For the reaction in (c), Kc = K ½ = (3. 8 x 104 L 2/mol 2) ½ = 1. 9 x 102

N 2(g) + 3 H 2(g) 2 NH 3(g) ØThis process for making ammonia is done at high temperature and pressure. ØWhen complete all 3 components are present in the closed tank.
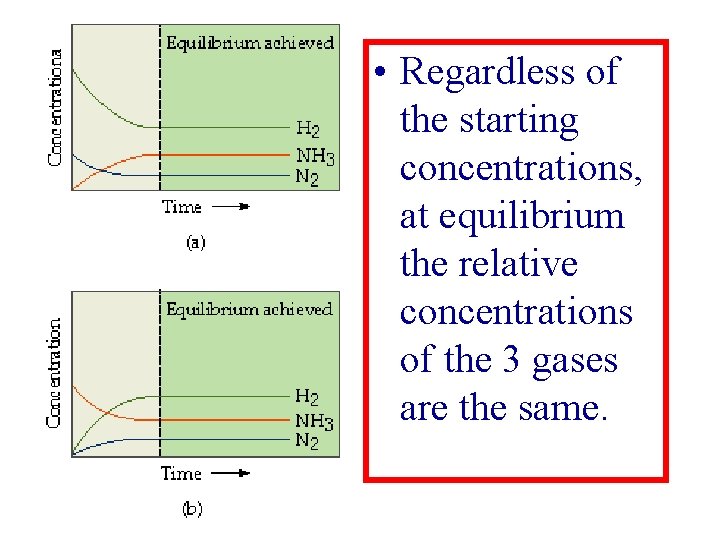
• Regardless of the starting concentrations, at equilibrium the relative concentrations of the 3 gases are the same.

Equilibrium constant for Haber Process • The equilibrium-constant depends only on the stoichiometry (3, 2, 1) of the reaction, not on its mechanism.

The following equilibrium process has been studied at 230ºC: 2 NO(g)+ O 2(g) 2 NO 2(g) In one experiment the concentration of the reacting species at equilibrium are found to be [NO]=0. 0542 M [O 2]=0. 127 M [NO 2]=15. 5 M. Calculate Kc of the reaction at this temperature.
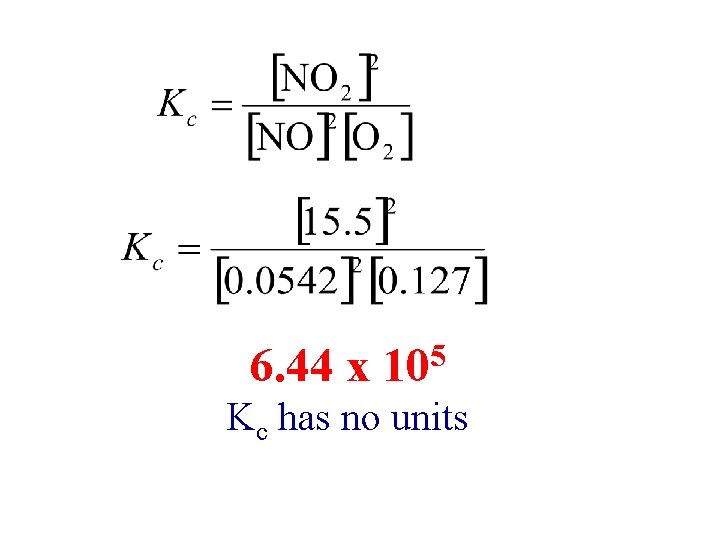
6. 44 x 5 10 Kc has no units

Kc in terms of pressure Example: a. A + b. B c. C + d. D PA is partial pressure of A in atm, PB is the partial pressure of B, etc.

Example The reaction forming nitrosyl chloride is 2 NO (g) + Cl 2 (g) 2 NOCl (g) If the pressures at equilibrium are PNOCl = 1. 2 atm, PNO=5. 0 x 10 -2 atm, and PCl 2 = 3. 0 x 10 -1 atm, what is the value of Kp? Kp = PNOCl 2 = (1. 2)2 = 1. 9 x 103 atm-1 (PNO )2(PCl 2) (5. 0 x 10 -2)2(3. 0 x 10 -1)

• Values for Kc and Kp are usually different. • It is possible to calculate one from the other. • n is the change in the number of moles of gas. Δn = nproducts - nreactants
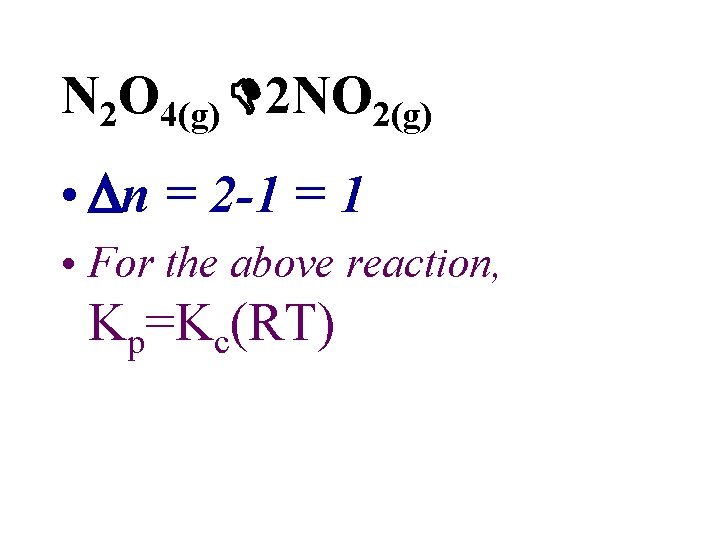
N 2 O 4(g) 2 NO 2(g) • n = 2 -1 = 1 • For the above reaction, Kp=Kc(RT)

For the equilibrium 2 SO 3(g) 2 SO 2(g) + O 2(g) at temperature 1000 K, Kc has a value 4. 08 x 10 -3. Calculate the value for Kp. Kp = 4. 08 x 10 -3 (. 0821 x 1000)1 0. 335

A 3: 1 starting mixture of hydrogen, H 2, and Nitrogen, N 2 comes to equilibrium at 500 °C. The mixture at equilibrium is 3. 506% NH 3, 96. 143% N 2, and 0. 3506% H 2 by volume. The total pressure in the reaction vessel was 50. 0 atm. What is the value of Kp and Kc for the reaction? N 2 + 3 H 2 2 NH 3 Hint: Use Dalton’s Law of partial pressures to determine the partial pressure of each component in the reaction. PA = X A PT Since we know we have 100%, Partial Pressure = (% of substance /100 X total pressure)

P NH 3 = 0. 03506 x 50. 0 atm = 1. 75 atm P N 2 = 0. 96143 x 50. 0 atm = 48. 1 atm P H 2 Kp = = 0. 003506 x 50. 0 atm = 0. 175 atm (P NH 3)2 (P N 2) (P H 2)3 = (1. 75)2 (48. 1) (0. 175)3 = 11. 9 Δn 1 Kc = Kp RT ( ) Kc = K p ( 1 0. 0821 x 773 ) -2 = 4. 79 x 104

Homework #28 • KP = K(RT)Δn, • K = Δn = 2 - 3 = -1; • K = 0. 25 × (0. 08206 × 1100) = 23 28

• When Kc is very large the numerator is much larger than the denominator. • The equilibrium lies to the right, or to the product side of the equation (mostly products formed). K = [products] [reactants]

• When the equilibrium constant is very small the equilibrium lies to the left, or the reactant side (mostly reactants). • K>>1 Products favored. • K<<1 Reactants favored.

• Substances that are in equilibria and are in different phases. Ca. CO 3(s) Ca. O(s)+ CO 2(gas) Equilibrium constant

• The concentration of a pure solid or liquid is a constant. • If a pure solid or pure liquid are involved in a heterogeneous equilibrium, its concentration is not included in the equilibrium-constant expression for the Rx.

• Write the equilibrium expression Kc and Kp for the following reaction: 3 Fe(s)+ 4 H 2 O(g) Fe 3 O 4(s) + 4 H 2(g)

Magnitude of K determines whether reaction will proceed. Using K we can predict: 1. Direction of reaction 2. Calculate concentrations of reactants and products at equilibrium.

Reaction Quotient (Q) • This is used when you are given information about reactants and products when they have not yet reached equilibrium. • Allows us to determine direction reaction WILL go. --may be given concentrations before reaction begins. --may be given concentrations determined during a reaction. • Calculate Q as you do K. N 2(g) + 3 H 2(g) 2 NH 3(g) • Compare Q and K as follows.

Reaction Quotient (Q) and reaction direction • Q=K when system is at equilibrium • Q>K reaction goes to left(reactants) • Q<K reaction goes to right(products)

Direction of a Reaction Step 1: Calculate the reaction quotient, Qc Step 2: Compare Qc to Kc

case 1: Qc > Kc Too much product! Large numerator. case 2: Qc = Kc At equilibrium case 3: Qc < Kc Too much reactants! Large denominator.

N 2(g)+ 3 H 2(g) 2 NH 3(g) • At the start of a reaction, there are 0. 249 mol N 2, 3. 21 x 10 -2 mol H 2, and 6. 42 x 10 -4 mol NH 3 in a 3. 50 L reaction vessel at 200º C. If the equilibrium constant (Kc) for the reaction is 0. 65 at this temperature, decide whether the system is at equilibrium. If it is not, predict which way the net reaction will proceed.

Initial Concentrations
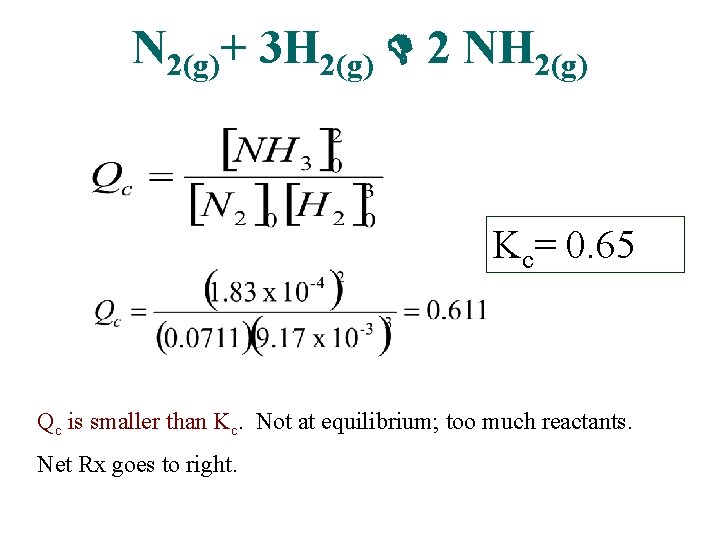
N 2(g)+ 3 H 2(g) 2 NH 2(g) Kc= 0. 65 Qc is smaller than Kc. Not at equilibrium; too much reactants. Net Rx goes to right.

Problem N 2(g) + 3 H 2(g) 2 NH 3(g) In the reaction above, the value of Kc at 500°C is 6. 0 x 10 -2. At some point during the reaction, the concentrations of each material were measured. At this point, the concentrations of each substance were [N 2] = 1. 0 x 10 -5 M, [H 2] = 1. 5 x 10 -3 M, and [NH 3] = 1. 5 x 10 -3 M. Determine the direction that the reaction was most likely to proceed when the measurements were taken. (. 0015)3 = (1. 0 x 10 -5) (1. 5 x 10 -3 )3 Qc=1. 0 x 105 Most likely will go to the left (reactant side)

Extent of reaction Kc >> 1 (any # > 10) Forward rxn goes to near completion Kc << 1 (any # < 0. 1) Reverse rxn goes to near completion

Homework #34 • Determine Q for each reaction, and compare this value to Kp (2. 4 × 103) to determine which direction the reaction shifts to reach equilibrium. Note that, for this reaction, K = Kp since Δn = 0. a. Q = = 2. 2 × 103 Q < Kp so the reaction shifts right to reach equilibrium. 44

Homework #34 Cont. b. Q= = 4. 0 × 103 > Kp Reaction shifts left to reach equilibrium. c. Q= at equilbrium = 2. 4 × 103 = Kp; 45

Calculating Equilibrium Constant “When all Equilibrium Concentrations are not known. ”

• Equilibrium concentrations are often unknown. • If one equilibrium concentration is known, we can use stoichiometry to find the equilibrium concentration of the other species.

Equilibrium Concentrations Initial concentration Change in concentration Equilibrium concentration Called an ICE diagram

When 2. 00 mole each of hydrogen (H 2) and (I 2) are mixed in an evacuated 1. 00 L vessel, 3. 50 moles of HI are produced. What is the value of the equilibrium constant, Kc? Procedure for calculating equilibrium concentrations. H 2(g) + I 2(g) H 2 2 HI(g) I 2 2 HI Start Δ Finish 1) First, we design our table so there is a column for each component in the equation, and rows for start, Δ, and finish.

H 2(g) + I 2(g) Initital 2 HI(g) H 2 I 2 2 HI 2. 0 0 Change +3. 50 Equilibrium 3. 50 2) Next, we fill in what we know. *Since HI is a product, it is 0 at the start of the reaction. *We also know that at equilibrium, 3. 50 mol of HI exist.

H 2(g) + I 2(g) Start 2 HI(g) H 2 I 2 2 HI 2. 0 0 Δ +3. 50 Finish 3. 50 3) We now use stoichiometry to fill in the rest of the table.

H 2(g) + I 2(g) 2 HI(g) H 2 I 2 2 HI Start 2. 0 0 Δ -1. 75 Finish 3. 50 mol HI +3. 50 1 mol H 2 2 mol HI = 1. 75 mol H 2 *It is negative because it is being used up in the reaction.

H 2(g) + I 2(g) 2 HI(g) H 2 I 2 2 HI Start 2. 0 0 Δ -1. 75 +3. 50 Finish 0. 25 3. 50 mol HI 1 mol I 2 2 mol HI = 1. 75 mol I 2 *Now we can fill in the “finish” column.
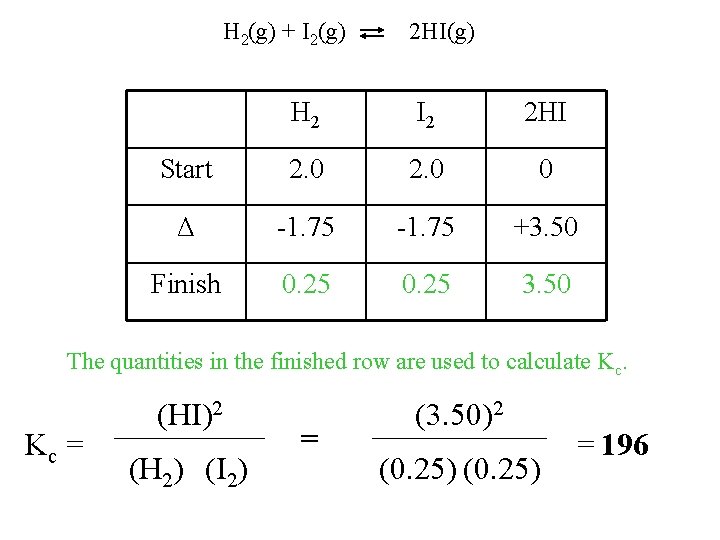
H 2(g) + I 2(g) 2 HI(g) H 2 I 2 2 HI Start 2. 0 0 Δ -1. 75 +3. 50 Finish 0. 25 3. 50 The quantities in the finished row are used to calculate Kc. Kc = (HI)2 (H 2) (I 2) = (3. 50)2 (0. 25) = 196

You try this one! Nitric oxide gas, NO, and oxygen gas, O 2, react to form the poisonous gas nitrogen dioxide, NO 2, in the reaction shown below: 2 NO(g) + O 2(g) 2 NO 2(g) 10. 0 moles of NO and 6. 00 moles of O 2 are placed into an evacuated 1. 00 L vessel, where they begin to react. At equilibrium, there are 8. 80 moles of NO 2 present. Calculate the value of Kc, assuming that the temperature remains constant throughout the reaction.

10. 0 moles of NO and 6. 00 moles of O 2 are placed into an evacuated 1. 00 L vessel, where they begin to react. At equilibrium, there are 8. 80 moles of NO 2 present. Calculate the value of Kc, assuming that the temperature remains constant throughout the reaction. 2 NO(g) + O 2(g) Kc= 2 NO 2(g) 2 NO O 2 2 NO 2 Initial 10 6 0 Change 8. 8 4. 4 8. 8 Equilibrium 1. 2 1. 6 8. 8 (NO 2)2 (O 2) (NO)2 = 33. 6

When 3. 0 mol of I 2 and 4. 0 mol of Br 2 are placed in a 2. 0 L reactor at 150 o. C, the following reaction occurs until equilibrium is reached: I 2(g) + Br 2(g) 2 IBr(g) Chemical analysis then shows that the reactor contains 3. 2 mol of IBr. What is the value of the equilibrium constant Kc for the reaction? Consider M since we have a 2. 0 L vessel.

3. 0 mol of I 2 and 4. 0 mol of Br 2 are placed in a 2. 0 L reactor at 150 o. C. Chemical analysis then shows that the reactor contains 3. 2 mol of IBr. What is the value of the equilibrium constant Kc for the reaction? I 2(g) + Br 2(g) 2 IBr(g) Initial I 2 Br 2 2 IBr 3 4 0 Change 3. 2 Equilibrium 3. 2 Kc=3. 0

If you are dealing with the pressures of gases at equilibrium, you will be given: moles, temperature, and volume. is the key to calculating the pressure of each gas at equilibrium. Stoichiometric equivalents may also be used.

Example: 2 SO 3(g) 2 SO 2(g) + O 2(g) Initially, 0. 01219 moles of SO 3 gas in a 2. 0 L vessel are heated to a temperature of 1000 K where it reaches equilibrium. At equilibrium the vessel is found to contain 0. 00487 moles of SO 3 gas. A) Calculate the equilibrium partial pressures of SO 2(g) and O 2(g). B) Calculate Keq.

1) We must first calculate the pressures from the information given in the problem. PV = n. RT P = n. RT/V P =. 01219(0. 8206)(1000) / (2. 0) = 0. 500 atm This is the initial pressure of SO 3 gas. initial Δ finish 2 SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3 2 SO 2 . 500

PV = n. RT P = n. RT/V P =. 00487(0. 8206)(1000) / (2. 0) = 0. 200 atm This is the pressure of SO 3 gas at equilibrium. initial 2 SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3 2 SO 2 . 500 Δ finish . 200
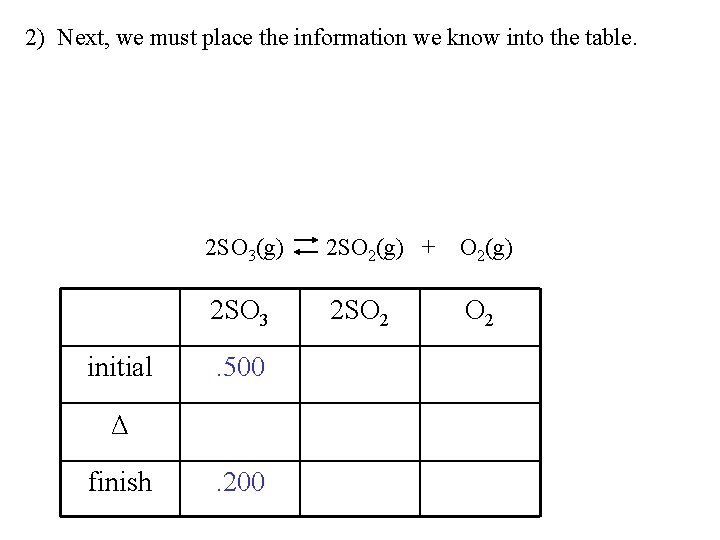
2) Next, we must place the information we know into the table. 2 SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3 2 SO 2 initial . 500 0 0 Δ -. 300 finish . 200

2) Now we use stoichiometric relationships to fill in the rest. Notice the 2: 2 ratio of SO 3 and SO 2. Notice the 2: 1 ratio of SO 3 and O 2. 2 SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3 2 SO 2 initial . 500 0 0 Δ -. 300 +. 150 finish . 200

3) Now we add the columns. The finish row gives us the partial pressures of each gas at equilibrium. 2 SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3 2 SO 2 initial . 500 0 0 Δ -. 300 +. 150 finish . 200 . 300 . 150

4) Write equilibrium constant expression and solve for Keq. 2 SO 3(g) 2 SO 2(g) + O 2(g) 2 SO 3 2 SO 2 initial . 500 0 0 Δ -. 300 +. 150 finish . 200 . 300 . 150

Homework #42 2 NH 3(g) Initial Change Equil. 2 M -1 M 1 M K= ⇌ N 2(g) + 3 H 2(g) 0 0 → +0. 5 M +1. 5 M 0. 5 M 1. 5 M 67
![Homework #42 Cont. [N 2] = 0. 50 M; [H 2] = 1. 5 Homework #42 Cont. [N 2] = 0. 50 M; [H 2] = 1. 5](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-68.jpg)
Homework #42 Cont. [N 2] = 0. 50 M; [H 2] = 1. 5 M [NH 3]= 1 M K= = 1. 7 68

• Often only Kc and beginning concentrations are known. • Must be able to solve for equilibrium concentrations.

Problem H 2(g) + I 2(g) 2 HI(g) • A mixture of 0. 500 mol H 2 and 0. 500 mol I 2 was placed in a 1. 00 L stainless steel flask at 430ºC. Calculate the concentrations of H 2, I 2, and HI at equilibrium. The equilibrium constant Kc for the reaction is 54. 3 at this temperature.
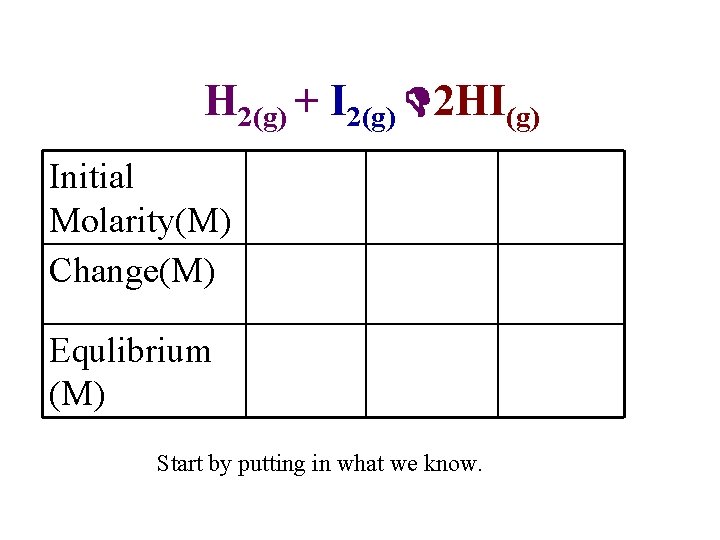
H 2(g) + I 2(g) 2 HI(g) Initial 0. 500 Molarity(M) Change(M) -x Equlibrium (M) 0. 500 0. 00 -x +2 x 0. 500 -x Start by putting in what we know. 2 x

H 2(g) + I 2(g) 2 HI(g) H 2 Start (M) Δ (M) I 2 0. 500 M -x Finish (M) 0. 500 -x 2 HI 0 -x 2 x 0. 500 -x 2 x

Must now plug x back into “finish” row to calculate actual concentrations.
![At equilibrium the concentrations are: • • [H 2]= (0. 500 -0. 393) = At equilibrium the concentrations are: • • [H 2]= (0. 500 -0. 393) =](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-74.jpg)
At equilibrium the concentrations are: • • [H 2]= (0. 500 -0. 393) = 0. 107 M [I 2] = (0. 500 -0. 393) = 0. 107 M [HI] = 2(0. 393) = 0. 786 M Answers can be checked by calculating Kc using the equilibrium concentrations.

Homework #46 Initial Change Equil. H 2(g) + I 2(g) ⇌ 2 HI(g) K = 100. 1. 00 M -x -x → +2 x 1. 00 – x 1. 00 + 2 x K = 100. = ; Taking the square root of both sides: 10. 0 = 75

46 Cont. 10. 0 = 10. 0 - 10. 0 x = 1. 00 + 2 x, 12. 0 x = 9. 0, x = 0. 75 M [H 2] = [I 2] = 1. 00 - 0. 75 = 0. 25 M; [HI] = 1. 00 + 2(0. 75) = 2. 50 M 76

Shouldn’t need for AP exam but you never know!! H 2(g) + I 2(g) 2 HI(g) • For the same reaction and temperature, suppose the initial concentrations of HI, H 2, and I 2 are 0. 0224 M, 0. 00623 M, & 0. 00414 M respectively. Calculate the concentrations at equilibrium.
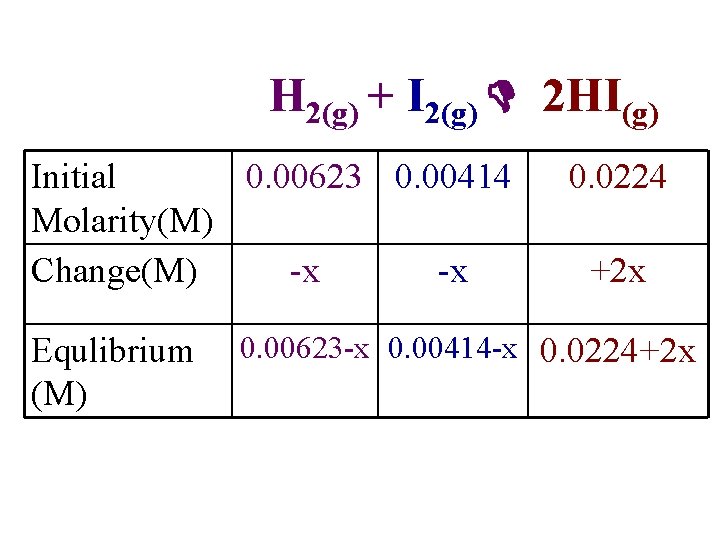
H 2(g) + I 2(g) 2 HI(g) Initial 0. 00623 0. 00414 Molarity(M) Change(M) -x -x Equlibrium (M) 0. 0224 +2 x 0. 00623 -x 0. 00414 -x 0. 0224+2 x
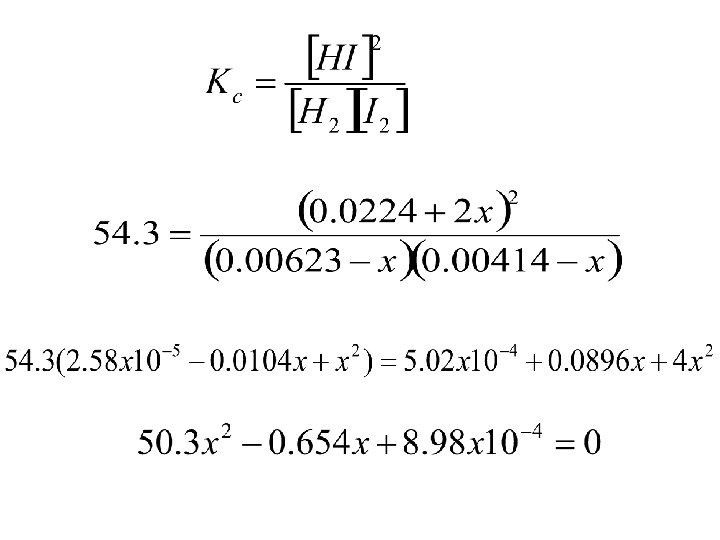

• This is a quadratic equation of the form: ax 2 + bx + c = 0 The solution is: a= 50. 3; b = -0. 654; c = 8. 98 x 10 -4 X = 0. 0114 M or x = 0. 00156 M The first solution in impossible since it says more I 2 reacted
![At equilibrium the concentrations are: • [H 2]= (0. 00623 -0. 00156) = 0. At equilibrium the concentrations are: • [H 2]= (0. 00623 -0. 00156) = 0.](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-81.jpg)
At equilibrium the concentrations are: • [H 2]= (0. 00623 -0. 00156) = 0. 00467 M • [I 2] = (0. 00414 -0. 00156) = 0. 00258 M • [HI] = (0. 0224+2(0. 00156)= 0. 0255 M

5% Rule • Some problems have an x that is really small in comparison to the equilibrium number. • Usually happens when K is very small also • Assume to ignore the x in the equilibrium concentration to avoid using quadratic equation. • Is a valid assumption when the x value compared to the equilibrium concentration is less than 5% 82

Example of 5% rule • #52 N 2 O 4(g) ⇌ 2 NO 2(g) Initial 1. 0 mol/10. 0 L 0 Change -x +2 x Equil. 0. 10 – x 2 x K= = 4. 0 × 10 -7 83

#52 Continued K= = = 4. 0 × 10 -7; Because K has a small value, assume that x is small compared to 0. 10 so that 0. 10 - x ≈ 0. 10. Solving: 4. 0 × ≈ , 4 x 2 = 4. 0 × 10 -7 , x = 1. 0 × 10 -4 M • Checking the assumption by the 5% rule: × 100 = 0. 10% • Because this number is less than 5%, we will say that the assumption is valid. 84
![#52 Final Answer • [N 2 O 4] = 0. 10 - 1. 0 #52 Final Answer • [N 2 O 4] = 0. 10 - 1. 0](http://slidetodoc.com/presentation_image_h2/a0a9674735b22a8b11b133b2c32e9230/image-85.jpg)
#52 Final Answer • [N 2 O 4] = 0. 10 - 1. 0 × 10 -4 = 0. 10 M; • [NO 2] = 2 x = 2(1. 0 × 10 -4) = 2. 0 × 10 -4 M 85

• If a stress is applied to a system in a state of equilibrium, the equilibrium will shift so to relieve that stress. Stress could be a change in: • temperature • pressure • concentration of one component.

Effects of Changes on the System 1. Concentration: The system will shift away from the added component or toward a removed component. 2. Temperature: K will change depending upon the type of reaction. Since exothermic produces heat, it will shift away from added, endo the opposite.

In Reactant or Product Concentration • Adding a substance to a system at equilibrium will shift the reaction by consuming some of the added item. • Removing a substance from a system at equilibrium will shift the reaction to form more of the removed substance.

Effects of Changes on the System (continued) 3. Pressure: a. Addition of inert gas does not affect the equilibrium position. b. Decreasing the volume shifts the equilibrium toward the side with fewer moles.

Effects of Volume & Pressure • Reducing the volume of a gas equilibrium system (at constant T) will cause a shift in the direction that reduces the number of moles of gas in the system. N 2 O 4 2 NO 2 • Reducing volume favors which direction? • Shift to left.
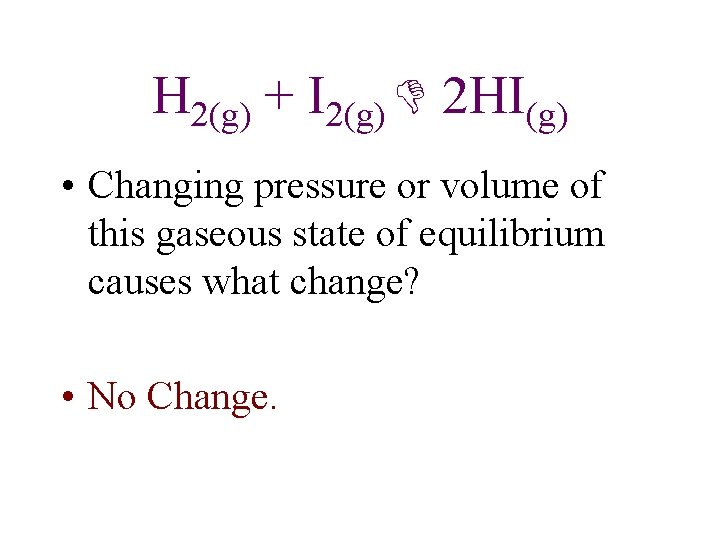
H 2(g) + I 2(g) 2 HI(g) • Changing pressure or volume of this gaseous state of equilibrium causes what change? • No Change.

• Equilibrium constant values (Keq) always change with temperature change, but stays the same when pressure or concentration is changed. • Enthalpy of the reaction plays a major roll in how heat effects the equilibrium.

Le Châtelier’s Principle Effect of Temperature Changes • The equilibrium constant is temperature dependent. • For an endothermic reaction, H > 0 and heat can be considered as a reactant. • For an exothermic reaction, H < 0 and heat can be considered as a product. • Adding heat (i. e. heating the vessel) favors away from the increase: – if H > 0, adding heat favors the forward reaction, – if H < 0, adding heat favors the reverse reaction. 93

When temperature is increased the reaction is shifted in the direction that absorbs heat. • Endothermic: + (shift to rt) Reactants + heat Products • Exothermic: - (shift to left) Reactants Products + heat

Consider the following equilibrium systems, and predict the direction of the net reaction in each case as a result of pressure ( volume) on the system at constant temperature. 2 Pb. S(s) + 3 O 2(g) 2 Pb. O(s) + 2 SO 2(g) Shift to right. PCl 5(g) PCl 3(g)+Cl 2(g) Shift to left. H 2(g) + I 2(g) 2 HI(g) No influence on position of equilibrium.

Consider the following equilibrium process: N 2 F 4(g) 2 NF 2(g) º= 38. 5 k. J Predict the changes in the equilibrium if : a) The reacting mixture is heated at constant pressure. Shift to right. b) NF 2 gas is removed at constant T and Pressure. Shift to right.

Example 13. 15 & 16 • 15—Predict the shift in equilibrium when volume is reduced in each of these(causing increased pressure): a. P 4 (s) +6 Cl 2 (g) 4 PCl 3 (l) Since there are no moles of gas on the right, volume reduction will favor that side. b. PCl 3 (g) + Cl 2 (g) PCl 5 (g) Since there are fewer moles of gas on the right, the equilibrium will shift that direction. c. PCl 3 (g) + 3 NH 3 (g) P(NH 2)3 (g) + 3 HCl (g) Since there are 4 moles on either side of the equation, there is no shift due to pressure change. 16—For each of these reactions, predict the shift of equilibrium with an increase in temperature N 2 (g) + O 2 (g) 2 NO (g) Hº= 181 k. J This reaction is endothermic, so heat acts as an added reactant, favoring the products to the right 2 SO 2 (g) + O 2 (g) 2 SO 3 (g) Hº= -198 k. J This reaction is exothermic, heat a product, so it shifts to the left.

Summary of Le. Chatelier Concentration: Equilibruim shifts away from what is added, toward what is removed. Temperature: Equilibrium shifts away from added temperature, depending on type of reaction, Exo-toward reactants Endo-toward products Pressure: Equilibrium shifts toward less molecules when pressure is added

Effects of Catalysts • Catalysts lower the energy barrier between reactant and products. – Ea for both forward and reverse are lowered to the same extent. • Catalysts increase the rate at which equilibrium is reached. • Does NOT change the composition of the equilibrium mixture.

3 O 2(g) 2 O 3(g) º=284 k. J When the above reaction is at equilibrium: 1. What is the effect of pressure by volume? Shift to right. 1. O 2? Shift to right. 2. Temperature? Shift to left. 3. Adding catalyst? No change, catalysts only help increase the rate at which equilibrium is reached.
- Slides: 100