Chemical Equilibrium Chapter 6 SCH 4 U Chapter

Chemical Equilibrium Chapter 6 (SCH 4 U) Chapter 15 (AP)

Chemical Equilibrium • • 15. 1 The Concept of Equilibrium 15. 2 The Equilibrium Constant 15. 3 Interpreting and Working with Equilibrium Constants 15. 4 Heterogeneous Equilibrium 15. 5 Calculating Equilibrium Constants 15. 6 Applications of Equilibrium Constants 15. 7 Le Chatalier Principle
The Concept of Equilibrium

Equilibrium: the extent of a reaction In stoichiometry we talk about theoretical yields, and the many reasons actual yields may be lower. Another critical reason actual yields may be lower is the reversibility of chemical reactions: some reactions may produce only 70% of the product you may calculate they ought to produce. Equilibrium looks at the extent of a chemical reaction.

Equilibrium: is a state in which there are no observable changes as time goes by. Chemical equilibrium is achieved when: • the rates of the forward and reverse reactions are equal and • the concentrations of the reactants and products remain constant Physical equilibrium H 2 O (l) H 2 O (g) Chemical equilibrium N 2 O 4 (g) 2 NO 2 (g) 14. 1

Rate of sale of cookies = Rate of replacing cookies
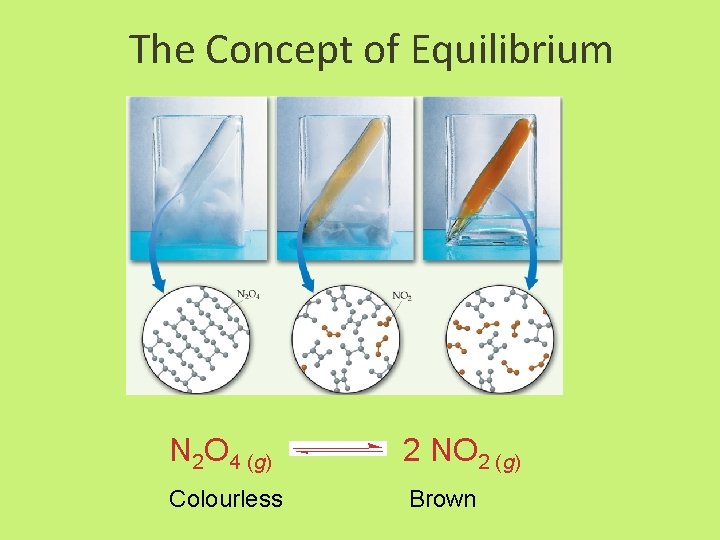
The Concept of Equilibrium N 2 O 4 (g) 2 NO 2 (g) Colourless Brown

Elementary Reactions • Forward Reaction N 2 O 4(g) 2 NO 2 (g) Rate= kf[N 2 O 4] • Reverse Reaction 2 NO 2 N 2 O 4(g) Rate=kr[NO 2]2 At equilibrium, the rate of the forward reactions are equal. kf[N 2 O 4]=kf [NO 2]2 [N 2 O 4] = kf = Equilibrium Constant kf
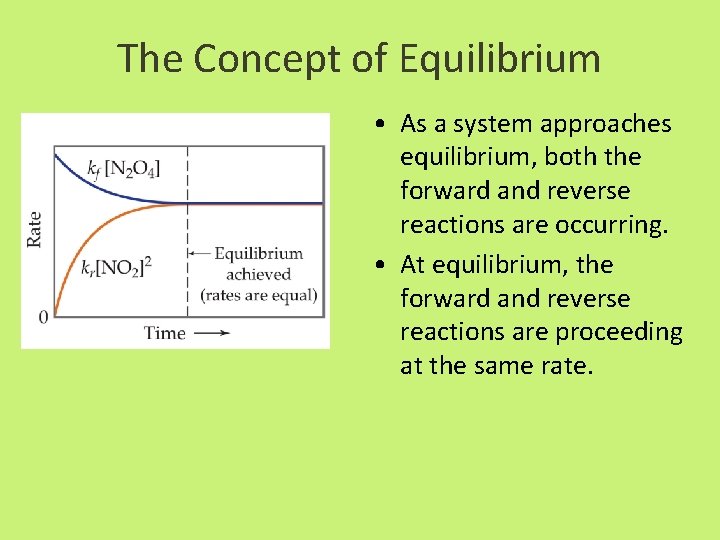
The Concept of Equilibrium • As a system approaches equilibrium, both the forward and reverse reactions are occurring. • At equilibrium, the forward and reverse reactions are proceeding at the same rate.
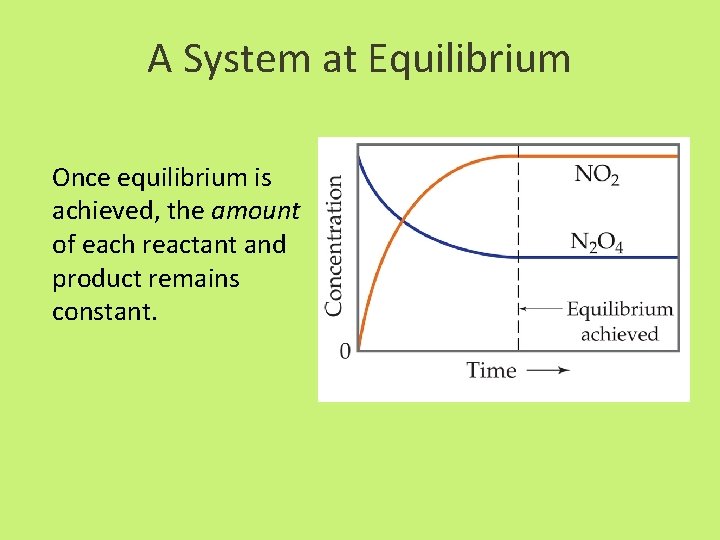
A System at Equilibrium Once equilibrium is achieved, the amount of each reactant and product remains constant.

The Concept of Equilibrium • Consider colorless frozen N 2 O 4. At room temperature, it decomposes to brown NO 2: N 2 O 4(g) 2 NO 2(g). • At some time, the color stops changing and we have a mixture of N 2 O 4 and NO 2. • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO 2 builds up, there is a chance that two NO 2 molecules will collide to form N 2 O 4. – At the beginning of the reaction, there is no NO 2 so the reverse reaction (2 NO 2(g) N 2 O 4(g)) does not occur.

The Concept of Equilibrium • As the substance warms it begins to decompose: N 2 O 4(g) 2 NO 2(g) • When enough NO 2 is formed, it can react to form N 2 O 4: 2 NO 2(g) N 2 O 4(g). • At equilibrium, as much N 2 O 4 reacts to form NO 2 as NO 2 reacts to re-form N 2 O 4 • The double arrow implies the process is dynamic.
![The Concept of Equilibrium As the reaction progresses – [A] decreases to a constant, The Concept of Equilibrium As the reaction progresses – [A] decreases to a constant,](http://slidetodoc.com/presentation_image_h2/877f8424abd267bee14b8e6385ddc2c4/image-13.jpg)
The Concept of Equilibrium As the reaction progresses – [A] decreases to a constant, – [B] increases from zero to a constant. – When [A] and [B] are constant, equilibrium is achieved.
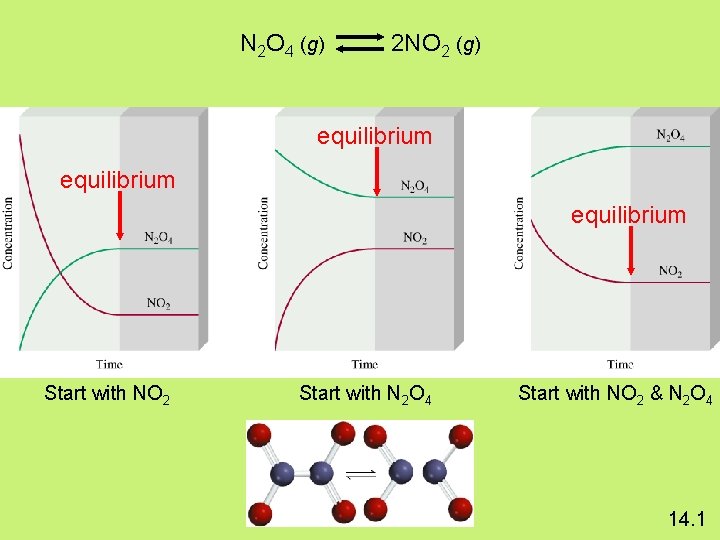
N 2 O 4 (g) 2 NO 2 (g) equilibrium Start with NO 2 Start with N 2 O 4 Start with NO 2 & N 2 O 4 14. 1
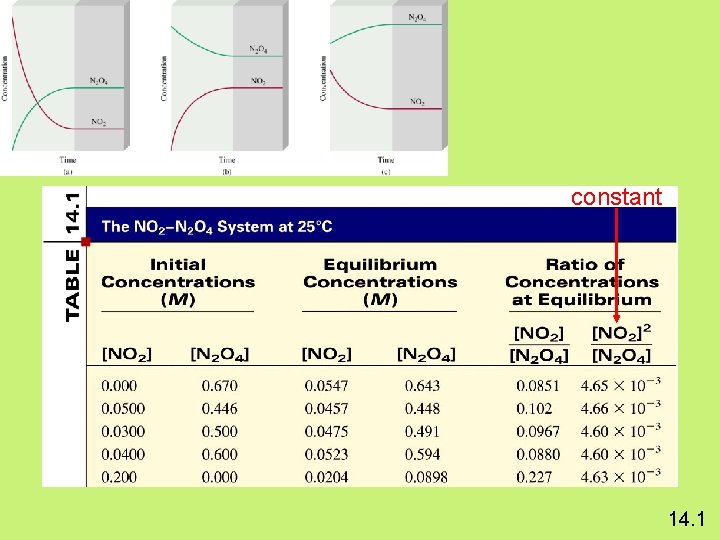
constant 14. 1
The Haber Process N 2(g) + 3 H 2(g) 2 NH 3(g) • Catalyst, pressure of several hundred atm, and several hundred degree Celsius temperature. • Closed system, meaning that N 2 and H 2 cannot be completely consumed.

Recap from Last Class Conditions that Apply to All Equilibrium System : • Equilibrium is achieved in a reversible process when the rates of opposing changes are equal • Observable (macroscopic) properties of a system at equilibrium are constant. • Equilibrium can only be reached in a closed system • Equilibrium can be approached from any direction. http: //www. mhhe. com/physsci/chemistry/animations/chan g_7 e_esp/kim 2 s 2_5. swf

Thermodynamics & Equilibrium • Can you predict what will happen if you - let go of a pencil you are holding above the ground ? - add drops of food coloring to water? - begin to burn a piece of paper in air? - leave an iron nail outside over winter? These are all examples of favorable (spontaneous) changes. That is, changes that have a natural tendency to happen under certain conditions.

Spontaneous Reaction • spontaneous reaction = one that, given the necessary activation energy, proceeds without continuous outside assistance • Example: a sparkler – Needs light from a flame for activation – Once lit, the available fuel combusts quickly and completely, releasing large amounts of energy as heat and light

Enthalpy & Entropy Changes Together Determine Spontaneity • Endothermic = + ∆H • Exothermic = - ∆H • Exothermic reactions tend to proceed spontaneously a) Ba(OH)2(aq) + 2 NH 4 NO 3(aq) Ba(NO 3)2(aq) + 2 NH 4 OH(l) b) NH 4 NO 3(s) NH 4 +(aq) + NO 3 -(aq)

Entropy • enthalpy is not the only factor that determines whether a chemical or physical change occurs spontaneously • entropy, S = a measure of the randomness or disorder of a system, or the surroundings
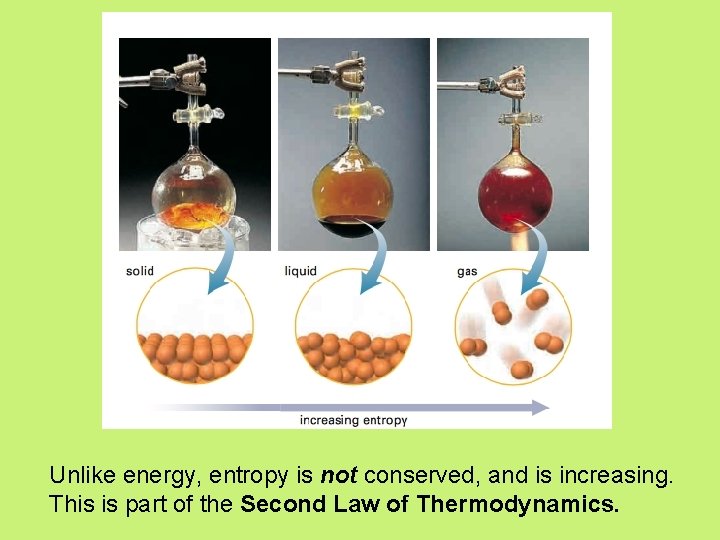
Unlike energy, entropy is not conserved, and is increasing. This is part of the Second Law of Thermodynamics.

Entropy • Increase entropy = increase randomness = +∆S • When entropy increases in a reaction, the entropy of the products, Sproducts, is greater than the entropy of the reactants, Sreactants, yielding an overall positive change in entropy, S.

Entropy • decrease entropy = decrease randomness = -∆S • When entropy decreases in a reaction, the entropy of the products, Sproducts, is less than the entropy of the reactants, Sreactants, yielding an overall negative change in entropy, S.
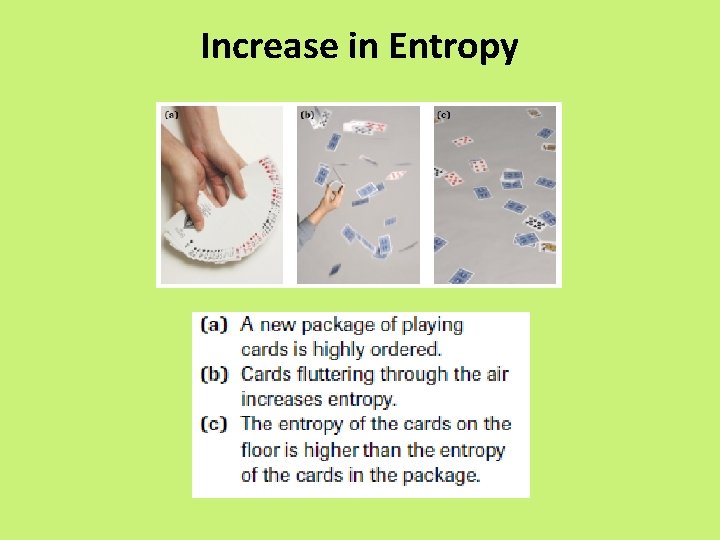
Increase in Entropy
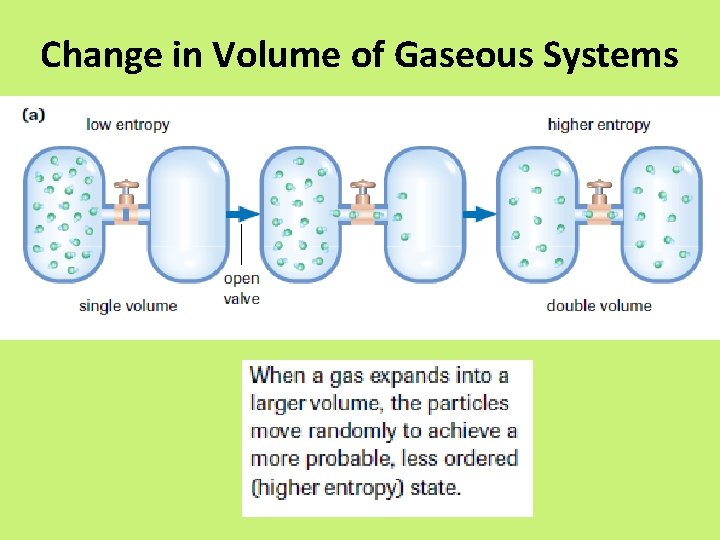
Change in Volume of Gaseous Systems
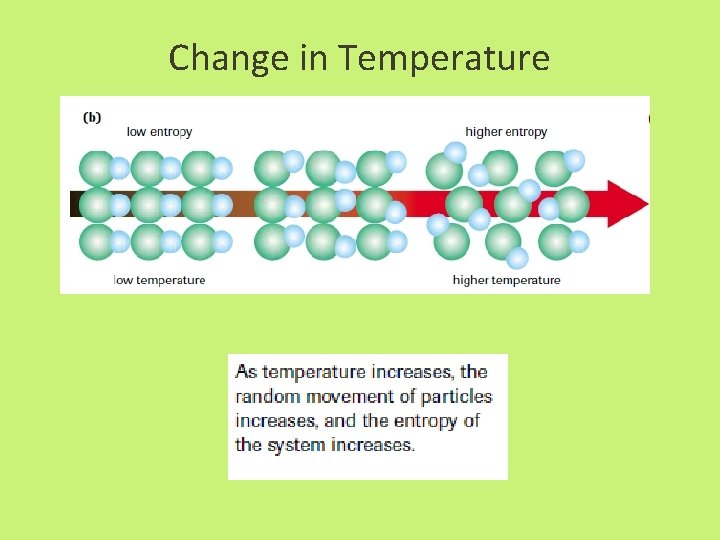
Change in Temperature
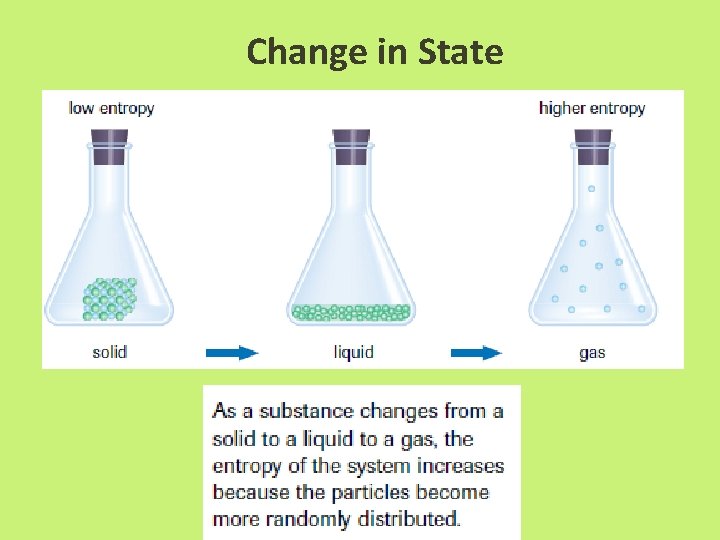
Change in State

In Chemical Reactions…

Enthalpy, Entropy, and Spontaneous Change • Changes in the enthalpy, ∆H, and entropy, ∆S, of a system help us to predict whether a change will occur spontaneously • Exothermic reactions (-∆H) involving an increase in entropy (+∆S) occur spontaneously, because both changes are favoured • Endothermic reactions (+∆H) involving a decrease in entropy (-∆S) are not spontaneous because neither change is favoured

Enthalpy, Entropy, and Spontaneous Change • But what happens in cases where the energy change is exothermic (favoured) and the entropy decreases (not favoured)? • Or when the energy change is endothermic (not favoured) but entropy increases (favoured)? • In these situations, the temperature at which the change occurs becomes an important consideration as well as free energy

Free Energy • free energy (or Gibbs free energy), G = energy that is available to do useful work • In general, a change at constant temperature and pressure will occur spontaneously if it is accompanied by a decrease in Gibbs free energy, G -∆G = spontaneous +∆G = nonspontaneous

Second Law of Thermodynamics “Law of Entropy” • all changes that occur in the universe. All changes, whether spontaneous or not, are accompanied by an increase in the entropy (overall disorder) of the universe Mathematically, Suniverse > 0

Second Law of Thermodynamics • a system’s entropy, Ssystem, can decrease (the system becomes more ordered), so long as there is a larger increase in the entropy of the surroundings, Ssurroundings, so that the overall entropy change, Suniverse, is positive.

Predicting Spontaneity • The spontaneity of any reaction carried out at constant temperature and pressure can be predicted by calculating the value of G using the following equation, called the Gibbs-Helmholtz equation:
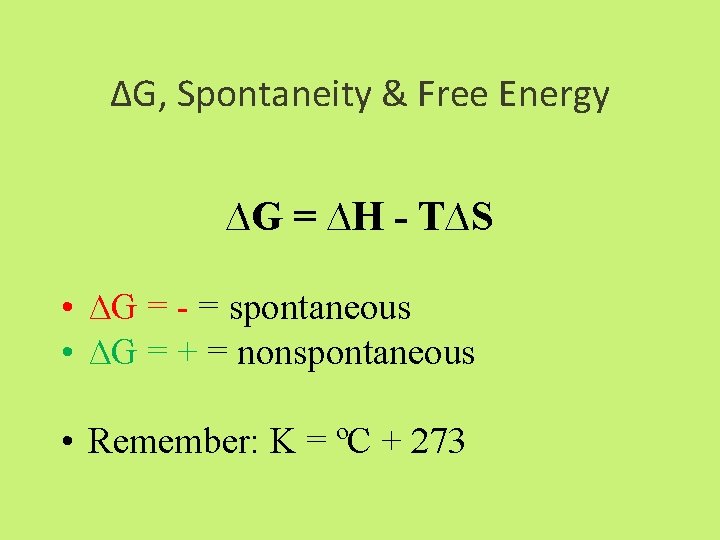
∆G, Spontaneity & Free Energy ∆G = ∆H - T∆S • ∆G = - = spontaneous • ∆G = + = nonspontaneous • Remember: K = ºC + 273
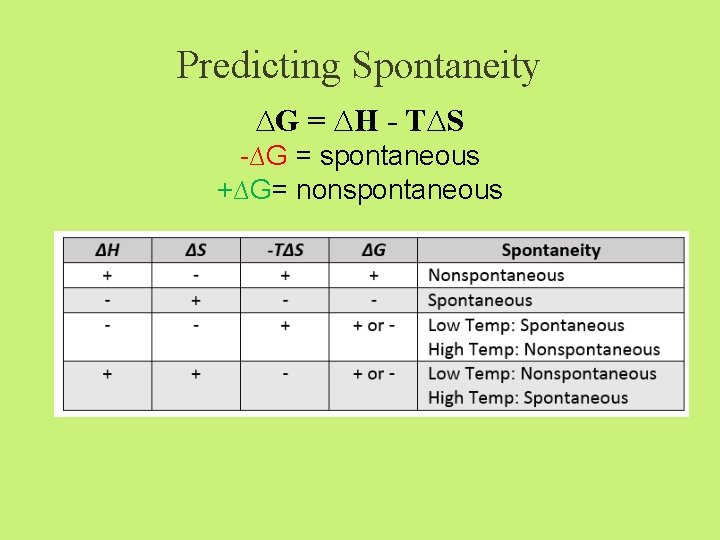
Predicting Spontaneity ∆G = ∆H - T∆S -∆G = spontaneous +∆G= nonspontaneous

When a maximum state of disorder is reached- we say that a chemical system is at equilibrium. Therefore, equilibrium occurs when a system has reached maximum entropy

The Equilibrium Constant • No matter the starting composition of reactants and products, the same ratio of concentrations is achieved at equilibrium. • For a general reaction the equilibrium constant expression is where Kc is the equilibrium constant.
![N 2 O 4 (g) K= 2 NO 2 (g) [NO 2]2 [N 2 N 2 O 4 (g) K= 2 NO 2 (g) [NO 2]2 [N 2](http://slidetodoc.com/presentation_image_h2/877f8424abd267bee14b8e6385ddc2c4/image-40.jpg)
N 2 O 4 (g) K= 2 NO 2 (g) [NO 2]2 [N 2 O 4] = 4. 63 x 10 -3 a. A + b. B K= c. C + d. D [C]c[D]d Law of Mass Action [A]a[B]b Equilibrium Will K >> 1 Lie to the right Favor products K << 1 Lie to the left Favor reactants 14. 1

Homogenous equilibrium applies to reactions in which all reacting species are in the same phase. N 2 O 4 (g) 2 NO 2 (g) Heterogenous equilibrium applies to reactions in which all reacting species in different phases. Ca. CO 3 (s) Ca. O (s) + CO 2 (g) 14. 2
![Homogeneous Equilibrium CH 3 COOH (aq) + H 2 O (l) [CH 3 COO-][H Homogeneous Equilibrium CH 3 COOH (aq) + H 2 O (l) [CH 3 COO-][H](http://slidetodoc.com/presentation_image_h2/877f8424abd267bee14b8e6385ddc2c4/image-42.jpg)
Homogeneous Equilibrium CH 3 COOH (aq) + H 2 O (l) [CH 3 COO-][H 3 O+] Kc = [CH 3 COOH][H 2 O] CH 3 COO- (aq) + H 3 O+ (aq) [H 2 O] = constant [CH 3 COO-][H 3 O+] Kc = [CH 3 COOH] General practice not to include units for the equilibrium constant.

Writing Equilibrium Constant Expressions • The concentrations of the reacting species in the condensed phase are expressed in M. In the gaseous phase, the concentrations can be expressed in M or in atm. • The concentrations of pure solids, pure liquids and solvents do not appear in the equilibrium constant expressions. • The equilibrium constant is a dimensionless quantity. • In quoting a value for the equilibrium constant, you must specify the balanced equation and the temperature. • If a reaction can be expressed as a sum of two or more reactions, the equilibrium constant for the overall reaction is given by the product of the equilibrium constants of the individual reactions. 14. 2

Practice Problems
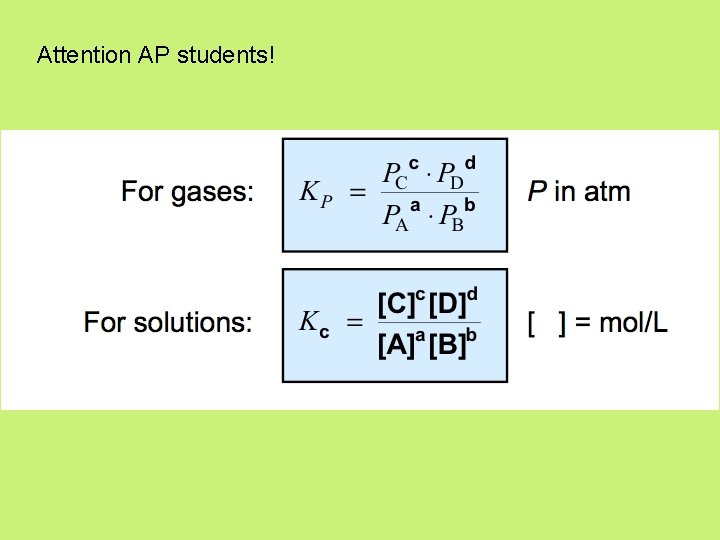
Attention AP students!


Kc vs. Kp (AP) • Kc is generally different in value than Kp, however you can calculate one from the other. Kp= Kc (RT)Δn • Δn = (moles of gaseous product) – (moles of gaseous reactant )

Practice Problem(AP) In the synthesis of ammonia from nitrogen and hydrogen, N 2(g) + 3 H 2 (g) 2 NH 3 (g) Kc= 9. 60 at 300°C. Calculate the Kp for the reaction.

Calculating an Equilibrium Constant

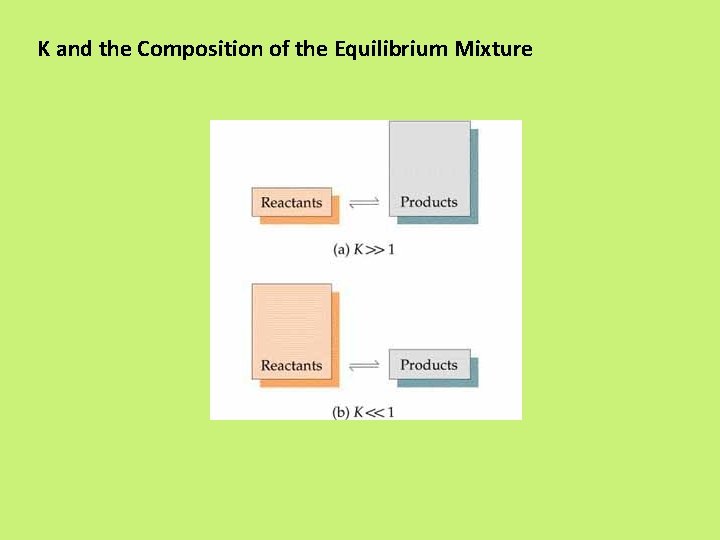
K and the Composition of the Equilibrium Mixture

Measuring Equilibrium Concentrations
- Slides: 52