Chemical Equilibrium Chapter 14 Chemical Equilibrium 14 1

Chemical Equilibrium

Chapter 14 Chemical Equilibrium • 14. 1 the concept of equilibrium and the equilibrium constant • 14. 4 writing equilibrium constant expression • 14. 4 what does the equilibrium constant tell us • 14. 5 factors that effect chemical equilibrium

The Concept of Equilibrium and the Equilibrium Constant q Few reaction are proceed in one direction. q Most reactions are reversible. q Equilibrium is a state in which there are no observable changes as time goes by. q Chemical equilibrium: is achieved when: qthe rates of the forward and reverse reactions are equal qand the concentrations of the reactants and products remain constant

The Concept of Equilibrium and the Equilibrium Constant 1. Physical Equilibrium between two phases of the same substances 2. Chemical Equilibrium between two or more different chemical substances

The Equilibrium Constant q A, b, c, & d: are the stoichiometric coefficients for A, B, C , & D q K is the equilibrium constant. Concentration of products Equilibrium constant Concentration of reactants • [A], [B], etc. are the equilibrium concentrations • K ≈ [products] / [reactants] • K >> 1 ; favors products >>> Lie to the right • K << 1 favors reactants >>> Lie to the left • K ≈ 1: roughly equal concentration of reactants and products ﺛﺎﺑﺖ ﺍﻻﺗﺰﺍﻥ = ﻛﺴﺮ ﻫﻮ ﻧﺎﺗﺞ ﺿﺮﺏ : ﺍﻟﺒﺴﻂ ﺗﺮﻛﻴﺰ ﺍﻟﻨﻮﺍﺗﺞ ﻛﻞ ﻣﺮﻓﻮﻉ ﺍﻟﻰ ﺍﺱ ﻳﺴﺎﻭﻱ ﻋﺪﺩ ﻣﻮﻻﺗﻪ ﻓﻲ ﻣﻌﺎﺩﻟﺔ ﺍﻟﺘﻔﺎﻋﻞ ﺍﻟﻤﻮﺯﻭﻧﻪ ﻫﻮ ﻧﺎﺗﺞ ﺿﺮﺏ : ﻭﺍﻟﻤﻘﺎﻡ ﺗﺮﻛﻴﺰ ﺍﻟﻤﺘﻔﺎﻋﻼﺕ ﻛﻞ ﻣﺮﻓﻮﻉ ﺍﻟﻰ ﺍﺱ ﻳﺴﺎﻭﻱ ﻋﺪﺩ ﻣﻮﻻﺗﻪ ﻓﻲ ﻣﻌﺎﺩﻟﺔ ﺍﻟﺘﻔﺎﻋﻞ ﺍﻟﻤﻮﺯﻭﻧﻪ

The Equilibrium Constant • K is a constant at a given temperature • Solids and pure liquid drop out of the expression & water drops out when the solvent is water • K has no unit

Writing Equilibrium Constant Expressions n Homogeneous Equilibrium: all species are in the same phase n Example: homogenous gas-phase equilibrium of the dissociation of N 2 O 4, n. K is given by: Concentration Units Pressure Units

Writing Equilibrium Constant Expressions n Relationship between Kc & Kp : ¨ Kc ≠ Kp ¨ ∆n = moles of gases products – moles of gases reactants ¨ Kc = Kp, when ∆n = 0

Example 14. 1: Write expressions for Kc, and Kp if applicable, for the following reversible reactions at equilibrium: • Solution: No gases → Only Kc: Not written (neglected) Homogeneous

Homogeneous Solution: Gases → Kp & Kc can be written, as follows:
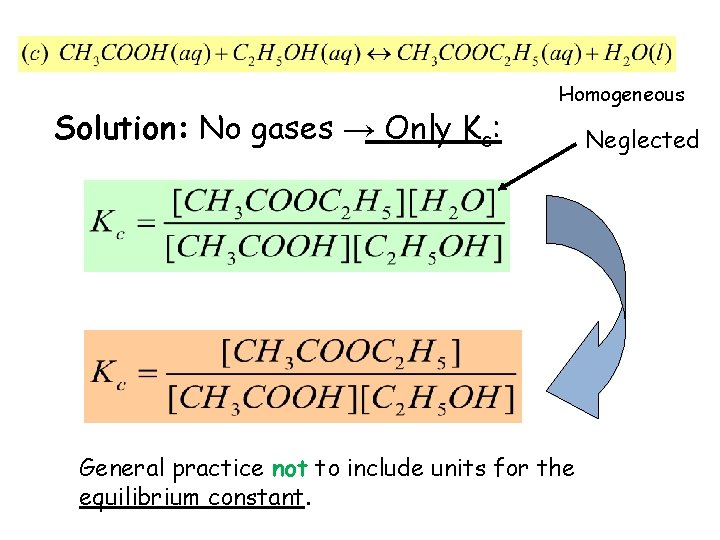
Solution: No gases → Only Kc: Homogeneous General practice not to include units for the equilibrium constant. Neglected

Example 14. 2: The following equilibrium process has been studied at 230 o. C: Homogeneous In one experiment, the concentrations of the reacting species at equilibrium are found to be [NO] = 0. 0542 M, [O 2] = 0. 127 M, and [NO 2] = 15. 5 M. Calculate the equilibrium constant (Kc) of this reaction at this temperature. Solution: Large magnitude favors products >>> Lie to the right

Example 14. 3: The equilibrium constant Kp for the reaction Homogeneous is 158 at 1000 K. What is the equilibrium partial pressure of O 2 if the PNO 2 = 0. 400 atm and PNO = 0. 270 atm? Solution: 2 2 2

Example 14. 4: Methanol (CH 3 OH) is manufactured industrially by the reaction: Homogeneous The equilibrium constant (Kc) for the reaction is 10. 5 at 220 o. C. What is the value of Kp at this temperature? Solution:

Writing Equilibrium Constant Expressions n Heterogeneous Equilibrium: species are of different phases. [Ca. CO 3] = constant Neglected [Ca. O] = constant Kc = [CO 2] The concentration of solids and pure liquids are not included in the expression for the equilibrium constant.

Example 14. 5: Write the equilibrium constant expression Kc, and Kp if applicable, for each of the following heterogeneous systems: Heterogeneous Solution: All are gases → Kp Solid neglected
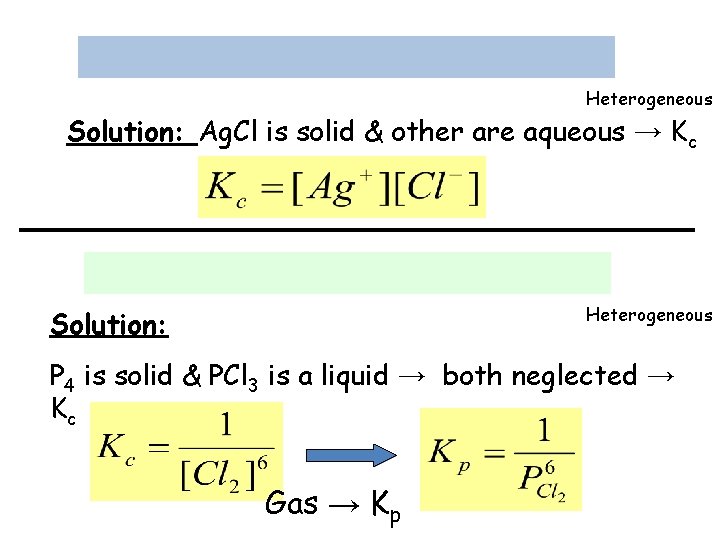
Heterogeneous Solution: Ag. Cl is solid & other are aqueous → Kc Heterogeneous Solution: P 4 is solid & PCl 3 is a liquid → both neglected → Kc Gas → Kp

Example 14. 6 Consider the following equilibrium at 295 K: NH 4 HS (s) ⇄ NH 3 (g) + H 2 S (g) Heterogeneous The partial pressure of each gas is 0. 265 atm. Calculate Kp and Kc for the reaction? Solution:
- Slides: 18