Chemical Equilibrium Chapter 13 1 Equilibrium Weve already

Chemical Equilibrium Chapter 13 1

Equilibrium We’ve already used the phrase “equilibrium” when talking about reactions. In principle, every chemical reaction is reversible. . . capable of moving in the forward or backward direction. 2 H 2 + O 2 2 H 2 O Some reactions are easily reversible. . . Some not so easy. . . 2

Equilibrium: the extent of a reaction • In stoichiometry we talk about theoretical yields, and the many reasons actual yields may be lower. • Another critical reason actual yields may be lower is the reversibility of chemical reactions: some reactions may produce only 70% of the product you may calculate they ought to produce. 3

The Concept of Equilibrium • Consider colorless frozen N 2 O 4. At room temperature, it decomposes to brown NO 2: • At some time, the color stops changing and we have a mixture of N 2 O 4 and NO 2. • Chemical equilibrium is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. At that point, the concentrations of all species are constant. • Using the collision model: – as the amount of NO 2 builds up, there is a chance that two NO 2 molecules will collide to form N 2 O 4. – At the beginning of the reaction, there is no NO 2 so the reverse reaction 2 NO 2 (g) N 2 O 4 (g) does not occur. 4
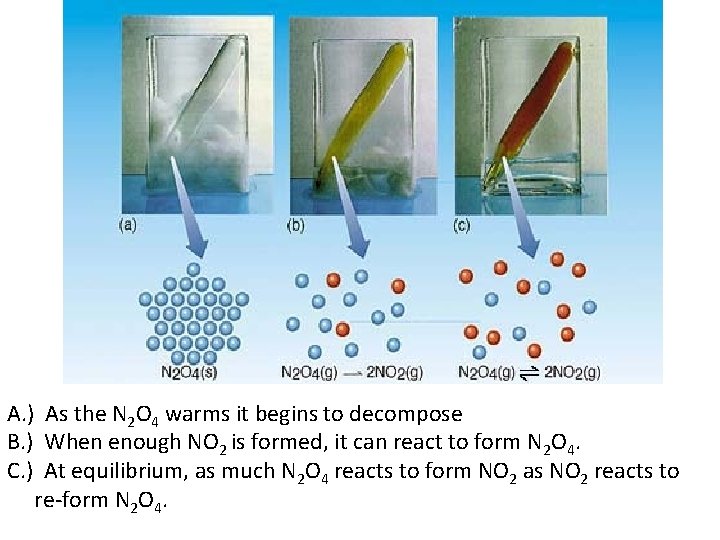
A. ) As the N 2 O 4 warms it begins to decompose B. ) When enough NO 2 is formed, it can react to form N 2 O 4. C. ) At equilibrium, as much N 2 O 4 reacts to form NO 2 as NO 2 reacts to re-form N 2 O 4.
![The Concept of Equilibrium As the reaction progresses – [A] decreases to a constant, The Concept of Equilibrium As the reaction progresses – [A] decreases to a constant,](http://slidetodoc.com/presentation_image/3dfc9f6a24b9fd01bdf8e4f6c8f6e7c8/image-6.jpg)
The Concept of Equilibrium As the reaction progresses – [A] decreases to a constant, – [B] increases from zero to a constant. – When [A] and [B] are constant, equilibrium is achieved. 6

The Equilibrium Constant • No matter the starting composition of reactants and products, the same ratio of concentrations is achieved at equilibrium. • For a general reaction: a. A (g) + b. B (g) c. C (g) + d. D (g) • The value of Kc is the equilibrium constant and the information on the right side is considered the Equilibrium expression. 7

The Equilibrium Constant • Kc is based on the molarities of reactants and products at equilibrium. • We generally omit the units of the equilibrium constant. • Note that the equilibrium constant expression has products over reactants. • For the reverse reaction: Equilibrium expression = reciprocal of the original expression. Kc = 1 Kc 8

The Equilibrium Constant • An equilibrium can be approached from any direction. Forward Reaction Reverse Reaction 9

The Equilibrium Expression • Write the equilibrium expression for the following reaction: 10

For the reaction: CO(g) + 2 H 2 (g) CH 3 OH(g) The equilibrium concentrations are: [Co] = 2. 58 M [H 2] = 0. 280 M [Ch 3 OH] = 2. 93 M Calculate the Kc for this reaction. Calculate the Kc for the reverse reaction

Heterogeneous Equilibria • Reactants and products are present in more than one phase • Molar concentrations of solids and pure liquids are constant. (You can’t find the concentration of something that isn’t a solution!) • We ignore the concentrations of pure liquids and pure solids in equilibrium constant expressions. • The amount of CO 2 formed will not depend greatly on the amounts of Ca. O and Ca. CO 3 present. Kc = [CO 2] 12

The Equilibrium Constant in Terms of Pressure • If KP is the equilibrium constant for reactions involving gases, we can write: • KP is based on partial pressures measured in atmospheres. 13

Large Value for the Equilibrium Constant Kc ˃( 103) • Product favored over reactants. • Reaction proceeds almost to completion • Position of reaction is said to be to the right Small Value for the Equilibrium Constant Kc ˂( 10 -3) • Reactants are favored over products. • Reaction hardly proceeds forward at all. • Position of reaction is said to be to the left. • 10 -3 ˂ Kc ˂ 103 good amounts of both R and P’s

Predicting the Direction of Reaction • We define Q, the reaction quotient, for a reaction at conditions NOT at equilibrium as where [A], [B], [P], and [Q] are molarities at any time. • Q = K only at equilibrium. 15
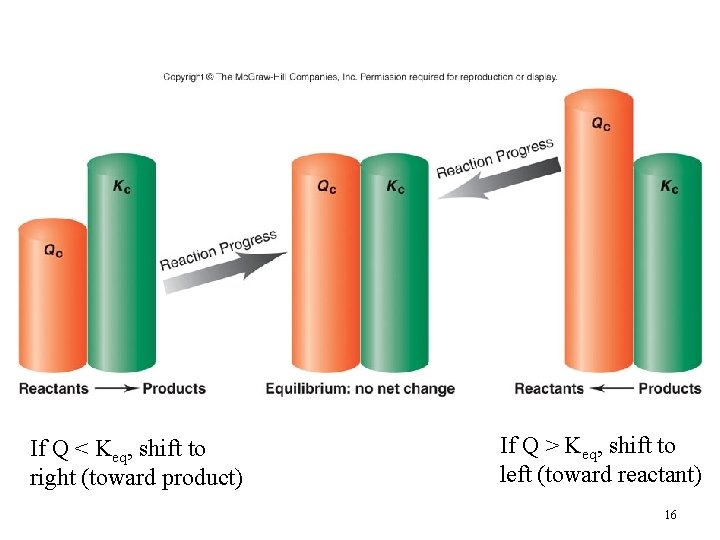
If Q < Keq, shift to right (toward product) If Q > Keq, shift to left (toward reactant) 16

Predicting the Direction of Reaction • Reaction quotient Qc is the same as the equilibrium constant expression except that the concentrations are not necessarily equilibrium values. • If Q > K then the reverse reaction must occur to reach equilibrium (go left) • If Q < K then the forward reaction must occur to reach equilibrium (go right) 17
![Practice Problem Kp = 4. 18 x 10 -9 at 425⁰ C [HBr] = Practice Problem Kp = 4. 18 x 10 -9 at 425⁰ C [HBr] =](http://slidetodoc.com/presentation_image/3dfc9f6a24b9fd01bdf8e4f6c8f6e7c8/image-18.jpg)
Practice Problem Kp = 4. 18 x 10 -9 at 425⁰ C [HBr] = 0. 75 M [H 2] = [Br 2] = 2. 5 x 10 -4 2 HBr (g) H 2 (g) + Br 2 (g) Is the reaction mixture at equilibrium? In which direction will the equilibrium proceed?

Calculating Equilibrium Constants Steps to Solving Problems: 1. Write an equilibrium expression for the balanced reaction. 2. Write an ICE table I =initial concentration C= Change in concentration E = Equilibrium concentration (In constructing the table, define x as the concentration of one of the substances that reacts on going into equilibrium. ) 3. Fill in the given amounts and solve for x. – – Use stoichiometry (mole ratios) on the change in concentration line. Calculate the equilibrium concentrations from the value of x 4. Check your results by substituting them into the equilibrium equation. 19

Practice Problem: The reaction PCl 5 PCl 3 (g) + Cl 2 (g) Kc = 85. 0 at a temperature of 760⁰ C Calculate the equilibrium concentrations of all three species if the initial concentration of PCl 5 is 5. 00 M. Make sure that you use an ICE box and show all your work.

Le Châtelier’s Principle • If you disturb an equilibrium, it will shift to undo the disturbance. • “Shift Happens” • Three types of Equilibrium Stress: ü Changes in concentration of reactants or products. ü Changes in temperature ü Changes in pressure 21

Change in Reactant or Product Concentrations • Adding a reactant or product shifts the equilibrium away from the increase. • Removing a reactant or product shifts the equilibrium towards the decrease. • To optimize the amount of product at equilibrium, we need to flood the reaction vessel with reactant and continuously remove product (Le Châtelier). 22

Effects of Volume and Pressure slide 1 There is almost no effect on reactions that are in aqueous solutions. The story is very different with Gasses! The system shifts to remove gases and decrease pressure. • An increase in pressure favors the direction that has fewer moles of gas. • In a reaction with the same number of product and reactant moles of gas, pressure has no effect. 23

Effects of Volume and Pressure slide 2 • An increase in pressure (by decreasing the volume) favors the formation of colorless N 2 O 4. • The instant the pressure increases, the system is not at equilibrium and the concentration of both gases has increased. • The system moves to reduce the number moles of gas (i. e. the reverse reaction is favored). • A new equilibrium is established in which the mixture is lighter because colorless N 2 O 4 is favored. 24

Effect of Temperature Changes • The equilibrium constant is temperature dependent. • For an endothermic reaction: • H > 0 , heat can be considered as a reactant and is placed on that side. • Adding heat would favor the forward reaction • Removing heat would favor the reverse reaction. • For an exothermic reaction • H < 0 , heat can be considered as a product and is placed on that side. • Adding heat would favor the reverse reaction. • Removing heat would favor the forward reaction. 25

Solubility • The solubility of a substance is the amount of that substance that will dissolve in a given amount of solvent. • A substance is said to be soluble if more than 1 g of that substance dissolves in 100 m. L solvent. • If less than 0. 1 g dissolves in 100 m. L solvent, the substance is said to be insoluble. • Substances with solubility between these limits are called "sparingly soluble“ or slightly soluble.

Solubility Product Principle • Another equilibrium situation to consider is slightly soluble products • Ksp is the solubility product constant • Ksp can be found on a chart at a specific temperature • Since the product is solid on the left side, only the products (ions) are involved in the Ksp expression 27
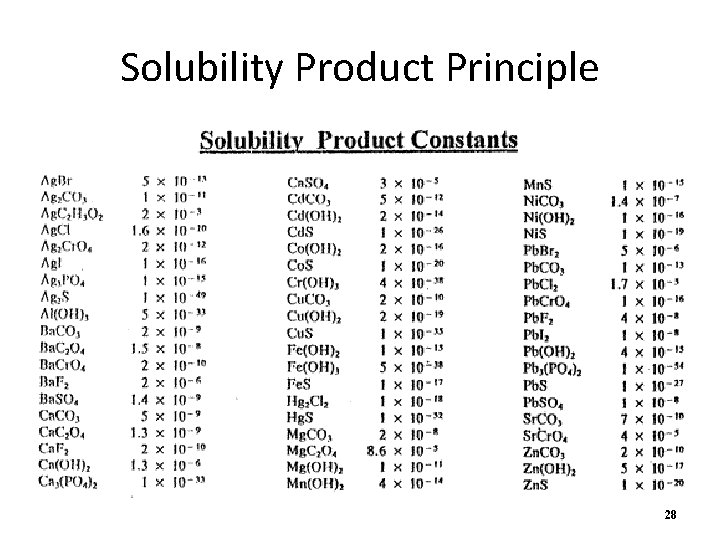
Solubility Product Principle 28

Solubility Practice Problem Find the concentration of ions present in calcium fluoride (in water) and the molar solubility. Ksp = 2 X 10 -10 Ca. F 2(s) --> Ca+2 + 2 F- 1. Write the Ksp expression. 2. Substitute x values into the expression If x = [Ca+2 ], then [F-] = 2 x 3. Solve for X

Solubility Practice Problem Solution 4 x 3 = 2 X 10 -10 x 3 = 5 X 10 -11 x = 3. 68 X 10 -4 [Ca+2 ] = x = 3. 68 X 10 -4 [F-] = 2 x = 7. 37 X 10 -4 Solubility of Ca. F 2 = 3. 68 X 10 -4
- Slides: 30