Chemical Equilibrium A reversible reaction can go in

Chemical Equilibrium A reversible reaction can go in either the forward or reverse directions A + B C + D Equilibrium is dynamic as both the forward and reverse reactions continue At equilibrium the forward and reverse rates are equal

Le Chateliers Principle If a stress is applied to a system at equilibrium, the system adjusts to relieve the stress Co. Cl 42 - + H 2 O H= + Endothermic ( Cooling) Co(H 2 O)62+ + 4 Cl. H= - Exothermic (Heating)

Le Chatelier’s Principle If a stress is applied to a system at equilibrium, the system adjusts to relieve the stress Cr 2 O 72 - + H 2 O Cr. O 42 - + 2 H+

Le Chateliers Principle If a stress is applied to a system at equilibrium, the system adjusts to relieve the stress Fe. Cl 3 + CNS- Fe(CNS)2 - + 3 Cl-

Le Chatelier’s Principle Co. Cl 42 - + H 2 O Co(H 2 O)62+ + H= + 4 Cl. H= - Exothermic (Heating) Endothermic ( Cooling) What happens if the red solution is heated ? Heating is a stress Le Chatelier states the opposing reaction occurs The cooling reaction occurs ie the reverse reaction Turning red into blue
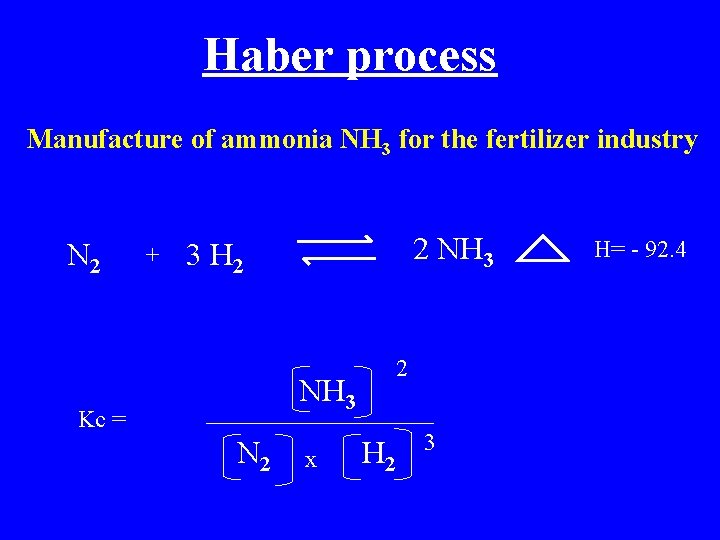
Haber process Manufacture of ammonia NH 3 for the fertilizer industry N 2 + 2 NH 3 3 H 2 2 NH 3 Kc = N 2 x H 2 3 H= - 92. 4

Haber process Manufacture of ammonia NH 3 for the fertilizer industry N 2 + 3 H 2 2 NH 3 H= - 92. 4 Le Chatelier’s principle predicts the yield of NH 3 is maximised by 1. Low temperature 2. High pressure Actual temp used is 500 o. C (as lower temp reduces the rate ) Pressure used is 200 ATM

Contact process Manufacture of Sulfuric acid H 2 SO 4 2 SO 2 + O 2 2 SO 3 H= - 196 Le Chatelier’s principle predicts the yield of SO 3 is maximised by 1. Low temperature 2. High pressure Actual temp used is 450 o. C (as low temp reduces the rate ) Pressure used is 1 ATM
- Slides: 8