Chemical Equilibrium 1 What is chemical Equilibria 2

Chemical Equilibrium 1. What is chemical Equilibria 2. demo 3. What is it 4. Le. Chatelier’s Principle 5. Stresses – Concentration, Pressure, Temperature, Volume 6. Percent Reaction 7. Equilibrium Expression – Ke, Law of Mass Action

1. Demo – textbook pg 423 Shakin’ the Blues 250 m. L of water, add 6 drops methylene blue and 5. 0 g of potassium hydroxide, and 3. 0 g glucose or dextrose to 250 m. L flask. Stir mixture until solids have dissolved Stopper the flask and set it on the bench. Observe the colour of the solution Shake solution, note changes. Set on table and leave it standing until another change is noticed Repeat 2 previous steps. Compare to a reaction like HCl and Zn which once complete is done.

Chemical Equilibria a. A + b. B c. C + d. D *when the rate of the forward = the rate of the reverse in Chemical Equilibrium OR dynamic equilibrium we say the system is This means … 1. A, B, C and D are all present in the container 2. Concentration of all species remains constant. 3. At the macroscopic level (visual) nothing seems to be going on (pressure, temp, colour) 4. At the microscopic level (atomic) both reactions are taking place at the same rate (forward and reverse) It does NOT mean… a. That the concentrations of all the species are the same.
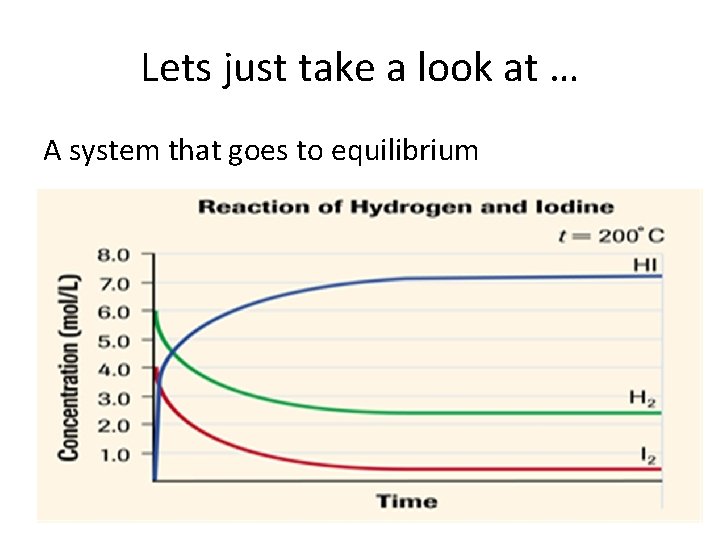
Lets just take a look at … A system that goes to equilibrium

Driving either the forward Rxn or reverse Rxn to proceed when a system is at equilibrium Le Chatelier`s Principle When a stress is applied to a System at equilibrium, the System WILL react so as to relieve that stress one of the reactions will take place either the FORWARD or REVERSE

Quick Terminology Rxn that takes place We say equilibrium Favored Direction shifts to the Start to produce Forward Reaction Right Forward Products Reverse Reaction Left Reverse Reactants
![The Stressors: 1. Concentration Stress If you increase the [ ] of the reactants/products, The Stressors: 1. Concentration Stress If you increase the [ ] of the reactants/products,](http://slidetodoc.com/presentation_image_h2/666cf9b6f4c8670722d306e91f58e10e/image-7.jpg)
The Stressors: 1. Concentration Stress If you increase the [ ] of the reactants/products, the system will react to decrease the [ ] of the reactants/products which will cause an increase in the [ ] of the products/reactants Eg. : N 2 (g) + 3 H 2 (g) 2 NH 3 (g) ΔHR= -300 k. J Stress:
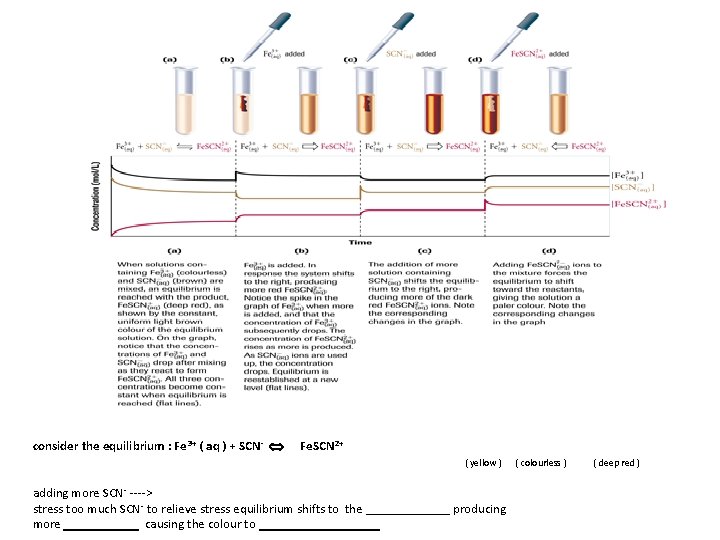
consider the equilibrium : Fe 3+ ( aq ) + SCN- Fe. SCN 2+ ( yellow ) adding more SCN- ----> stress too much SCN - to relieve stress equilibrium shifts to the _______ producing more causing the colour to _________ ( colourless ) ( deep red )

Consider the equ. SO 2(g) + 1/2 O 2 (g) SO 3(g) In what direction would the equation shift if More O 2 is pumped in : A: to the right B: to the left Some SO 2 is removed from the system A: to the right B: To the left Consider the equ. SO 2(g) + 1/2 O 2 (g)�SO 3(g), If SO 3 is removed from the system, the concentration of O 2 will increase A: True B: False

2. Temperature Stress If you add heat ( T) ‘ENDO’ rxn is favoured If you remove heat ( T) “EXO” rxn is favoured Ex. N 2 (g) + 3 H 2 (g) 2 NH 3 (g) ΔHR= -300 k. J a) Label forward / reverse as endo or exo
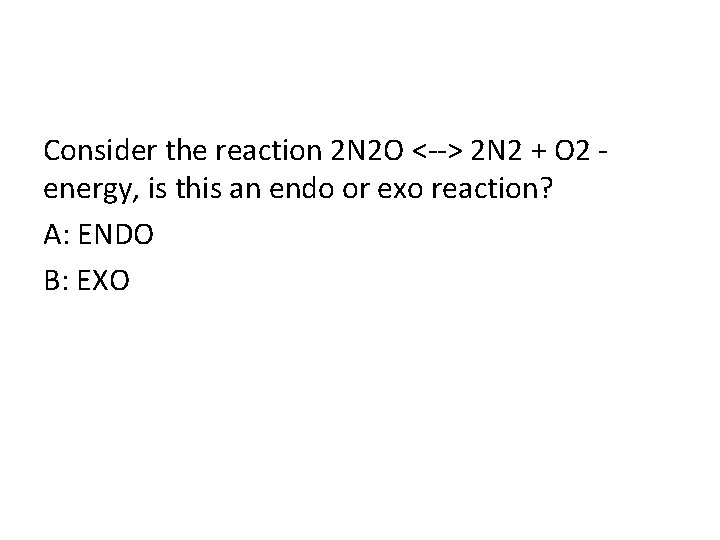
Consider the reaction 2 N 2 O <--> 2 N 2 + O 2 energy, is this an endo or exo reaction? A: ENDO B: EXO

Plickers: Consider the reaction N 204 + energy <--> 2 NO 2. This is an exothermic reaction. A: True B: False
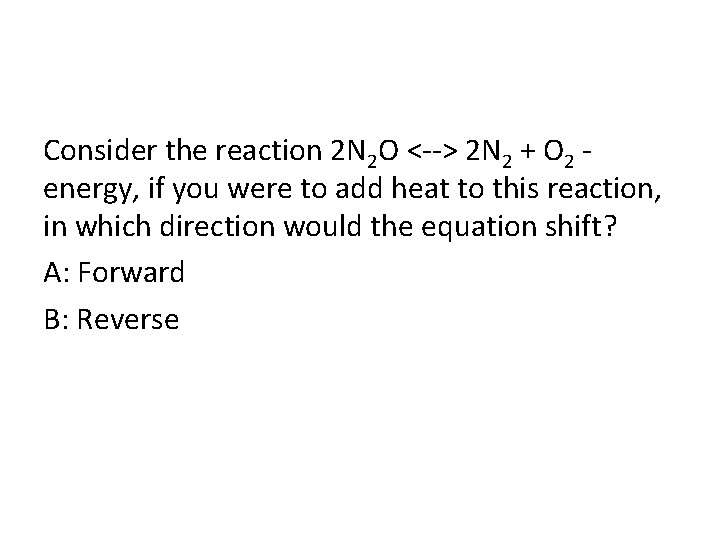
Consider the reaction 2 N 2 O <--> 2 N 2 + O 2 energy, if you were to add heat to this reaction, in which direction would the equation shift? A: Forward B: Reverse
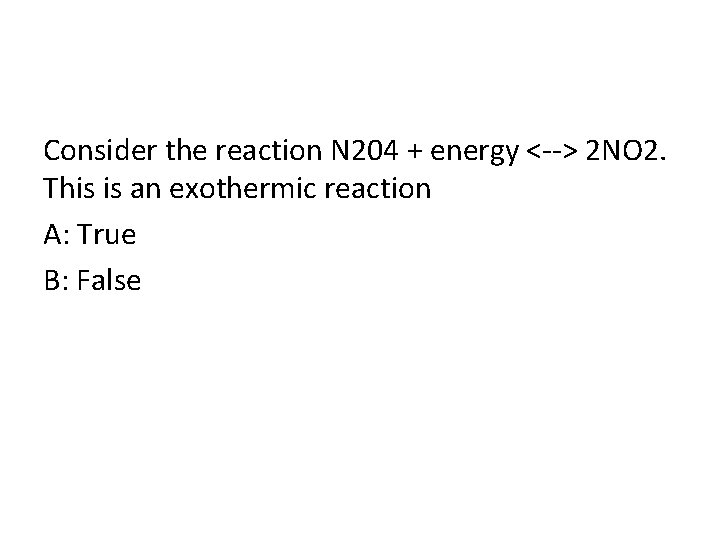
Consider the reaction N 204 + energy <--> 2 NO 2. This is an exothermic reaction A: True B: False

Consider the reaction N 2 O 4 + energy <--> 2 NO 2. , in which direction would the reaction shift and what would happen to the concentration of the product if the reaction was cooled? A: Right, Increase B: Right, Decrease C: Left, Increase D: Left, Decrease

Consider the equation N 2 O 4 (g) + 59 k. J 2 NO 2 (g) (colourless) (brown/reddish) - increase in temp. ; stress is too much energy (_______), to relieve the stress equ. shifts to the using up some of the energy (__________) ; reddish brown colour ________ since there is an increase in conc. and a decrease in conc.

3. Pressure Stress If you increase pressure the system will react to decrease the pressure (goes toward the side with the lower # of gas phase particles) Eg. : N 2 (g) + 3 H 2 (g) 2 NH 3 (g) ΔHR= -300 k. J Stress: Increase in P = shift to the side with least # of gas moles to relive the stress Decrease in P = shift to the side with more moles of gas to relieve the stress

Discuss with a partner, then vote as a class – explain your reasons. Click for the answer, Then click again for the explanation. Consider the reaction Eg. N 2 O 4 (g) + energy 2 NO 2 (g), how would the system react To being moved from a 1 L container to a 2 L container? A: equilibrium will move in the forward direction B: equilibrium will move in the reverse direction An increase in volume = a decrease in pressure so the system will shift towards the side With more moles of gas (this will increase the pressure again, reducing the stress of Decreased pressure due to increased volume)

4. Volume Stress: Translate to a pressure stress (gr. 11 V/P relationships) increase V = decrease P decrease V= increase P 5. Addition of a Catalyst: NO EFFECT, just speeds up reaction 6. Addition of an INERT gas: not altogether sure yet

Le. Chatelier’s Graphical Representation • Follow the video and copy the example into your book

Four types of equilibria 1. Gaseous 2. Solubility 3. Phase 4. Acid/Base Use your textbooks to further explain what is happening microscopically during solubility equilibrium and phase equilibriums. Use examples and pictures if necessary. Textbook questions 1 -3, 5 on page. 428

Use your textbook to outline the difference and significance between using an open and closed system (still on your paper).

Work on Le. Chatelier Worksheet provided.

Law of Mass Action At equilibrium ALL species are present … the concentrations are constant Consider: a. A + b. B < -- > c. C + d. D Equilibrium Law Expression: ** only if (g) or (aq) ** if (l) or (s) sub in the #1
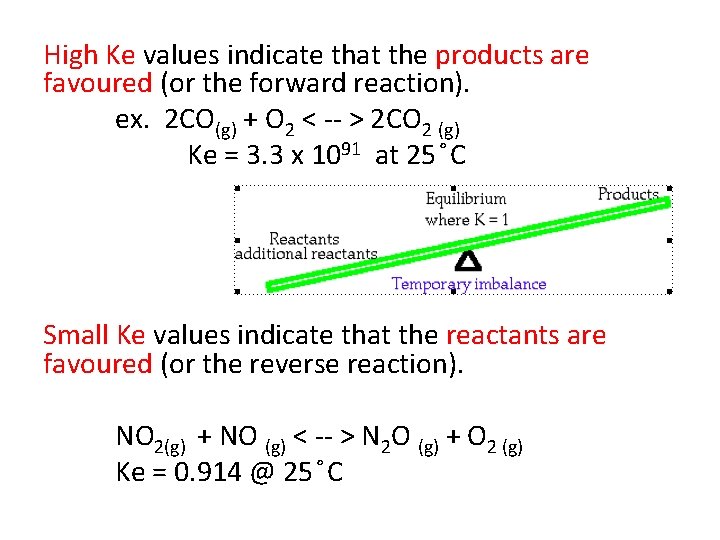
High Ke values indicate that the products are favoured (or the forward reaction). ex. 2 CO(g) + O 2 < -- > 2 CO 2 (g) Ke = 3. 3 x 1091 at 25 C Small Ke values indicate that the reactants are favoured (or the reverse reaction). NO 2(g) + NO (g) < -- > N 2 O (g) + O 2 (g) Ke = 0. 914 @ 25 C
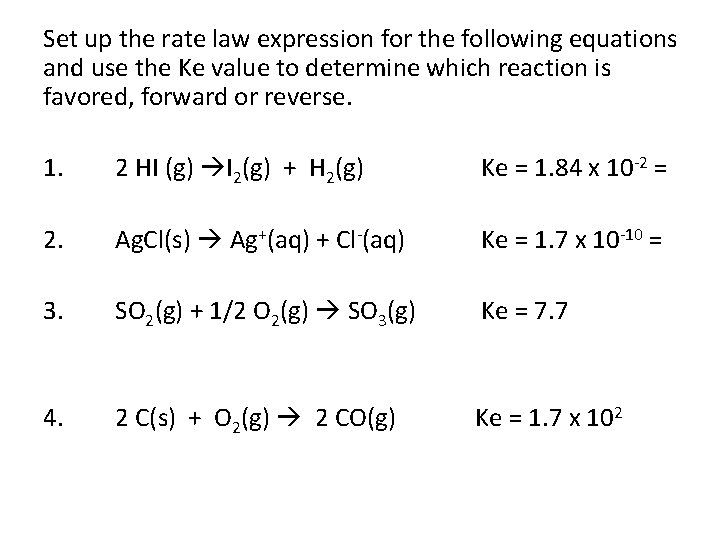
Set up the rate law expression for the following equations and use the Ke value to determine which reaction is favored, forward or reverse. 1. 2 HI (g) I 2(g) + H 2(g) Ke = 1. 84 x 10 -2 = 2. Ag. Cl(s) Ag+(aq) + Cl-(aq) Ke = 1. 7 x 10 -10 = 3. SO 2(g) + 1/2 O 2(g) SO 3(g) Ke = 7. 7 4. 2 C(s) + O 2(g) 2 CO(g) Ke = 1. 7 x 102

Go to smart board for examples

Gaseous/ Chemical Equilibria Go to Smart Board
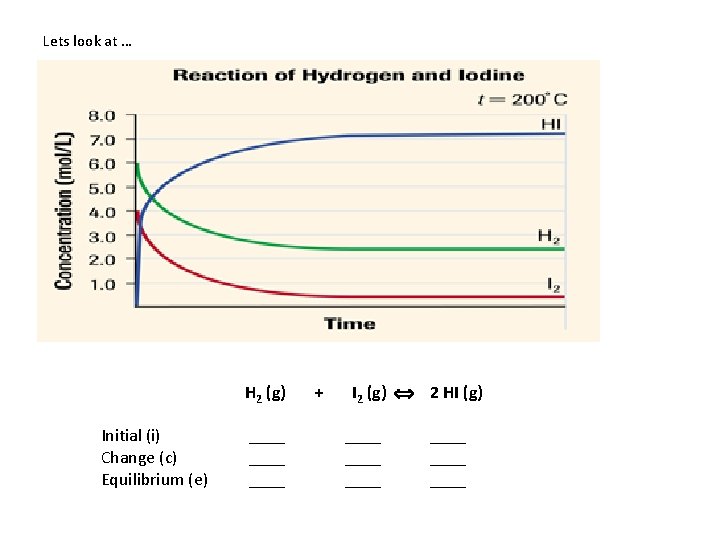
Lets look at … H 2 (g) Initial (i) Change (c) Equilibrium (e) ____ + I 2 (g) 2 HI (g) ____ ____

Solubility Equilibria smartboard
![ACID/BASE EQUILIBRIA Preview/Review 1. Determining p. H 1. p. H = -log[H+1 ] 2. ACID/BASE EQUILIBRIA Preview/Review 1. Determining p. H 1. p. H = -log[H+1 ] 2.](http://slidetodoc.com/presentation_image_h2/666cf9b6f4c8670722d306e91f58e10e/image-31.jpg)
ACID/BASE EQUILIBRIA Preview/Review 1. Determining p. H 1. p. H = -log[H+1 ] 2. p. OH = log [OH-1] 3. p. H + p. OH = 14 2. Defn’s monoprotic/ monobasic – one diprotic/dibasic = 2 polyprotic/polybasic =

. 3. Neutralization # moles H+1 = # moles OH-1 MH+1 VH+1 = MOH-VOH# moles H+ = MOH-VOH 4. Acid / Base Equation

Something a little different… determining p. H of a mixture of an acid and base 1. Calculate the # _______ and # of ______ (Mx. V = n) (BECAREFUL – write out equation) 2. Identify the one in ________. 3. Calculate [ _________] = =
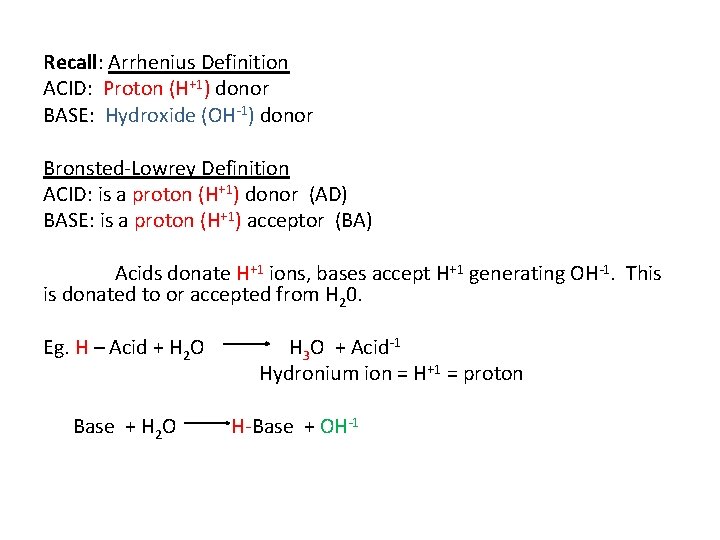
Recall: Arrhenius Definition ACID: Proton (H+1) donor BASE: Hydroxide (OH-1) donor Bronsted-Lowrey Definition ACID: is a proton (H+1) donor (AD) BASE: is a proton (H+1) acceptor (BA) Acids donate H+1 ions, bases accept H+1 generating OH-1. This is donated to or accepted from H 20. Eg. H – Acid + H 2 O Base + H 2 O H 3 O + Acid-1 Hydronium ion = H+1 = proton H-Base + OH-1

Strong Acid (SA): acid that ionizes 100% no equilibria eg. HCl H+ + Cl-1 HCl, HNO 3, H 2 SO 4, HBr, HI, HCl. O 4 Strong Base (SB): base that ionizes 100% eg. Na. OH Na+ + OH-1 • Li. OH, Na. OH, KOH, Rb. OH, Cs. OH
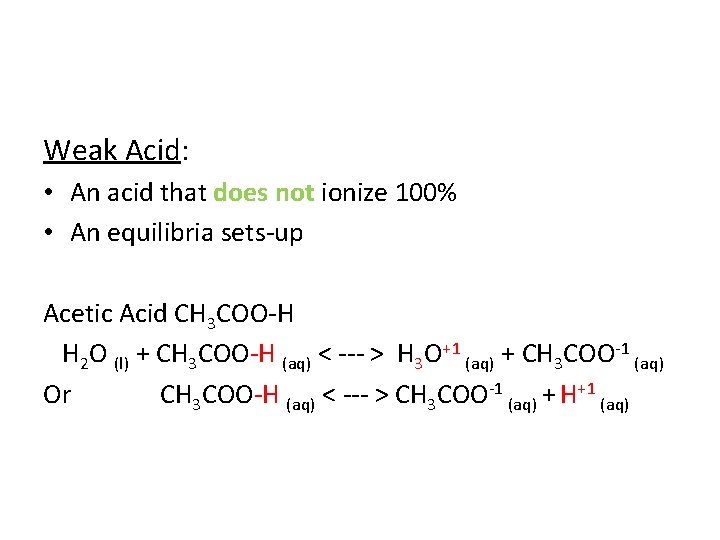
Weak Acid: • An acid that does not ionize 100% • An equilibria sets-up Acetic Acid CH 3 COO-H H 2 O (l) + CH 3 COO-H (aq) < --- > H 3 O+1 (aq) + CH 3 COO-1 (aq) Or CH 3 COO-H (aq) < --- > CH 3 COO-1 (aq) + H+1 (aq)
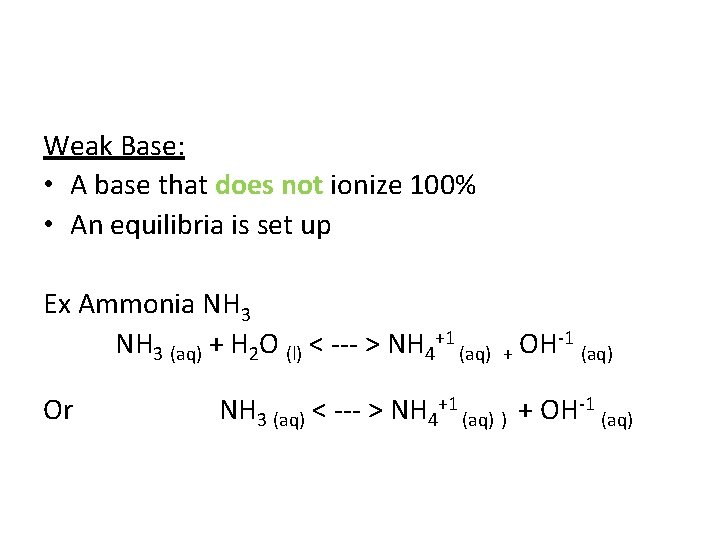
Weak Base: • A base that does not ionize 100% • An equilibria is set up Ex Ammonia NH 3 (aq) + H 2 O (l) < --- > NH 4+1 (aq) + OH-1 (aq) Or NH 3 (aq) < --- > NH 4+1 (aq) ) + OH-1 (aq)
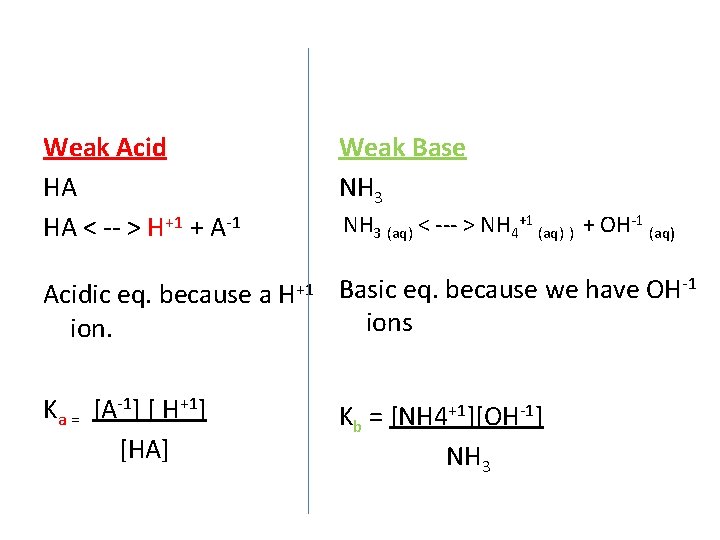
Weak Acid HA HA < -- > H+1 + A-1 Weak Base NH 3 (aq) < --- > NH 4+1 (aq) ) + OH-1 (aq) -1 Acidic eq. because a H+1 Basic eq. because we have OH ions ion. Ka = [A-1] [ H+1] [HA] Kb = [NH 4+1][OH-1] NH 3

Handout – do acidic/basic equilibriums Then to smart board for determining p. H of weak acid or base. Do example then in partners try the 2 examples on white page then take up.

Salt Hydrolysis • Salts are strong electrolytes that completely ionize in water, the cations or anions created MAY react with water to create a solution that is acidic, basic or neutral • A neutral salt solution generally occurs when a salt containing an anion or cation of a strong acid or base (HSO 4 -) is dissolved in water – there is NO reaction therefore no p. H sets up ex. Na. NO 3 Na+ + NO 3 - The NO 3 does not react with water to set up an equilibrium
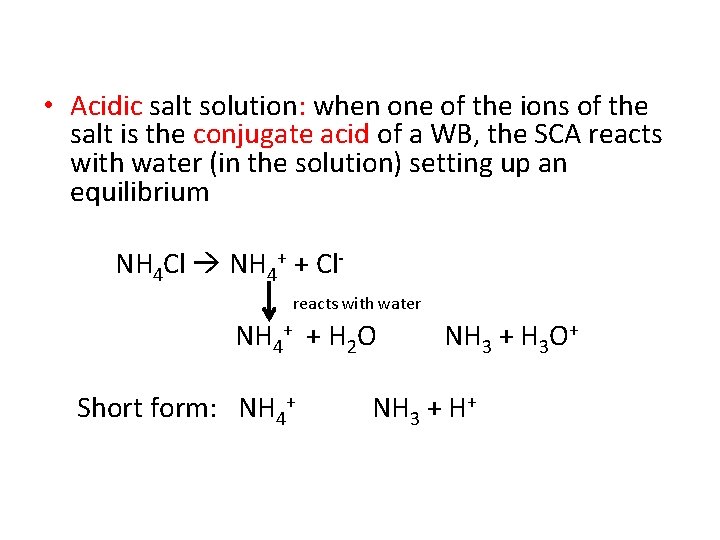
• Acidic salt solution: when one of the ions of the salt is the conjugate acid of a WB, the SCA reacts with water (in the solution) setting up an equilibrium NH 4 Cl NH 4+ + Clreacts with water NH 4+ + H 2 O Short form: NH 4+ NH 3 + H 3 O+ NH 3 + H+
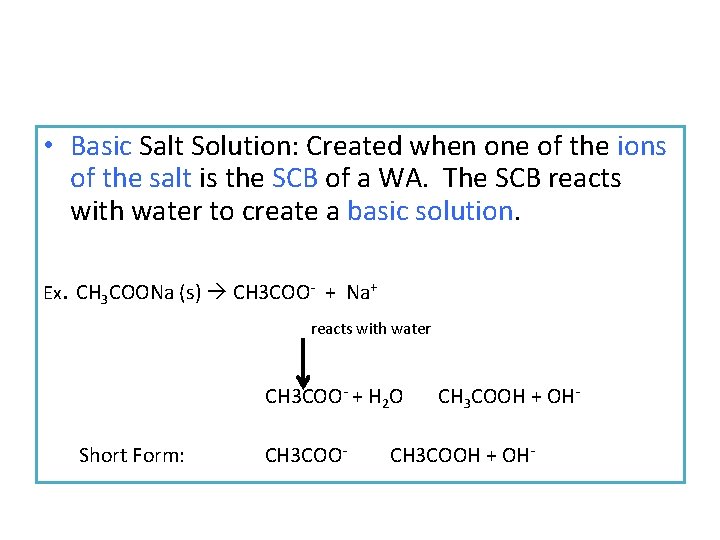
• Basic Salt Solution: Created when one of the ions of the salt is the SCB of a WA. The SCB reacts with water to create a basic solution. Ex. CH 3 COONa (s) CH 3 COO- + Na+ reacts with water CH 3 COO- + H 2 O Short Form: CH 3 COO- CH 3 COOH + OH-

Titrations • Mixing an acid and base until an endpoint is reached. The p. H of the endpoint is based on the indicator being used. ACID + BASE SALT + H 2 O • If the salt produces a strong conjugate as one of its ions, hydrolysis takes place and it will create an acidic/basic solution instead of a neutral one Ex. HCl + NH 3 NH 4 Cl + H 2 O

Buffers in the Blood n n n n The p. H of blood is 7. 35 – 7. 45 Changes in p. H below 6. 8 and above 8. 0 may result in death The major buffer system in the body fluid is H 2 CO 3/HCO 3 Some CO 2, the end product of cellular metabolism, is carried to the lungs for elimination, and the rest dissolves in body fluids, forming carbonic acid that dissociates to produce bicarbonate (HCO 3 -) and hydronium (H 3 O+) ions. More of the HCO 3 - is supplied by the kidneys. CO 2 + H 2 O ↔ H 2 CO 3 + H 2 O ↔ H 3 O+ + HCO 3 - 44
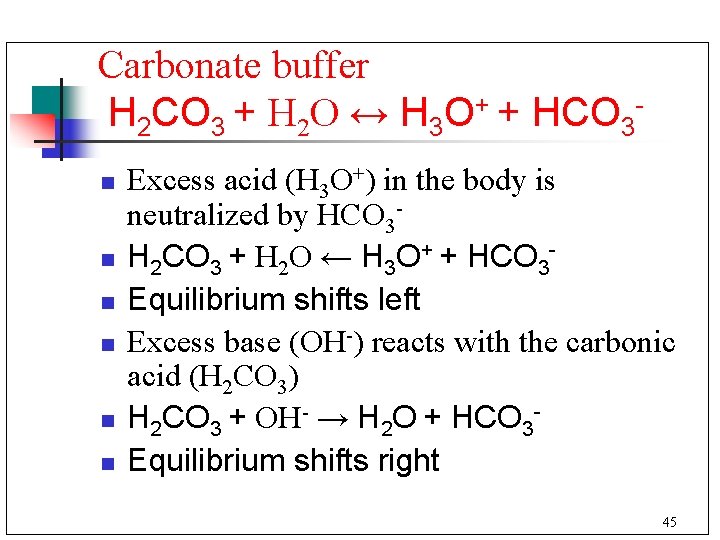
Carbonate buffer H 2 CO 3 + H 2 O ↔ H 3 O+ + HCO 3 n n n Excess acid (H 3 O+) in the body is neutralized by HCO 3 H 2 CO 3 + H 2 O ← H 3 O+ + HCO 3 Equilibrium shifts left Excess base (OH-) reacts with the carbonic acid (H 2 CO 3) H 2 CO 3 + OH- → H 2 O + HCO 3 Equilibrium shifts right 45

The effectiveness of the blood buffer n n n If the p. H of 100 m. L of distilled water is 7. 35 and one drop of 0. 05 M HCl is added, the p. H will change to 7. 00. To change 100 m. L of “normal” blood from p. H of 7. 35 to 7. 00, approximately 25 m. L of 0. 05 M HCl is needed. With 5. 5 L of blood in the average body, more than 1300 m. L of HCl would be required to make the same change in p. H. 46

Importance of the bicarbonatecarbonic acid buffering system 1. 2. 3. H 2 CO 3 dissociates into CO 2 and H 2 O, allowing H 3 O+ to be eliminated as CO 2 by the lungs Changes in PCO 2 modify the ventilation rate HCO 3 - concentration can be altered by kidneys 47

Interpretation of Clinical Blood Gas Data H 2 O CO 2 + H 2 O ↔ H 2 CO 3 ↔ H 3 O+ + HCO 3 - n n n The concentration of carbonic acid in the body is associated with the partial pressure of CO 2. When CO 2 level rises, producing more H 2 CO 3, the equilibrium produces more H 3 O+, which lowers the p. H – acidosis. Decreasing of CO 2 level due to a hyperventilation, which expels large amounts of CO 2, leads to a lowering in the partial pressure of CO 2 below normal and the shift of the equilibrium from H 2 CO 3 to CO 2 and H 2 O. This shift decreases H 3 O+ and raises blood p. H – alkalosis. 48

Respiratory Acidosis: CO 2 ↑ p. H ↓ n n n Symptoms: Failue to ventilate, suppression of breathing, disorientation, weakness, coma Causes: Lung disease blocking gas diffusion (e. g. , emphysema, pneumonia, bronchitis, and asthma); depression of respiratory center by drugs, cardiopulmonary arrest, stroke, poliomyelitis, or nervous system disorders Treatment: Correction of disorder, infusion of bicarbonate 49

Respiratory Acidosis: CO 2 ↑ p. H ↓ n n n Symptoms: Failue to ventilate, suppression of breathing, disorientation, weakness, coma Causes: Lung disease blocking gas diffusion (e. g. , emphysema, pneumonia, bronchitis, and asthma); depression of respiratory center by drugs, cardiopulmonary arrest, stroke, poliomyelitis, or nervous system disorders Treatment: Correction of disorder, infusion of bicarbonate 50

Metabolic (Nonrespiratory) Alkalosis: H+ ↓ p. H ↑ n n n Symptoms: Depressed breathing, apathy, confusion Causes: Vomiting, diseases of the adrenal glands, ingestions of access alkali Treatment: Infusion of saline solution, treatment of underlying diseases 51

n Buffers are also important in many consumer, commercial, and industrial applications. Fermentation and the manufacture of antibiotics require buffering to optimize yields and to avoid undesirable side reactions. The production of various cheeses, yogurt, and sour cream are very dependent on controlling p. H levels, since an optimum p. H is needed to control the growth of micro-organisms and to allow enzymes to catalyze fer- mentation processes. Sodium nitrite and vinegar are widely used to preserve food; part of their function is to prevent the fermentation that takes place only at certain p. H values. 52
- Slides: 52